1. Juliette Eye Institute, Albuquerque, NM, USA; University of New Mexico School of Medicine, Department of Ophthalmology, Albuquerque, NM, USA
2. Shammas Eye Medical Center, Lynwood, CA, USA; Department of Ophthalmology, the Keck School of Medicine of USC, Los Angeles, CA, USA
*Paid consultants for Alcon Vision, LLC
Introduction
The SS-OCT technology is the latest generation of ocular biometers to measure ocular dimensions. The technology provides a longitudinal cross-sectional overview of the entire length of the eye. In this article, the unique benefits of the ARGOS® SS-OCT biometer are summarized.
Fast measurements
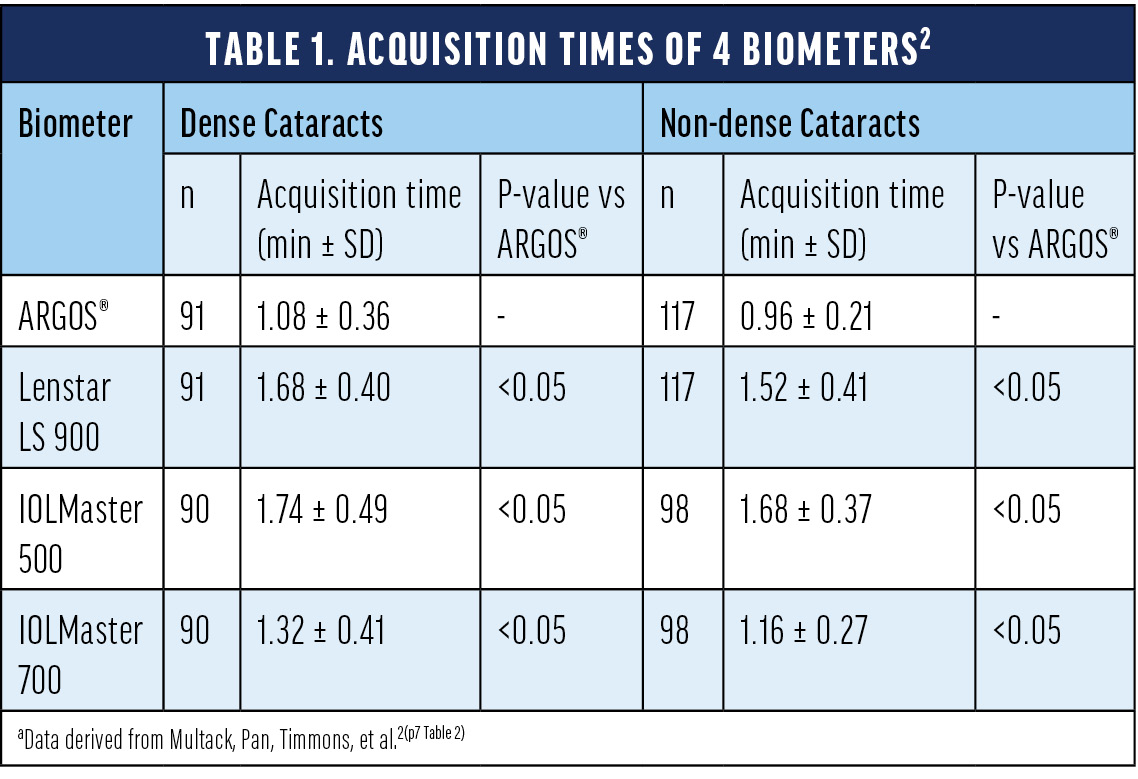
With increasing rates in cataract surgery globally,1 it is more and more important to evaluate the patient’s ocular biometry in a comprehensive, yet efficient, cataract workflow in the clinic. The ARGOS® biometer measures dense and non-dense cataracts in a fast manner as shown by a recent study investigating the acquisition time of various optical biometers.2 Specifically, the authors evaluated ARGOS®, Lenstar LS 900 (optical low-coherence reflectometry, OLCR), IOLMaster 500 (partial coherence interferometry, PCI), and IOLMaster 700 (SS-OCT). The time to obtain measurements with ARGOS® for either dense and non-dense cataracts was significantly shorter as compared to the other evaluated biometers (Table 1).2
Acquisition rates
Optical penetration of dense cataracts can be challenging and applanation A-scan ultrasound may be used to measure the axial length of a patient.3 When using applanation A-scan ultrasound, one encounters a larger variability in axial length measurements and prediction error as compared to optical biometers.4
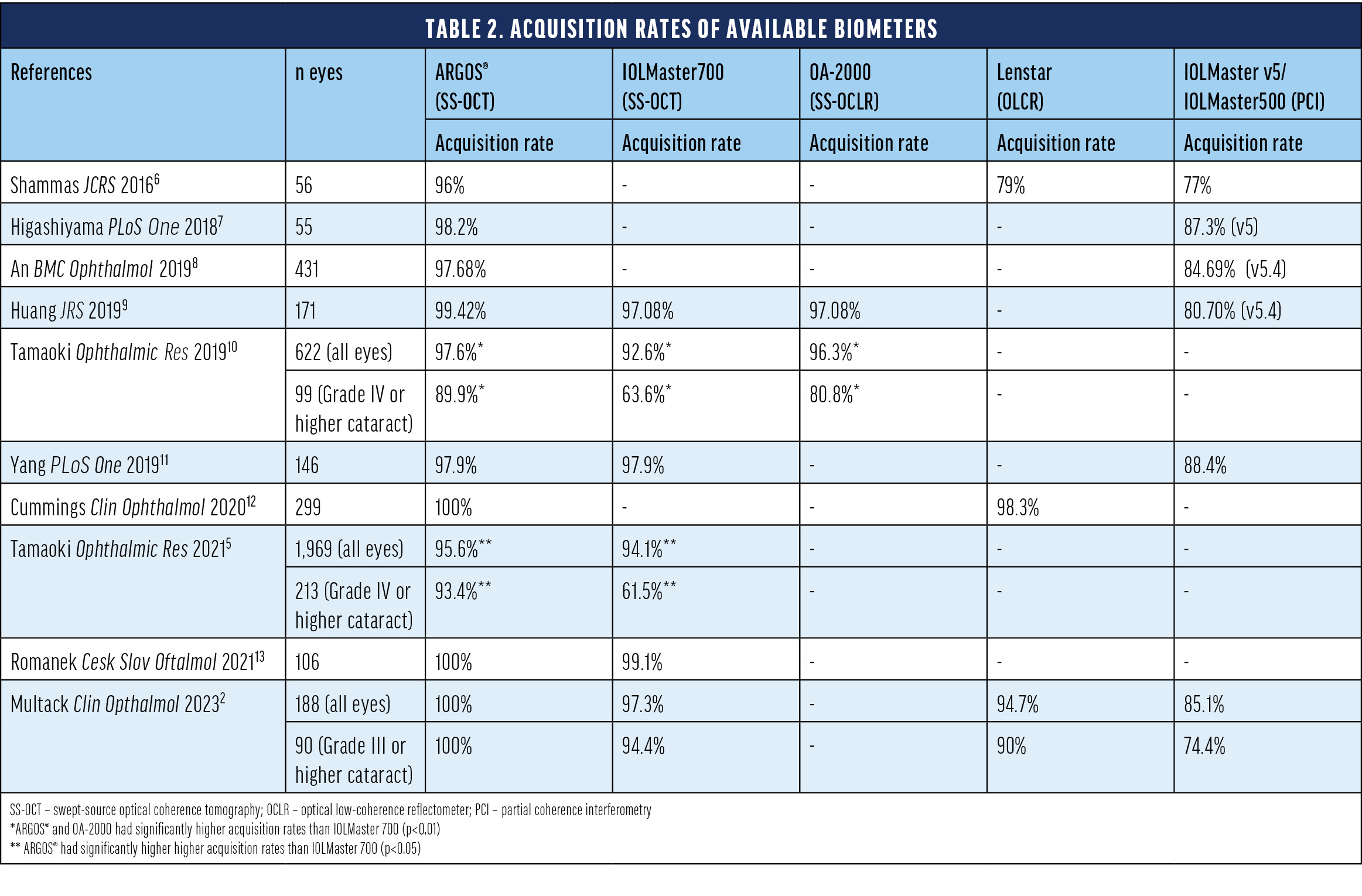
While SS-OCT imaging allows for better penetration of denser cataracts, ARGOS® improves penetration even further with the enhanced retinal visualization (ERV) mode.15 This unique mode improves the overall acquisition rates (Table 2) and reduces the need for applanation A-scans.14 Comparative studies showed that ARGOS® has the highest acquisition rates among tested optical biometers (Table 2). The improved acquisition capability becomes even more apparent in dense cataracts for which up to 100% of the axial length could be captured with ARGOS® (Table 2).2,5,10
An improved acquisition rate reduces the need for applanation ultrasound biometry supporting the efficiency of the clinic. In an economic budget impact model comparing the acquisition failure rates and need for ultrasound biometry, it was shown that using the ARGOS® biometer could result in substantially fewer measurements with ultrasound biometry (Figure 1).14 Additionally, time and cost efficiencies were identified as potential benefits.14
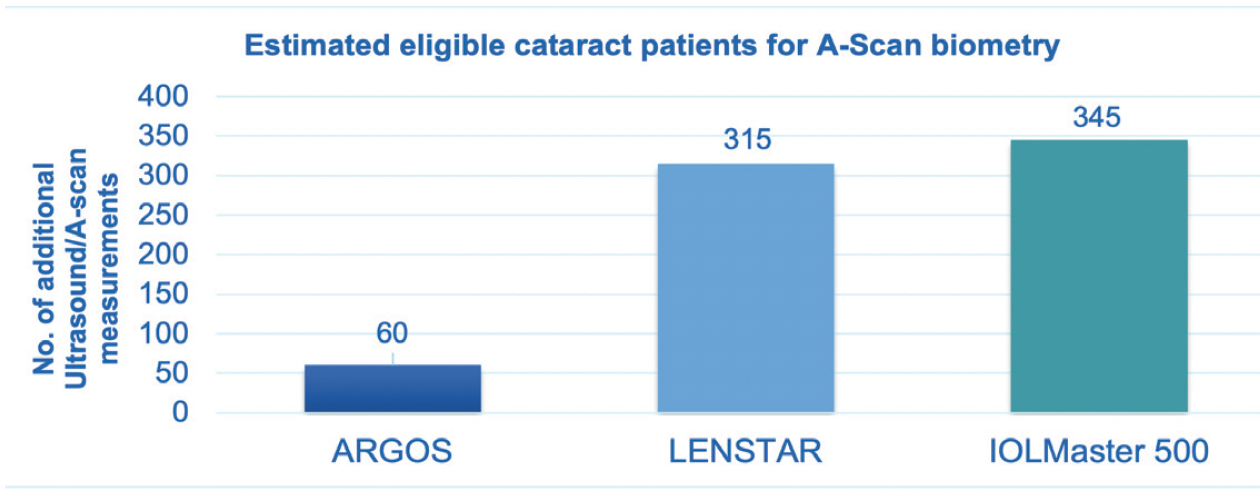
Figure 1. Potential number of additional ultrasound biometry/A-Scan performed annually with different optical biometers in a practice setting that operates on a patient mix with moderate and advanced cataract (n=1,500).14
Alcon Vision Planner Software
Another feature of the ARGOS® SS-OCT biometer is the Alcon Vision Planner that allows for surgical planning directly at the system or on any other networked device. The ARGOS® measurements feed automatically into the surgical plan. The software allows for a quick review of multiple formulas, IOLs, as well as astigmatic management options (Figure 2).15 Once the surgical plan has been finalized, it can be printed or exported to other Alcon systems.
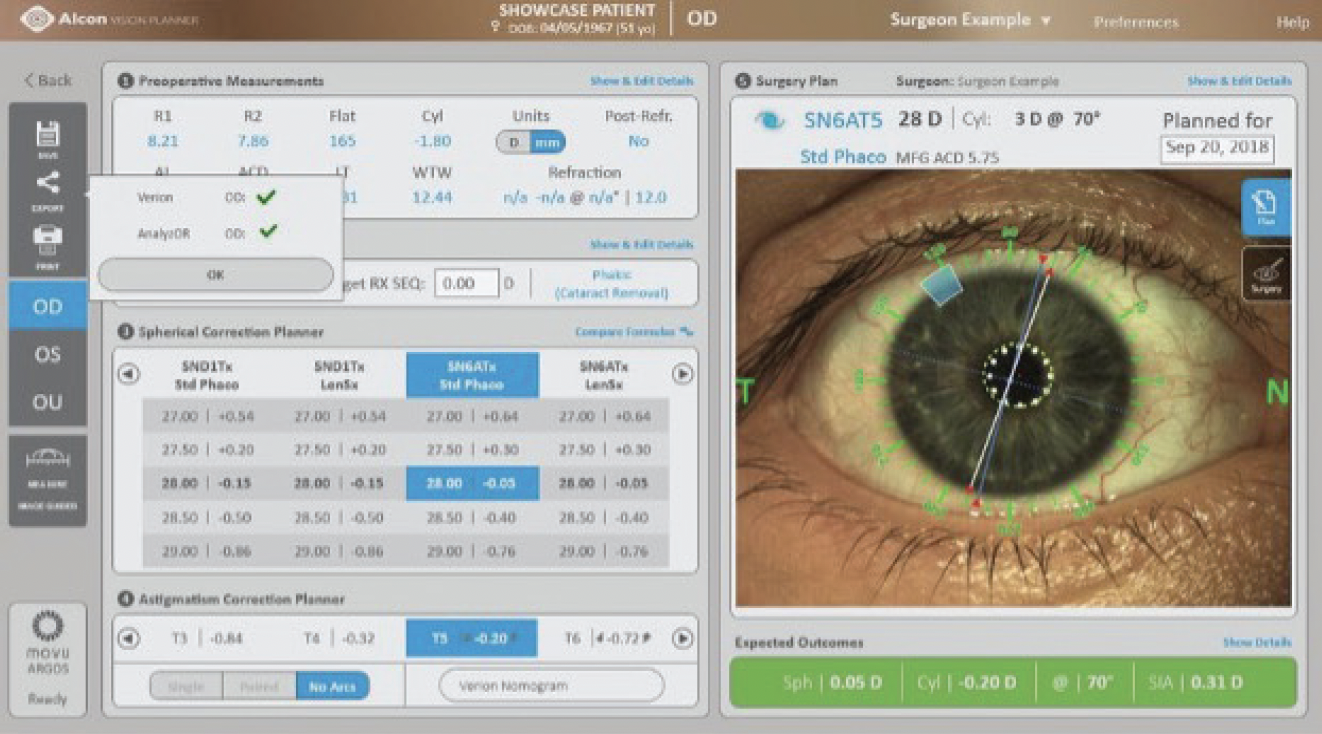
Figure 2. The Alcon Vision Planner provides a quick overview of multiple formulas, IOL options and astigmatic corrections.
After surgery, the software can support refractive outcome optimization, including optimization of the SIA by measuring postoperative keratometry and optimization of the A-constant by entering subjective refractions (sharable with the AnalyzORTM).15
Conclusions
The ARGOS® SS-OCT biometer offers several benefits including fast measurements in patients with dense and non-dense cataracts and the highest acquisition rates among optical biometers, particularly for dense cataracts.2,5,10 Improved acquisition rates of the ARGOS® biometer reduce the need for ultrasound biometry and the integrated Alcon Vision Planner provides convenient surgical planning directly at the device or at any other networked computer.14,15
IMPORTANT PRODUCT INFORMATION ARGOS® OPTICAL BIOMETER
Caution: Federal (USA) law restricts this device to the sale by or on the order of a physician.
Indication: ARGOS® is a non-invasive, noncontact biometer based on swept-source optical coherence tomography (SS-OCT). The device is intended to acquire ocular measurements as well as perform calculations to determine the appropriate intraocular lens (IOL) power and type for implantation during intraocular lens placement.
Intended Use: The Reference Image functionality is intended for use as a preoperative and postoperative image capture tool. It is intended for use by ophthalmologists, physicians, and other eye-care professionals and may only be used under the supervision of a physician.
Warnings / Precautions:
• Only properly trained personnel with experience may operate the device and control software and interpret the results.
• Factors that influence the measurement of patient’s eyes are listed in the User Manual (Table 1): pseudophakic eye, wearing contact lenses, fixation problem, cornea opacity, non-intact cornea, refractive surgery, blood in the vitreous humor, retinal detachment, keratoconus, asteroid hyalosis, ambient light in the room, and deformation of the corneal shape. Please consider the guidance provided in Table 1 when you encounter these factors.
• Optical Radiation - This device is equipped with a Class 1 laser light source.
Attention: Refer to the ARGOS® User Manual for a complete description of proper use and maintenance, optical and technical specifications, as well as a complete list of warnings and precautions.
© 2023 Alcon Inc. 06/2023 US-ARB-2300047
1. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28:98-103.
2. Multack S, Pan LC, Timmons SK, et al. Impact of a swept source-optical coherence tomography device on efficiency in cataract evaluation and surgery: a time-and-motion study. Clin Ophthalmol. 2023;17:1-13.
3. Mylonas G, Sacu S, Buehl W, Ritter M, Georgopoulos M, Schmidt-Erfurth U. Performance of three biometry devices in patients with different grades of age-related cataract. Acta Ophthalmol. 2011;89(3): e237-241.
4. Olsen T. Improved accuracy of intraocular lens power calculation with the Zeiss IOLMaster. Acta Ophthalmol Scand. 2007;85(1):84-87.
5. Tamaoki A, Kojima T, Hasegawa A, et al. Evaluation of axial length measurement using enhanced retina visualization mode of the swept-source optical coherence Tomography biometer in dense cataract. Ophthalmic Res. 2021;64(4):595-603.
6. Shammas Hj, Ortiz S, Shammas MC, Kim SH, Chong C. Biometry measurements using a new large-coherence-length swept-source optical coherence tomographer. J Cataract Refract Surg. 2016;42(1):50-61.
7. Higashiyama T, Mori H, Nakajima F, Ohji M. Comparison of a new biometer using swept-source optical coherence tomography and a conventional biometer using partial coherence interferometry. PLoS One. 2018;13(4):e0196401.
8. An Y, Kang EK, Kim H, Kang MJ, Byun YS, Joo CK. Accuracy of swept-source optical coherence tomography based biometry for intraocular lens power calculation: a retrospective cross-sectional study. BMC Ophthalmol. 2019;19(1):30.
9. Huang J, Chen H, Li Y, et al. Comprehensive comparison of axial length measurement with three swept-source OCT-based biometers and partial coherence interferometry. J Refract Surg. 2019;35(2):115-120.
10. Tamaoki A, Kojima T, Hasegawa A, et al. Clinical evaluation of a new swept-source optical coherence biometer that uses individual refractive indices to measure axial length in cataract patients. Ophthalmic Res. 2019;62(1):11-23.
11. Yang CM, Lim DH, Kim HJ, Chung TY. Comparison of two swept-source optical coherence tomography biometers and a partial coherence interferometer. PLoS One. 2019;14(10):e0223114.
12. Cummings AB, Naughton S, Coen AM, Brennan E, Kelly GE. Comparative analysis of swept-source optical coherence tomography and partial coherence interferometry biometers in the prediction of cataract surgery refractive outcomes. Clin Ophthalmol. 2020;14:4209-4220.
13. Romanek J, Slukova K. Comparison of optical biometers Argos and IOL Master 700. Cesk Slov Oftalmol. 2021;77:295-299.
14. Shammas HJ, Hsiao CWC, Jawla S. Presented at: The American Society of Cataract and Refractive Surgery (ASCRS) annual meeting; May 15-19, 2020; Boston, MA.
15. Alcon data on file.



