Champagne Cork Hyphema
Surgical management of a New Year’s injury.
Uday Devgan, MD

New Year’s Eve is supposed to be a fun night of celebration. In some social circles, champagne is enjoyed liberally on the occasion, but there is a danger associated with opening these bottles of sparkling wine pressurized with two to three times the Earth’s atmospheric force. The corks can become projectiles that can cause serious ocular injury.
The trauma case I describe here was caused by a champagne cork that hit the patient’s anterior segment and caused a large hyphema that filled 80% of the anterior chamber with blood (Figure 1). On presentation, about 2 weeks after the injury, that blood had clotted and was completely obscuring the pupil, causing the patient’s vision to be light perception only. The patient had an elevated IOP that was not well controlled despite maximal topical therapy plus systemic acetazolamide. An ultrasound study showed that the posterior segment was normal, with a clear vitreous and an attached retina. This patient needed surgical intervention to clear the hyphema and restore normal anatomy and function to his eye.
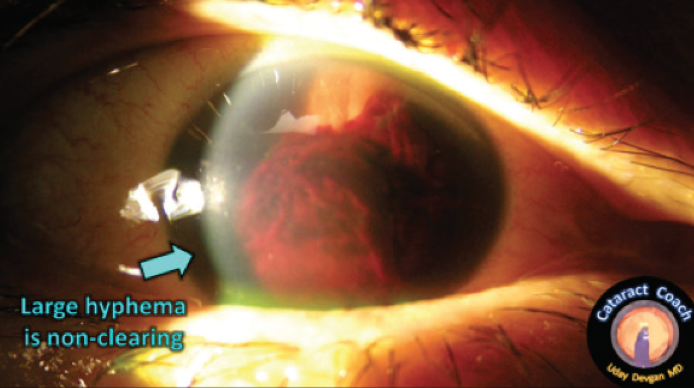
Figure 1. A severe hyphema filled about 80% of the anterior chamber, causing uncontrolled high IOP and complete obscuration of vision.
SURGICAL EVACUATION and Preventing A Rebleed
Two weeks after the trauma occurred, the blood simply was not clearing. I decided to perform a surgical evacuation of the hyphema and carefully remove the clot without touching the crystalline lens in order to avoid causing a cataract.
The technique for this surgery involved making two 1-mm incisions at the edge of the cornea and just barely nicking the limbal blood vessels. A 23-gauge instrument was placed in each of the incisions—an infusion cannula in one and a vitrectomy cutter in the other. I did not intend to perform a vitrectomy; I was instead using the vitrector to help aspirate and cut pieces from the blood clot.
To remove the clot, the suction of the vitrector was used to firmly grasp the coagulated material and then gently pull it away from the angle of the eye (Figure 2). The cutter action was engaged to break the clot into smaller pieces for easier aspiration once it had been freed from the iris.
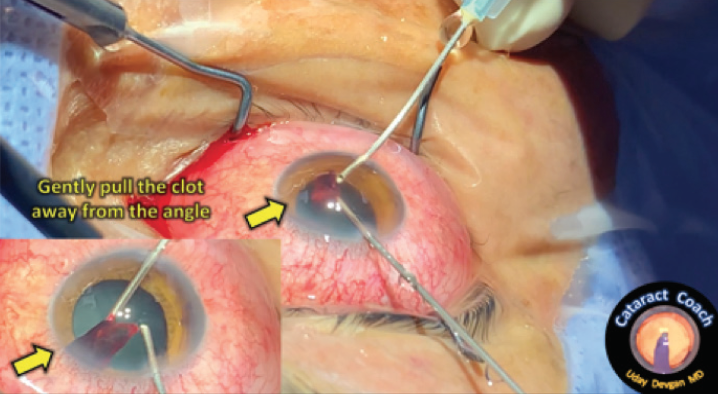
Figure 2. The vitrectomy cutter was used to remove the hyphema by engaging the blood clot with high vacuum and gently pulling the clot away from the angle.
Once the blood clot was removed, I wanted to ensure that rebleeding would not occur. This can be done in two ways: by adding epinephrine to the infusion fluid or by instilling it into the anterior chamber in a diluted concentration, which causes vasoconstriction, reducing the risk of a rebleed. A solution of 1:10,000 epinephrine mixed with balanced saline solution was injected to achieve this.
Phaco Settings
The settings of the phaco machine are of paramount importance to avoid causing an iatrogenic cataract during surgical evacuation of a hyphema. For this procedure, the phaco machine mode was set to I/A cut, which means that footpedal position 1 is irrigation, footpedal position 2 is aspiration, and footpedal position 3 is engaging the cutting function.
For this case, I dropped the cut rate down to 100 cuts per minute, which is about one cut every half-second. This was enough to break up the clot to aspirate it without clogging the port. The infusion pressure or bottle height must be raised significantly in order to deepen the anterior chamber and provide more room to work without risking touching the crystalline lens (Figure 3).
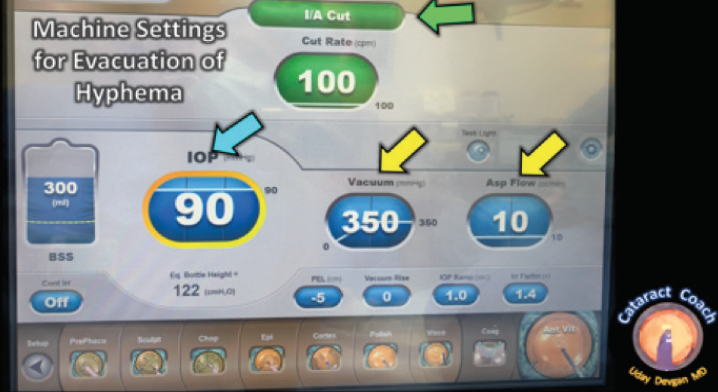
Figure 3. Suggested phaco settings for this procedure include using I/A cut mode (green arrow), a high infusion pressure (blue arrow), and strong vacuum with a low flow rate (yellow arrows).
Figures 1–3 courtesy of Uday Devgan, MD
Higher infusion pressure can also help prevent a rebleed as the clot is peeled away from the original site of bleeding. A vacuum level of 350 mm Hg provided enough grip and holding power to pull the clot away from the attachment points on the iris. Finally, the aspiration flow rate was intentionally kept low at just 10 mL/min so that it did not outstrip the inflow rate, especially with the small 23-gauge instrumentation.
Outcome
This surgical case took just a few minutes to complete, and the patient experienced a full recovery of normal vision. I suggested that he use safety glasses the next time he planned to open a champagne bottle. A full video of this case is available on CataractCoach.com.
The Psychological Toll of Ocular Trauma
A pellet gun injury changed the course of a young boy’s life.
Mark Kontos, MD

Managing patients with ocular trauma is pretty common for ophthalmologists. Most patients fade into the background of our memories. Some don’t, though. Some we remember vividly. This case is one of those to remember.
The injury itself was not unusual, a pellet gun shot to the eye of a 12-year-old boy. It was the aftermath and the consequences of the injury for him that have stayed with me over the years.
loss of useful vision
I was called to the ER on a summer’s afternoon to see a boy who had been accidently shot with a pellet gun in the left eye at close range by his best friend (a member of a prominent family in our community). It was clear on examination that the injury was severe and that the eye would not likely recover useful vision. As I was talking to the mother, who was visibly distraught, I realized we were acquainted. She was a single mother who worked at our country club. After several heroic attempts to save the eye by me, our cornea surgeon, and our retina surgeon, the eye was eventually removed, and a prosthesis was placed with very good cosmetic results. Unfortunately, this did not result in a happy ending for the boy.
Initially, things seemed to go well, and he seemed to be adjusting to his new situation. One day, however, his mom took me aside and said he was struggling in school and had become socially withdrawn. Before the injury, he had been a smart, outgoing, athletic kid who had been very popular in school. The injury had resulted in a subsequent lawsuit that caused him to lose his best friend and many of their common friends. He was no longer able to play organized sports because of his eye injury and the associated loss of binocularity. His dreams of playing football in high school and being in the military had shattered.
I started to notice the changes over time as well and felt he was at risk for depression and its consequences. He did not have a father figure in his life, so I tried to do what I could to help without being too intrusive. I was able to get one of the professional golfers at the club to help, and we got the boy involved in golfing and the snowboarding camp at our ski mountain.
Over the next couple of years, he seemed to be managing as well as could be expected, and I was no longer in close contact with his family. His mother had left her job at the club, and I was seeing him only for yearly examinations.
bad news
When he missed his appointment, I contacted his mother. Unfortunately, the news was not good. He had dropped out of school and had developed a significant drug addiction. After several unsuccessful attempts at rehabilitation, he left home and moved to Portland, Oregon. The mother lost all contact with him and became very depressed over the situation. I, too, was saddened by the course his life was taking.
It was hard not to think that the trajectory of his life would have been much different had that pellet just grazed his face instead of damaging his eye. He had been a happy, well-adjusted boy with a life full of possibilities. That all changed the day he lost his eye, and I remember it like it was yesterday.
Ruptured Corneal Graft
A challenging case, a poor outcome, and a lesson learned.
By Mark Lobanoff, MD

On a Friday in 2014, I was on call and feeling pretty tired from a busy week in the OR and clinic. At 11:45 pm, my phone rang, and my heart sank. On the other end of the line was an ER doctor calling from the local hospital. He sounded very concerned as he told me that one of my patients had fallen and hit his eye on the edge of a coffee table and that he appeared to have a ruptured globe and facial fractures. I immediately rushed to the hospital.
I knew this patient well. He was a 97-year-old retired pathologist I had gotten to know over the course of 10 years as his ophthalmologist. He was sharp as a tack and liked to trade stories with me during his many visits. His history included cataract surgery with placement of a posterior chamber IOL in 1984 and penetrating keratoplasty for a central corneal scar in 1994. I had been monitoring the patient for endothelial graft failure, and, 1 year before this accident, I had performed Descemet stripping endothelial keratoplasty to restore vision.
The scene in the ER was horrific. A large hematoma with embedded iris structures was protruding through a 300º rupture of his corneal graft (Figure 4). The patient needed immediate surgery, but I had a decision to make. Because he was taking warfarin for atrial fibrillation and had a history of an embolic stroke, I had to decide whether to reverse the coagulopathy before surgery or not. Doing so would make the surgical repair easier but risk a stroke. Otherwise, I could proceed with the patient’s international normalized ratio at 3.0. I also had to decide whether to request fresh frozen plasma if proceeding or prothrombin complex concentrate (PCC) if reversing the coumadin coagulopathy.
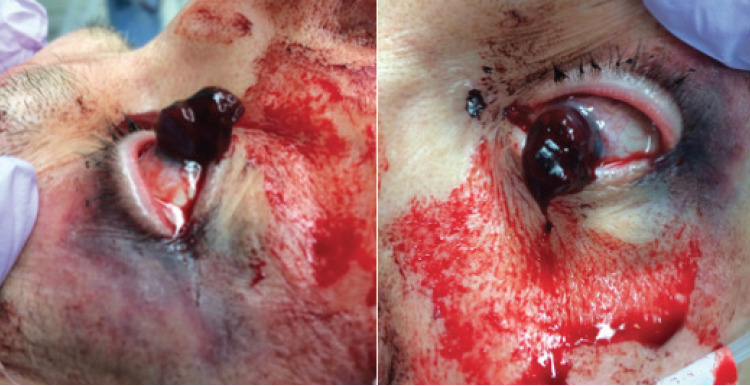
Figure 4. A large hematoma with embedded iris structures protruded through a 300º corneal graft rupture.
Surgical Repair
I decided to proceed with surgical repair without reversing the coumadin coagulopathy because it appeared that the bleeding had ceased. In the OR, I began by investigating the extruded contents. I carefully trimmed away the clot and removed about 75% of the avulsed devitalized iris. The trauma had caused the ciliary body and iris to separate, resulting in a severe hemorrhage. After debulking the hematoma, I closed the corneal wound with 24 interrupted 10-0 nylon sutures—a task made difficult by the large subconjunctival hematomas that developed every time I grasped the conjunctiva with forceps. Finally, after 3 hours, I felt I had achieved closure of the eye.
Outcome
Preoperative comuted tomography of the orbit had shown that the optic nerve and extraocular muscles were intact (Figure 5). The extensive hemorrhaging in both the anterior and posterior chambers, however, prevented a preoperative evaluation of the retina.
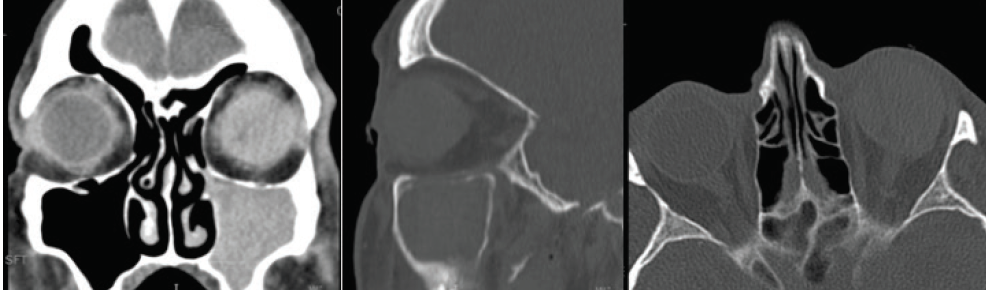
Figure 5. Preoperative computed tomography of the orbit revealed an intact optic nerve and extraocular muscles.
Figures 4 and 5 courtesy of Mark Lobanoff, MD
After 3 days, the patient’s vision improved to light perception. I was hoping that he had not suffered damage to his retina—that it was not an expulsive choroidal hemorrhage—but A- and B-scan ultrasound revealed large choroidal hemorrhages and a funnel retinal detachment. Given these findings, the decision was made to enucleate the eye.
A Lesson Learned
This was a difficult case with a very poor outcome. I learned an important lesson from it, however: I should have used PCC and reversed the coumadin coagulopathy before undertaking surgery. PCC works faster than fresh frozen plasma and does not have to be crossmatched with the patient’s blood type. The risk of stroke would have been very low, and the surgical exploration and repair would have been much easier had I decided to reverse the coumadin and use PCC preoperatively.
Traumatic Cataract
The tale of a traumatic cataract and three federal agencies.
By Michael E. Snyder, MD

An FBI agent—let’s call him Agent Iris—suffered a blunt injury to his left eye during a training exercise. The injury left him with significant photophobia and markedly reduced vision due to a mature, misshapen cataract and a 4.5-clock hour iridodialysis with a necrotic bridge of iris tissue (Figure 6). After a lengthy discussion of options, Agent Iris elected to undergo phacoemulsification of the cataract with vitrectomy and implantation of a Cionni Capsular Tension Ring (Morcher) and CustomFlex ArtificialIris (HumanOptics).

Figure 6. After a blunt injury to the left eye, a dense cataract, capsular plaque, iridodialysis, and zonules tethering the lens’ edge superonasally were observed. Pigment in the prolapsed vitreous was evident in the anterior chamber.
Red Tape
At the time, the CustomFlex ArtificialIris was not available in the United States. We therefore planned to obtain the device by making use of the FDA’s compassionate use device exemption (CUDE) program, which we had previously used in a handful of cases. Unfortunately, the Department of Labor, which manages employment benefits for FBI employees, was very slow to approve payment for the device, so Agent Iris waited and waited.
After 6 months, the Department of Labor approved payment for the custom iris device, and a CUDE request was submitted to the FDA. However, the FDA’s investigational device exemption (IDE) study protocol for the CustomFlex was already under negotiation, and the agency was unwilling to issue a CUDE for the device in anticipation of the launch of the IDE study. Agent Iris had to go back to waiting.
The Surgery
Agent Iris became the first patient in the FDA IDE study 20 months after presentation. His surgery began with a one-port pars plana anterior vitrectomy to remove the prolapsed vitreous gel. A highly dispersive OVD was used to tamponade and protect the remaining exposed aphakic space during phacoemulsification. A type 1G Cionni capsular tension ring (CTR) was threaded into the capsular bag, and the capsule was recentered by tethering the ring to the scleral wall with a permanent suture.
An IOL was implanted into the capsular bag, and the CustomFlex ArtificialIris was trephined to the correct size and implanted into the capsular bag. Trypan blue dye was used to visualize the capsule while the device unfolded in the capsular bag, because the red reflex is no longer visible when the opaque device begins to open. The necrotic iris tissue was removed using a vitrector, and the surgery was completed uneventfully. Agent Iris achieved a happy outcome with a marked reduction in photophobia, no glare, and a UCVA of 20/16 at his final visit (Figure 7).
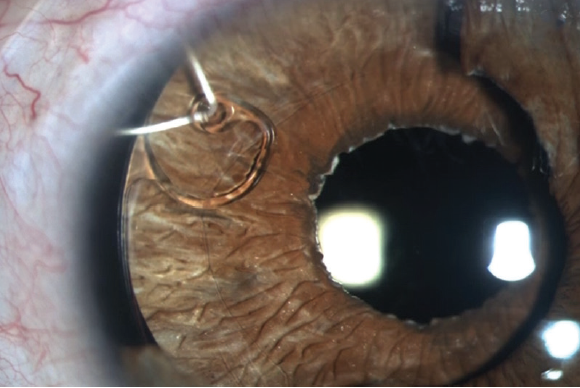
Figure 7. A Cionni ring and iris prosthesis are visible within the capsular bag postoperatively. The capsulorhexis is visible overlying the prosthesis, and the suture securing the CTR is visible in its anterior chamber path and along the episcleral surface under the conjunctiva in the upper right corner of the image. The color match of the device compares favorably to the residual native iris tissue, which can be seen temporally.
Figures 6 and 7 courtesy of Michael E. Snyder, MD
Challenges Not Uncommon
This story, although unique in the layers of federal bureaucracy involved, is not unique with regard to the administrative challenges that patients with traumatic eye injuries face, especially when they have other comorbid ocular injuries, such as the iris damage in this patient. His case was finally resolved after 2 years, but I have had other patients who had to wait even longer. One patient who presented during my early experience with CUDEs had to wait 8 years to get approval, which came only after the device had been approved by the FDA for more than a year.
Fortunately for similar patients today, the CustomFlex ArtificialIris is now approved by the FDA and covered by Medicare and an increasing number of commercial insurers. Procedure codes for insertion of the device have been approved and will become active in July.
Roman Candle Injury
Treating a remarkable child’s thermal injury taught me several lessons early in my career.
By Elizabeth Yeu, MD

Managing ocular trauma can be physically and emotionally draining for all involved—the patient, the patient’s support system, and the clinicians. One case that taught me several lessons occurred early in my career. The patient was a 4-year-old boy who came in as an emergency with a roman candle injury to the right eye that had occurred 1 or 2 days earlier. He was mature for his age and allowed me to perform a thorough slit-lamp examination.
Lesson No. 1: Thermal injuries can devastate the eye very quickly
The initial examination surprised me, as symblepharon was already beginning to form superiorly and inferiorly. The paracentral cornea had a small 0.5 x 1 mm perforation with iris plugging it, and the anterior chamber was formed. The corneal epithelium was denuded, with stromal thinning that was thinnest near the perforation. What a nightmare for this young child!
In the OR, I performed a repair of the corneal perforation. I released the iris plug and placed cyanoacrylate glue to close the small hole. After releasing the conjunctival adhesions within the superior and inferior fornices, I performed amniotic membrane transplantation (AMT) with a large graft, reforming the fornices and covering the palpebral and bulbar conjunctiva with the AMT. A large-diameter soft contact lens was placed, followed by a temporary tarsorrhaphy. I prescribed topical moxifloxacin four times per day, prednisolone acetate 1% four times per day, and 500 mg vitamin C daily.
Lesson No. 2: young children have a very robust healing potential
This patient required daily visits because the results of his clinical examinations changed quickly. Even the silk suture of the bolstered temporary tarsorrhaphy loosened much more quickly than in an adult patient. The AMT dissolved within 3 days. The large-diameter soft contact lens was allowing almost full conjunctival epithelialization without further symblepharon formation. The corneal surface was healing with conjunctivalized epithelium, which was not unexpected but was unfortunate, as I knew this meant limbal stem cell transplantation would be in his future. The most worrisome sign was slow reepithelialization of the central cornea. The corneal stroma was quite thin in the central 3 mm surrounding the area of the perforation. The anterior chamber was deep, which was a promising sign.
Lesson No. 3: Collagenase activity is fierce
I saw how quickly a cornea can decompensate and melt away after a thermal injury, and, coupled with the metabolism of a young child, the process literally seemed to happen overnight. The central corneal perforation enlarged within a few days as it underwent deturgescence. The patient required a tectonic penetrating keratoplasty (PKP). I explained to his parents that, for vision rehabilitation, the patient would almost certainly require repeat corneal transplantation and a possible allograft AMT. Thus, back we went to the OR for a tectonic PKP, multilayered AMT to the surface, a large-diameter bandage contact lens, and a temporary tarsorrhaphy.
This time, the cornea had a very difficult time reepithelializing postoperatively, and the patient’s peripheral cornea began to thin further. We decreased the topical medications, switched to preservative-free dexamethasone 0.1%, and maintained the oral vitamin C. The sutures loosened quickly. The clinical course necessitated multiple returns to the OR for this child, who willingly obliged, for suture removal or replacement and repeat AMTs.
A BRAVE PATIENT
After all of these heroic efforts, the patient presented 4 weeks after the PKP for a scheduled follow-up. He felt no pain, but endophthalmitis had consumed the eye, which was completely filled with exudate anteriorly and gas-forming exudate within the entire vitreous cavity. This precocious boy knew what this meant for him. He turned to his mother, who was in tears, and told her that he was ready to have the eye removed because he didn’t want her to cry over him anymore.
After the eye was removed, he always returned for his appointments, fully understood the importance of monocular protection, and was compliant with wearing polycarbonate spectacles at all times.
Ocular trauma can take devastating turns, especially thermal or chemical injuries. The resultant damage may be progressive in nature, necessitating sequential procedures, and the prognosis is guarded for extensive injuries.




