This content is for U.S. healthcare professionals only.
Indications and Usage
DURYSTA™ (bimatoprost intracameral implant) is indicated for the reduction of intraocular pressure (IOP) in patients with open angle glaucoma (OAG) or ocular hypertension (OHT).
INTRODUCTION
DURYSTA (bimatoprost intracameral implant) (Allergan, an AbbVie company) is the first FDA-approved sustained-release, biodegradable, intracameral implant for the reduction of intraocular pressure in patients with ocular hypertension or open-angle glaucoma. It provides a sustained release of bimatoprost directly to the targeted tissue for several months. DURYSTA is a physician-administered medication, and thus will have benefit verification, ordering, and billing requirements that differ from traditional pharmacy products. I. Paul Singh, MD, spoke with Fiaz Zaman, MD, FACS; Nathan Radcliffe, MD; Davinder Grover, MD, MPH; Richard A. Adler, MD, FACS; and Kasey Siters, COA, OSC, to discuss how they will implement this first-in-class treatment into their practices.
I. Paul Singh, MD: Having a pharmaceutical treatment modality that is administered by the physician without the patient having to administer the medication on their own for several months is very exciting. When a pharmaceutical treatment comes to market, there frequently are questions regarding how accessible it will be for our patients. Because a physician is administering the drug, DURYSTA is a procedure-based treatment covered as a medical benefit, not a pharmacy benefit. This means that it is a medical benefit covered under Medicare Part B, not Medicare Part D. As a medical benefit, DURYSTA was covered by traditional Medicare when it was approved. It is different from retail pharmacy products that fall under Part D, as well as devices that are covered under T-codes or Category III codes. How does that classification as a Part B benefit impact which patients you select to receive DURYSTA?
Nathan Radcliffe, MD: Having a therapy that is a medical benefit covered by traditional Medicare from day 1 helps ensure access to the medication I see fit for patients.
Davinder Grover, MD, MPH: We have a lot of patients with Medicare Advantage plans, which follow Medicare coverage guidelines with possible restrictions. It’s important to note that Medicare Advantage plans can place restrictions for use of a product (e.g., prior authorization), but they are required to provide the same coverage as traditional Medicare. Medicare covers DURYSTA now for on-label use with no prior authorization. This really helps streamline the process for us with our Medicare patients. With DURYSTA, we will have a subset of Medicare patients for whom we can have confidence in the coverage for on-label use. The situation may be different with commercial payers specific to the patients’ plan. It’s important to remember to run a benefit verification, as DURYSTA has coverage on a variety of commercial plans.
Dr. Singh: There are two codes needed when billing for DURYSTA: a procedure code (CPT code) and a drug code (HCPCS code), commonly referred to as a J-code (Figure 1). The CPT code for DURYSTA, 66030, is an established Category I code for injecting medication into the anterior chamber. If you administer DURYSTA into both eyes on the same date of service, the standard multiple procedure rules apply. The J-code is for the drug; upon approval by the FDA, DURYSTA was assigned the miscellaneous code, allowing physicians to bill for DURYSTA right away. A product-specific J-code has been established and is valid for dates of service on or after October 1, 2020, for all sites of service. This product-specific code is a Level II HCPCS code and is J7351, “Injection, bimatoprost, intracameral implant, 1 µg.” It’s important to note that the billing description for DURYSTA is 1 µg, yet it is a 10-µg implant, so you need to bill J7351 as 10 units. When offices are checking a patient’s benefits, they will need to check for both the CPT code and the J-code. Also include J7351 along with the CPT code on the CMS-1500 form, which is something you may not be used to doing. What do you think will be most challenging for practices new to J-codes?
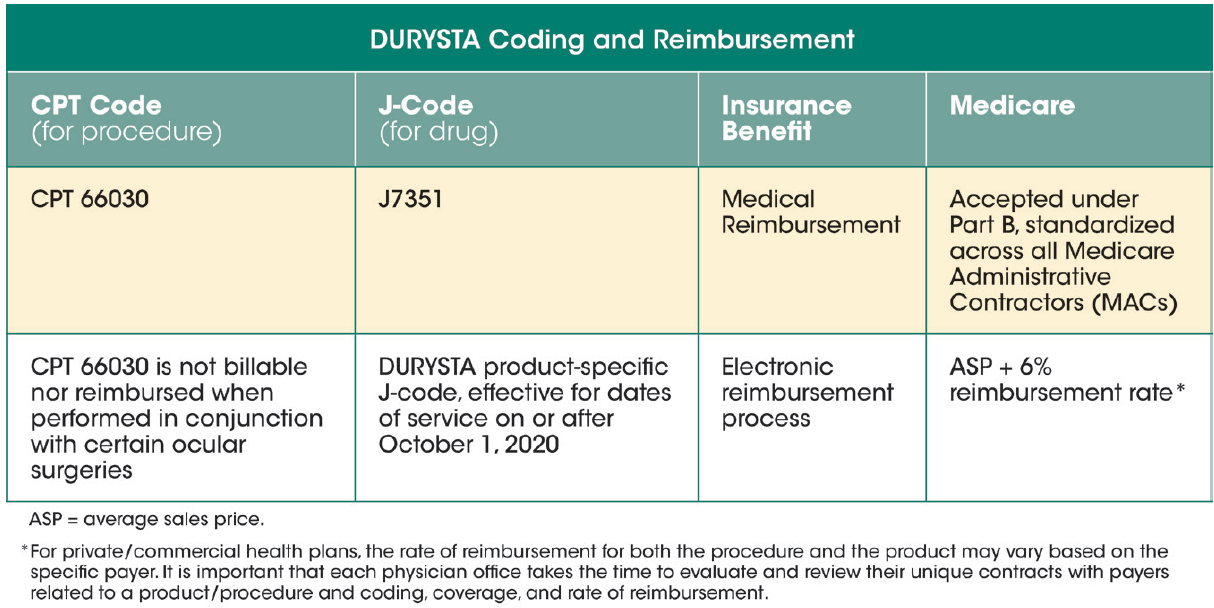
Figure 1. There are two codes needed when billing for DURYSTA: a procedure code (CPT code) and a drug code (HCPCS code), commonly referred to as a J-code.
Richard A. Adler, MD, FACS: I do my own coding, but other practices may use a scribe or have another system. How easy the transition is to start offering DURYSTA will depend on who is doing the billing and how easy it is to update the electronic medical or health record system.
Dr. Singh: It’s important for us to make sure our patients understand that there will be a cost to them, no matter how beneficial DURYSTA will be for them. Traditional Medicare will pay for 80% of the cost of DURYSTA. If a patient has supplemental insurance, it may be covered 100%, but that will depend on the plan. The benefit verification will also evaluate the coverage of the supplemental and secondary insurance. The real point here is that for those Medicare patients with supplemental or secondary insurance, we may be able to provide DURYSTA with low out-of-pocket costs. The Allergan EyeCue hub can assist with insurance verifications and patient out-of-pocket information (see Your Complete Resource for DURYSTA Reimbursement and Access). They also have a DURYSTA copay savings program for eligible commercially insured patients. Offices can go to DurystaHCP.com for further details on reimbursement resources and patient copay support for DURYSTA.
Do you foresee yourselves administering DURYSTA in both eyes on the same day, or do you think you will see how one eye responds prior to administering in the second eye?
YOUR COMPLETE RESOURCE FOR DURYSTA REIMBURSEMENT AND ACCESS
Allergan EyeCue is a comprehensive resource that provides 24/7 reimbursement assistance through one convenient online portal, to help ensure that your appropriate patients have access to DURYSTA. Through Allergan EyeCue, offices can:
- Conduct a benefit verification
- Request prior authorization support
- Access information about billing and coding
- Enroll eligible patients in the DURYSTA Savings Program
- Request claims assistance and appeals support
- Identify appropriate specialty pharmacy, if required by payer, and coordinate enrollment
Allergan EyeCue
- US based, with coast-to-coast coverage
- Staffed by dedicated agents
- 24/7 portal access
- Available by phone: Monday–Friday, 9 AM–8 PM ET
To register, visit DurystaHCP.com and navigate to the Reimbursement Section. Click on the "Go to Allergan EyeCue" link to register. Your Allergan Reimbursement Business Advisor can also help you enroll.
Fiaz Zaman, MD, FACS: I am starting with one eye until I get comfortable and start seeing results from my own patients. Then I will administer in the second eye. Similar to other procedures, I like to see how a patient responds in one eye before moving to repeat the procedure in the second eye.
Dr. Singh: In our practice, we have now started to implant DURYSTA in both eyes on the same day to decrease the number of times the patient has to come back to the office.
Dr. Adler: I feel I can present this similarly to how I present a cataract procedure. First we treat one eye, make sure nothing unexpected occurs, and then we treat the other eye.
ACQUIRING DURYSTA
Dr. Singh: When we administer DURYSTA to our patients will also be determined by how we decide to manage our supply. There are two ways to acquire DURYSTA. One is the buy-and-bill model, and the other is to use a specialty pharmacy (Figure 2). Traditional Medicare will require practices to use a buy-and-bill model. For my practice, the buy-and-bill model seems to be the most straightforward. You purchase the DURYSTA in advance, you use it as needed, and then you fill out the paperwork as you use it. You just need to remember to include the J-code along with the CPT code when you submit the 1500 form. The billing process is really the same as with other procedures. However, you do need some office process in place to purchase, log, and reconcile inventory with what you’ve purchased. Remember, Medicare Advantage and commercial payers may allow, or even require, the use of a specialty pharmacy.
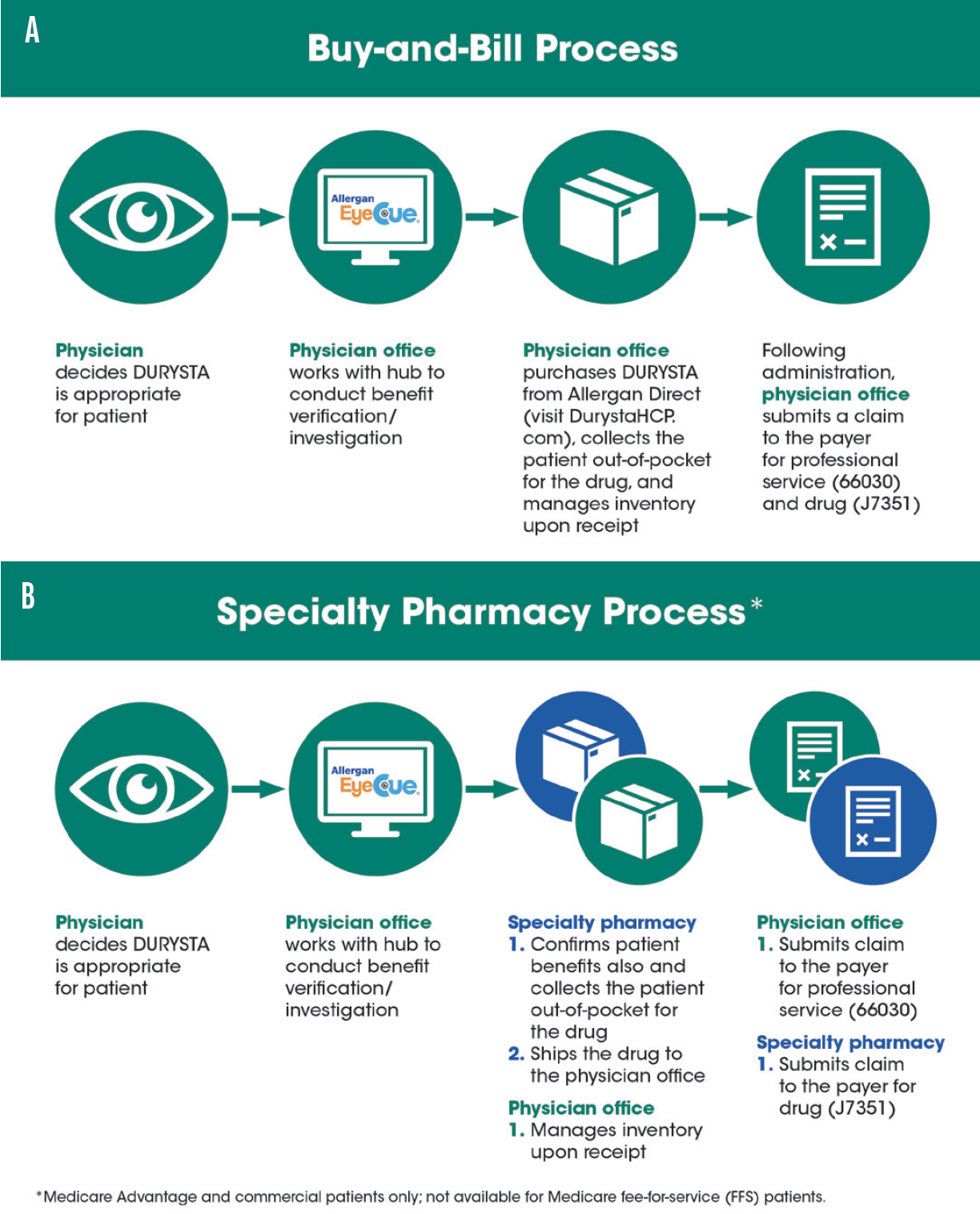
Figure 2. There are two ways to acquire DURYSTA. One is the buy-and-bill model (A), and the other is to use a specialty pharmacy (B).
Kasey Siters, COA, OSC: We have used specialty pharmacies for other physician-administered drugs, and the patient’s insurance determines which one you use. One advantage is that the pharmacy procures DURYSTA and bills the insurance company for the J-code. Then the pharmacy ships the drug to the office. This relieves the staff from having to procure the drug, but it does require verifying if the patient’s insurance is active and coordinating between the specialty pharmacy and the patient. Allergan has a specialty pharmacy network that works closely with their Allergan EyeCue hub, which can help streamline the process.
Dr. Adler: As we think about different practices, I believe that the specialty pharmacy option will be particularly appealing to the general ophthalmologist or a clinician who may not administer a large number of implants. However, most patients will likely be traditional Medicare patients at first. Luckily, it is possible to purchase only as much as you need and not have to maintain inventory.
PRACTICE INFRASTRUCTURE
Dr. Singh: Let’s move on to how DURYSTA administration will fit into practice flow. At a minimum, we need aseptic conditions, magnification, and the head stabilized. The full administration instructions are included in the accompanying Prescribing Information. Do you envision yourselves administering DURYSTA in your office in a minor operating room, or at an ambulatory surgery center? Also, will you schedule these patients together in blocks, or will you spread them throughout your normal day?
Dr. Zaman: It is most likely that our practice will mimic our retina colleagues and administer DURYSTA in the office. Participating in the clinical trials, we performed the procedure in the minor operating room. Initially, I will integrate these appointments into my regular clinic day. As I get more DURYSTA patients, then I will probably schedule them in blocks where I can have staff properly lined up.
Dr. Adler: One of the keys to success with DURYSTA is going to be the ability to effectively work it into the flow of a practice. We have topical hypotensive drops that work well, so there needs to be a distinct benefit for the patient to move to a different treatment. I think the opportunity to administer in the office will have the greatest appeal. Most doctors will not want to schedule this procedure separately and go to another room that is perhaps shared with another doctor. I don’t see myself creating blocks of time specifically for DURYSTA administration, but I do believe I will set a separate appointment for the procedure rather than try to fit it into an existing visit. It will take time to have the discussion and acquire patient consent, and I feel that may take a little bit too much time to squeeze into the existing visit.
Dr. Radcliffe: As glaucoma specialists, we are frequently reforming anterior chambers, moving tubes and intraocular lenses, and sometimes doing iridectomies in the office. For me, the office is the natural place to administer DURYSTA. I have already started introducing DURYSTA to my suitable patients, and enough of them have expressed interest in having it immediately that I will administer it once I have received consent and checked the insurance. I have two practices. At one, I perform selective laser trabeculoplasty on the spot, and at the other, I schedule it for a few weeks later. I think that my patients prefer to just get it done then and there. It does disrupt flow a little bit.
Mrs. Siters: In our experience with other injections, we typically keep them intermixed into the schedule. We have the discussion with the patient, provide them the information, tell them to think it over, and then schedule them to come back later. This keeps the clinic flow efficient. Sometimes patients feel very confident about an injection and want to get it on the spot, and if it’s approved by their insurance, the physicians will go ahead and get it done. This is particularly true if the patient has to travel to get to us or has other restrictions. Patients feel like they are being taken care of, and they don’t have to come back for another visit to receive the treatment.
Dr. Singh: Most likely, I will also mix same-day administration with scheduling a future appointment. Having that flexibility is a real benefit and allows me to tailor to what is best for individual patients. How we decide to schedule administration of DURYSTA will also influence how we organize the clinic infrastructure. Are you going to assign specific tasks to different personnel in your staff, such as ordering, checking benefits, scheduling, and assisting? Do you do that now with other procedures?
Mrs. Siters: We will have the front office verify eligibility. I already handle all ordering of medical supplies, so that will now include DURYSTA. I will have the scribes fully trained to be able to assist, set up, and bill. They will also be able to help educate the patient if there are any questions. That is typically what we do with all of our procedures, and it frees the doctor up to be able to come in, answer any remaining questions, perform the procedure, then move on to the next patient. All of the technicians are fully trained for every procedure, as well as to check eligibility and perform other tasks so the clinic doesn’t grind to a halt waiting on one person to do a specific task.
Dr. Adler: I think the infrastructure will look very different depending on the size of the practice. I’m in a multispecialty practice, but as the only ophthalmologist, I’m almost like a solo practitioner. I have one technician who does almost everything for me, other than front desk and billing. She will be responsible for ordering, checking insurance eligibility, and assisting with preparing for the actual administration. With this setup, I believe I will be scheduling specific appointments to administer DURYSTA and bringing patients back, rather than administering on the spot.
Dr. Zaman: I am in a large ophthalmology practice and we have a nice infrastructure in place already. In any size practice, I think you’ll be able to use the staff you have for the procurement of DURYSTA, and it won’t be difficult to implement. The biggest change will be organizing a buy-and-bill model for those that are not already doing that.
Dr. Singh: DURYSTA is an exciting addition to our treatment armamentarium to lower intraocular pressure in patients with open-angle glaucoma or ocular hypertension, and I think what we have discovered here is the feasibility of implementing it in the way that works best for the practice and patients.
DURYSTA™ Indications and Usage and Important Safety Information
Indications and Usage
DURYSTA™ (bimatoprost intracameral implant) is indicated for the reduction of intraocular pressure (IOP) in patients with open angle glaucoma (OAG) or ocular hypertension (OHT).
Important Safety Information
Contraindications
DURYSTA™ is contraindicated in patients with: active or suspected ocular or periocular infections; corneal endothelial cell dystrophy (e.g., Fuchs’ Dystrophy); prior corneal transplantation or endothelial cell transplants (e.g., Descemet’s Stripping Automated Endothelial Keratoplasty [DSAEK]); absent or ruptured posterior lens capsule, due to the risk of implant migration into the posterior segment; hypersensitivity to bimatoprost or to any other components of the product.
Warnings and Precautions
The presence of DURYSTA™ implants has been associated with corneal adverse reactions and increased risk of corneal endothelial cell loss. Administration of DURYSTA™ should be limited to a single implant per eye without retreatment. Caution should be used when prescribing DURYSTA™ in patients with limited corneal endothelial cell reserve.
DURYSTA™ should be used with caution in patients with narrow iridocorneal angles (Shaffer grade ˂ 3) or anatomical obstruction (e.g., scarring) that may prohibit settling in the inferior angle.
Macular edema, including cystoid macular edema, has been reported during treatment with ophthalmic bimatoprost, including DURYSTA™ intracameral implant. DURYSTA™ should be used with caution in aphakic patients, in pseudophakic patients with a torn posterior lens capsule, or in patients with known risk factors for macular edema.
Prostaglandin analogs, including DURYSTA™, have been reported to cause intraocular inflammation. DURYSTA™ should be used with caution in patients with active intraocular inflammation (e.g., uveitis) because the inflammation may be exacerbated.
Ophthalmic bimatoprost, including DURYSTA™ intracameral implant, has been reported to cause changes to pigmented tissues, such as increased pigmentation of the iris. Pigmentation of the iris is likely to be permanent. Patients who receive treatment should be informed of the possibility of increased pigmentation. While treatment with DURYSTA™ can be continued in patients who develop noticeably increased iris pigmentation, these patients should be examined regularly.
Intraocular surgical procedures and injections have been associated with endophthalmitis. Proper aseptic technique must always be used with administering DURYSTA™, and patients should be monitored following the administration.
Adverse Reactions
In controlled studies, the most common ocular adverse reaction reported by 27% of patients was conjunctival hyperemia. Other common adverse reactions reported in 5% 10% of patients were foreign body sensation, eye pain, photophobia, conjunctival hemorrhage, dry eye, eye irritation, intraocular pressure increased, corneal endothelial cell loss, vision blurred, iritis, and headache.
Please see full Prescribing Information.
© 2021 AbbVie. All rights reserved. DURYSTA™ and its design are trademarks of Allergan, Inc., an AbbVie company. DurystaHCP.com DUR146662 05/21
DURYSTA™ Important Safety Information for US Healthcare Professionals
Collapse -
Contraindications
DURYSTA™ is contraindicated in patients with: active or suspected ocular or periocular infections; corneal endothelial cell dystrophy (e.g., Fuchs’ Dystrophy); prior corneal transplantation or endothelial cell transplants (e.g., Descemet’s Stripping Automated Endothelial Keratoplasty [DSAEK]); absent or ruptured posterior lens capsule, due to the risk of implant migration into the posterior segment; hypersensitivity to bimatoprost or to any other components of the product.
Warnings and Precautions
The presence of DURYSTA™ implants has been associated with corneal adverse reactions and increased risk of corneal endothelial cell loss. Administration of DURYSTA™ should be limited to a single implant per eye without retreatment. Caution should be used when prescribing DURYSTA™ in patients with limited corneal endothelial cell reserve.
DURYSTA™ should be used with caution in patients with narrow iridocorneal angles (Shaffer grade ˂ 3) or anatomical obstruction (e.g., scarring) that may prohibit settling in the inferior angle.
Macular edema, including cystoid macular edema, has been reported during treatment with ophthalmic bimatoprost, including DURYSTA™ intracameral implant. DURYSTA™ should be used with caution in aphakic patients, in pseudophakic patients with a torn posterior lens capsule, or in patients with known risk factors for macular edema.
Prostaglandin analogs, including DURYSTA™, have been reported to cause intraocular inflammation. DURYSTA™ should be used with caution in patients with active intraocular inflammation (e.g., uveitis) because the inflammation may be exacerbated.
Ophthalmic bimatoprost, including DURYSTA™ intracameral implant, has been reported to cause changes to pigmented tissues, such as increased pigmentation of the iris. Pigmentation of the iris is likely to be permanent. Patients who receive treatment should be informed of the possibility of increased pigmentation. While treatment with DURYSTA™ can be continued in patients who develop noticeably increased iris pigmentation, these patients should be examined regularly.
Intraocular surgical procedures and injections have been associated with endophthalmitis. Proper aseptic technique must always be used with administering DURYSTA™, and patients should be monitored following the administration.
Adverse Reactions
In controlled studies, the most common ocular adverse reaction reported by 27% of patients was conjunctival hyperemia. Other common adverse reactions reported in 5% 10% of patients were foreign body sensation, eye pain, photophobia, conjunctival hemorrhage, dry eye, eye irritation, intraocular pressure increased, corneal endothelial cell loss, vision blurred, iritis, and headache.
Please see full Prescribing Information.









