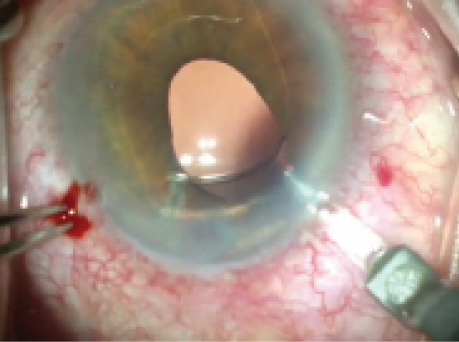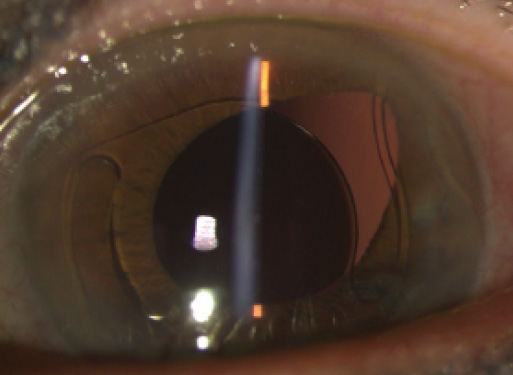
Figure 1. The haptic of the ACIOL has pulled the iris in the left eye superotemporally.
—Case prepared by Cathleen M. McCabe, MD.

STEVEN DEWEY, MD
This patient is obviously frustrated with the maintenance of a contact lens that either provides an artificial pupil or corrects anisometropia. My first step would be to place a punctal plug in the lower left eyelid and review the patient’s old records and her medical history. If alternative topical therapy such as a b-blocker would make her more comfortable, for example, this could be a simple temporizing option.
If the contact lens remained uncomfortable, the question about the distorted pupil would be whether it is trapped by the haptic or whether the iris is incarcerated in the incision. If it is the incision, the iris is likely stuck, and the recommended solution would be a coreoplasty. It could be achieved around an ACIOL, because most of the maneuvers take place below the iris plane. Obviously, care would need to be taken to avoid suturing the iris to the haptic.
If instead the haptic has trapped the iris, it could potentially be freed with manipulation using a forceps (MicroSurgical Technology). This procedure would be more challenging with the ACIOL in place, because the maneuvers typically take place anterior to the iris. A coreoplasty might still be the best option, but a gentle tug on the iris with the haptic retracted might free the iris rather easily.
Exchanging this ACIOL for a sutured or glued PCIOL would address the anisometropia but likely would not improve IOP control. The exchange would make the iris maneuvers easier by removing the physical obstruction of the ACIOL. It would also require a larger incision, however, and thus a longer recovery in addition to potentially worsening the patient’s ocular surface disease, which is the primary source of her current dissatisfaction.

SUSAN M. MacDONALD, MD
Proper placement of an ACIOL is critical to avoiding long-term complications. A malpositioned ACIOL can cause an irregular pupil, chronic inflammation, glaucoma, recurrent hyphema, cystoid macular edema, and corneal decompensation. This particular case presentation describes symptoms originating from iris capture of the ACIOL.
A complete eye examination, including an endothelial cell count and gonioscopy, would identify the details of the malpositioning and any coexisting conditions that could contribute to the patient’s symptoms such as dry eye and lid diseases. My discussion with the patient would include the risks and benefits of surgical intervention. The endothelial cell count would be crucial for predicting the cornea’s ability to tolerate a surgical intervention.
Because there is adequate iris tissue, my preference would be to reposition the lens and reestablish a normal pupil. If necessary, pupillary size could easily be reduced by releasing the peripheral iris, instilling a miotic intracamerally, and, if necessary, gently stretching the iris and possibly placing a suture. I would also consider treating the patient’s glaucoma at this time with endoscopic cyclophotocoagulation.
I would approach the case with peribulbar anesthesia. After placing a dispersive viscoelastic, I would use a Kuglen hook to grab the elbow of the haptic and pull it anteriorly and centrally. If this procedure released the iris, I would administer acetylcholine (Miochol-E; Bausch + Lomb) and place the haptic into the angle. I would then gently stretch the iris into its original configuration. If a suture were needed to reduce the size of the pupil, I would confirm placement with gonioscopy.

TIMOTHY PAGE, MD
The best approach to this complication is to avoid it by properly sizing the ACIOL and carefully walking the lens back and forth with a Kuglen hook to ensure there is no iris incarceration.
If the patient’s refractive error is significant, my first choice would be to explant the IOL and suture a three-piece lens to the iris. If the refraction is satisfactory, an attempt could be made to reposition the existing IOL.
After the administration of peribulbar anesthesia and sterile prep, I would create paracentesis incisions superotemporally and inferonasally and then inject an ophthalmic viscosurgical device (OVD). Using a Kuglen hook, I would push the temporal haptic and pull the nasal haptic while rotating the ACIOL counterclockwise until the haptic became free of the incarcerated iris. If corectopia persisted because of synechiae, I would administer carbachol intraocular solution (Miostat; Alcon) and use a blunt micrograsper and OVD to viscodissect any synechiae.
If the ACIOL would not rotate, I would exchange it. After creating a superior conjunctival peritomy, I would make a 6.5-mm, frowned, partial-thickness incision 1 mm posterior to the limbus. I would fashion a tunnel to the limbus and extend the incision 1 mm into the cornea. I would enter the anterior chamber with a keratome to create a triplanar incision, which I would then widen to 6.5 mm. Next, I would inject an OVD to tamponade vitreous.
I would grasp the haptic with a lens forceps to remove the ACIOL. Then, I would fold a three-piece IOL and place the haptics through the pupil with the optic above the iris plane. I would suture the haptics to the iris with 10–O nylon on a CIF-4 needle using Siepser knots.

R. BRUCE WALLACE III, MD
This patient’s treatment options could be better determined after further evaluation of her left eye. Gonioscopy would be prudent to determine the appearance of the iris-haptic contact in the superior temporal angle. Intraocular surgical revision could be difficult and high risk if significant fibrosis and/or neovascularization are apparent on gonioscopy. If just simple contact without any significant secondary effects is evident, a discussion of surgery is probably warranted. I would discontinue any anticoagulants or aspirin prior to surgery. I would carefully explain to this patient the risks associated with this procedure, especially considering the time that has elapsed since her original surgery.
Approaching the patient temporally, I would perform a 1.5-mm corneal incision near the limbus inferiorly and a 1.5-mm incision temporally. I would inject a miotic solution into the anterior chamber. Then, I would introduce a Lester hook through the inferior incision and simultaneously inject a dispersive viscoelastic agent gently through the temporal incision. I would engage the distal superior portion of the temporal haptic of the IOL with the Lester hook and gently pull the haptic inferiorly and anteriorly in hopes of disengaging it from the iris (with continued injection of the viscoelastic substance). The viscoelastic needle could also help bring the iris into proper position, and the haptic would be brought slightly anteriorly into the angle. I would perform irrigation and aspiration to remove the viscoelastic substance. All incisions would be stromally hydrated with balanced salt solution.

WHAT I DID: CATHLEEN M. McCABE, MD
After a thorough discussion of the risks and benefits of as well as alternatives to surgical intervention, the patient decided to proceed with a surgical solution. Options included possible release of the iris trapped behind the haptic or pupilloplasty to reconstruct a central, smaller pupil. Preoperative gonioscopy revealed no neovascularization in the angle and showed iris incarcerated in the angle behind the ACIOL’s haptic. The endothelial cell count and morphology were normal.
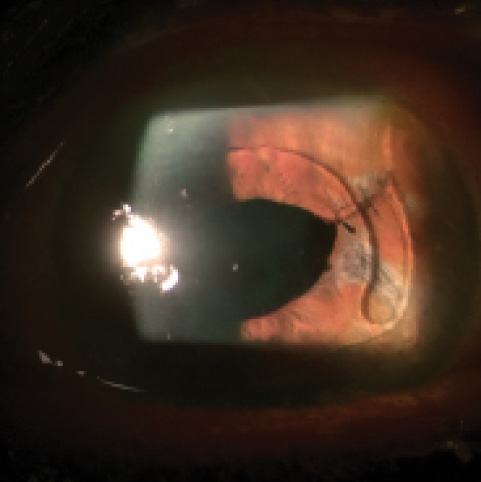
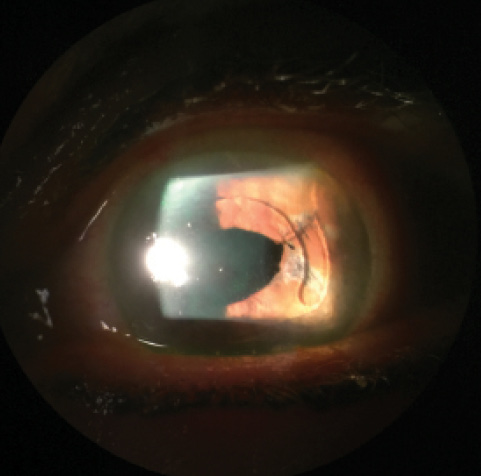
Figure 2. Postoperative appearance of repaired corectopia. The pupil no longer extends beyond the ACIOL’s optic, and the patient’s symptoms have resolved.
The patient was treated preoperatively with pilocarpine 2% topical drops. Intraoperatively, I instilled carbachol intraocular solution in the anterior chamber, followed by a dispersive viscoelastic (Viscoat; Alcon). Next, I used an Osher manipulator and an intraocular 23-gauge forceps (MicroSurgical Technology) to disengage the temporal haptic from the angle in an attempt to release the iris from the angle. Multiple attempts to gently extract the iris from the angle with intraocular graspers were unsuccessful. I performed a pupilloplasty with three interrupted 10–O Prolene sutures (Ethicon) in order to re-create a central pupil completely covered by the IOL. I used a micrograsper intraocularly to cinch the knots tightly while minimizing traction on the iris. I find this maneuver easier to perform than a Siepser knot, because the former is identical to tying a suture outside the eye. I instilled compounded triamcinolone and moxifloxacin in the anterior chamber to verify that no vitreous had presented and to provide an intracameral antibiotic and steroid medication.
Postoperatively, the patient’s IOP remained well controlled on her current medication, and her symptoms of glare and halos completely resolved (Figure 2). She is comfortably driving at night and is grateful for her improved quality of life. This case illustrates the importance of familiarity with intraoperative iris suturing techniques—a relatively straightforward procedure—for the treatment of iris defects and corectopia.
Section Editor Lisa Brothers Arbisser, MD
• emeritus position at Eye Surgeons Associates, the Iowa and Illinois Quad Cities
• adjunct professor, John A. Moran Eye Center, University of Utah, Salt Lake City
Section Editor Brandon D. Ayres, MD
• surgeon in the Cornea Service, Wills Eye Hospital, Philadelphia
Section Editor Cathleen M. McCabe, MD
• cataract and refractive specialist, The Eye Associates in Bradenton and Sarasota, Florida
• (941) 792-2020; cmccabe13@hotmail.com
• financial disclosure: consultant to and speaker for Alcon/Novartis and Bausch + Lomb/Valeant Pharmaceuticals International
Section Editor Audrey R. Talley Rostov, MD
• private practice with Northwest Eye Surgeons, Seattle medical advisory board, SightLife, Seattle
Steven Dewey, MD
• Pinnacle Eye Center, Colorado Springs, Colorado
• (719) 473-4507; deweys@prodigy.net
• financial disclosure: consultant to Abbott; has received royalties from MicroSurgical Technology
Susan M. MacDonald, MD
• assistant clinical professor, Tufts School of Medicine, Boston
• director of comprehensive ophthalmology, Lahey Clinic, Burlington and Peabody, Massachusetts
• (978) 538-4400; susan.m.macdonald@lahey.org
• financial disclosure: consultant to Alcon and Perfect Lens
Timothy Page, MD
• section head of anterior segment surgery, William Beaumont Hospital, Rochester, Michigan
• professor of ophthalmology, Oakland University, William Beaumont School of Medicine, Rochester, Michigan
• (248) 644-8060; tpagemd@yahoo.com
• financial interest: none acknowledged
R. Bruce Wallace III, MD
• medical director, Wallace Eye Surgery, Alexandria, Louisiana
• clinical professor of ophthalmology, Louisiana State University School of Medicine, New Orleans
• clinical professor of ophthalmology, Tulane School of Medicine, New Orleans
• (318) 448-4488; rbw123@aol.com