Cataract extraction is the most prevalent surgical procedure, and for over 30 years, phacoemulsification techniques have helped it to become one of the safest and most predictable surgeries.1 Historically, due to technological limitations with phacoemulsification systems, operating at high intraocular pressure (IOP) was necessary to create and maintain a stable anterior chamber with efficient aspiration flow and vacuum. We are gaining more and more new insights that show us just how high the pressure may become during surgery, as well as the potential impact on ocular structures. Recently, David Lubeck, MD, moderated a roundtable discussion on the topic of performing cataract surgery at a more physiologic IOP. He was joined by Jerry Hu, MD; Zaina Al-Mohtaseb, MD; and Arsham Sheybani, MD, a diverse group of anterior segment surgeons.
Defining intraocular pressure (IOP)
David Lubeck, MD: Thank you all for joining me today to discuss our motivation to consider and perform cataract surgery at a physiologic IOP. This topic has not been prioritized previously, and we all feel passionately that it is something we need to talk more about. Dr. Sheybani, would you please start the discussion of IOP in the context of cataract surgery?
Arsham Sheybani, MD: Simply stated, IOP is the fluid pressure of the eye. It is a measurement involving the magnitude of the force exerted by the aqueous humor on the internal surface area of the anterior eye.2 In healthy eyes, normal IOP pressure ranges between 11 and 21 mmHg and diurnal variances of 2 to 6 mmHg IOP with higher pressures typically found in the morning.2,3
IOP is a fine-tuned equilibrium between the production and drainage of aqueous humor. Any pressure changes in the eye can cause issues.2 A sudden increase in IOP exerts mechanical stress on the retinal nerve fiber layer, which can lead to ischemic effects.4 During cataract surgery, ingress and egress of fluid needs to balance. Without good balance, IOP will rise or fall, potentially causing issues in the eye.
Historical basis for supraphysiologic-IOP cataract surgery
Dr. Lubeck: The concept of phacodynamics was originally introduced by Barry Seibel, MD, in his classic text, Phacodynamics. The most recent edition publication was 1994, and since then, we, as a specialty, have not delved much into the nuanced evolution of phacodynamics—thus, the reason that we are here today, and thus the importance of regenerating the discussion about fluidics and the way our systems behave. Dr. Al-Mohtaseb, early in your career, why did you begin at a supraphysiologic IOP during cataract surgery?
Zaina Al-Mohtaseb, MD: It is important to remember that at that time technology was limited. Phacoemulsification machines were designed around safety, and high levels of irrigation pressures were necessary because the only way to move fluid through the eye was through gravity infusion. High IOP values maintained adequate chamber stability at efficient aspiration flow and vacuum and helped to compensate for dynamic pressure losses and chamber shallowing resulting from flow out of the eye. Otherwise, outflow of fluid from the eye would not be matched by inflow of fluid into the eye and would result in a shallow chamber.
We were not worried about the potential complications of having high pressures in the eye. What we did worry about was having a posterior capsular tear due to a surge or sudden shallowing in the anterior chamber. Back then, the settings were static because we could not monitor the feedback between the pressure in the eye and irrigation. As surgeons, our responsibility was to watch for the egress of fluid to prevent potential fluctuations. It was a very different system than what we have now.
Jerry Hu, MD: In the normal eye, the IOP is determined by the inflow and the outflow of the aqueous. Likewise, in the surgical model, the IOP is determined by the inflow and the egress of the fluid. In a passive fluid system where gravity is responsible for pushing the fluid through the infusion line, we only have two variables. If one variable is changing, we lose the stability of the anterior chamber. Whereas in a more active control system, we can dynamically modify or change the variable in response to the other changing variable to maintain a stable chamber and ensure safety of the surgery.
Detrimental effects of supraphysiologic-IOP cataract surgery and possible benefits of lower, more physiologic-IOP cataract surgery
Dr. Lubeck: Dr. Al-Mohtaseb, would you please review what is known about the effect of supraphysiologic IOP on the cornea and anterior segment and the possible benefits of operating at a more physiologic IOP?
Dr. Al-Mohtaseb: There is a good amount of data looking at supraphysiologic IOP during cataract surgery and how it affects the anterior segment. When you are operating at higher pressures, you can have corneal edema and also higher central corneal thickness after surgery. In one study, the data showed larger corneal volume 1 month postoperatively.5 The corneal thickness was increased at day 1,6-8 day 4,8 and day 7.6,7 There were also decreases in corneal clarity,7 a higher presence of Descemet’s folds,6,7 and greater anterior-segment inflammation (cells and flare)6-8 reported on day 1 postoperatively.
In regards to the corneal endothelial cell density, a number of studies in the past could not identify a difference when operating at higher versus lower levels of IOP.6,7,9 However, a recent study conducted in 2022 showed a higher protection rate of endothelial cell density with near-physiologic IOP (20 mmHg) versus high IOP (50 mmHg) at day 4 and at the 3-month follow-up visit, which was statistically significant (Figure 1).8 This is powerful because the phacoemulsification machine settings were very similar between the high IOP and near-physiologic-IOP groups.
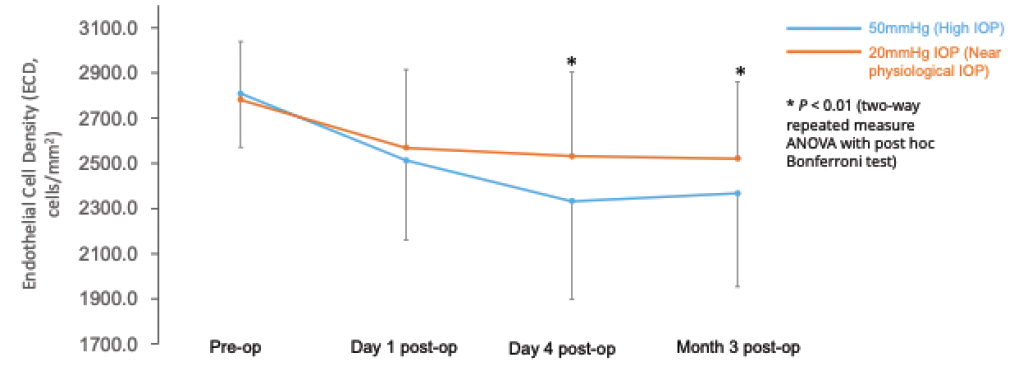
Figure 1. Comparison of endothelial cell density (ECD) in eyes operated under high IOP (50 mmHg, n=58 eyes) and near physiological IOP (20 mmHg, n=57 eyes). ECD was significantly less impacted at Day 4 and Month 3 postoperatively under near physiological IOP (P < 0.01).8
Now that we perform more premium cases, it is no longer just about how much edema remains at 1 week postoperatively; the clarity and lack of edema matter from the first day postoperatively. In my experience operating at near-physiologic IOP, I see less edema and a clearer cornea on postoperative day 1.6-8
Dr. Hu: My experience has been similar. I was operating with a bottle height of 115 (IOP pressure of about 85 mmHg), which is quadruple the upper limit of a normal IOP range. It was routine to see 2 to 3+ corneal edema on day 1 postoperative. Edema could take a week or two to resolve. This is more than a vanity or feel-good issue. In today’s world when we are working to evolve cataract surgery into lens-based refractive surgery, patients and referring/comanaging doctors demand not only good vision, but good vision sooner.
Dr. Lubeck: I believe that decreased edema at day 1 or week 1 is an indicator of improved corneal physiology. Our goal is to limit any adverse effects by providing what we believe is the best care for our patients. Dr. Sheybani, would you discuss the effect of operating at supraphysiological IOP in the posterior segment?
Dr. Sheybani: Cataract surgery with different irrigation systems and settings can impact the retinal tissue. Higher intraoperative IOP in the posterior segment reduces retinal blood flow. One study showed that intraoperative IOP of 55 to 60 mmHg resulted in absence of blood flow autoregulation mechanisms up to a complete stop in blood perfusion.10 The higher pressure correlates with macular thickness and tends to stick around longer compared to patients who had physiologic IOPs during surgery.11 While this may not change their ultimate visual acuity outcome, there might be rare cases where you could have macular ischemia issues.12 And I think we have all seen patients where everything else looks so good, but we wonder why their vision was not quite 20/20.
Dr. Hu: As a LASIK surgeon, I frequently see this phenomenon. When we create LASIK flaps and apply the LASIK suction ring to patients’ eyes, we tell them that they are going to lose their vision temporarily. Now, imagine the LASIK suction ring increasing the IOP level and inducing a similar rise in IOP that occurs during cataract surgery, only on a larger scale and for a longer duration. This is not insignificant—the potential impact on the retinal vasculature/ocular ischemia needs to be considered on a broader scale in the public health realm. If we know there is a potential element that can worsen this, and we know that we can perform the procedure safely without negatively affecting outcomes, then I think it is the right way to go.
Dr. Lubeck: Maintaining the stability of the anterior vitreous face (AVF) is another point of importance for operating at a more physiologic IOP (Figure 2). Dr. Vasavada has shown that high infusion pressures and irrigation can cause trampolining of the lens iris diaphragm and ruptures of the AVF.13
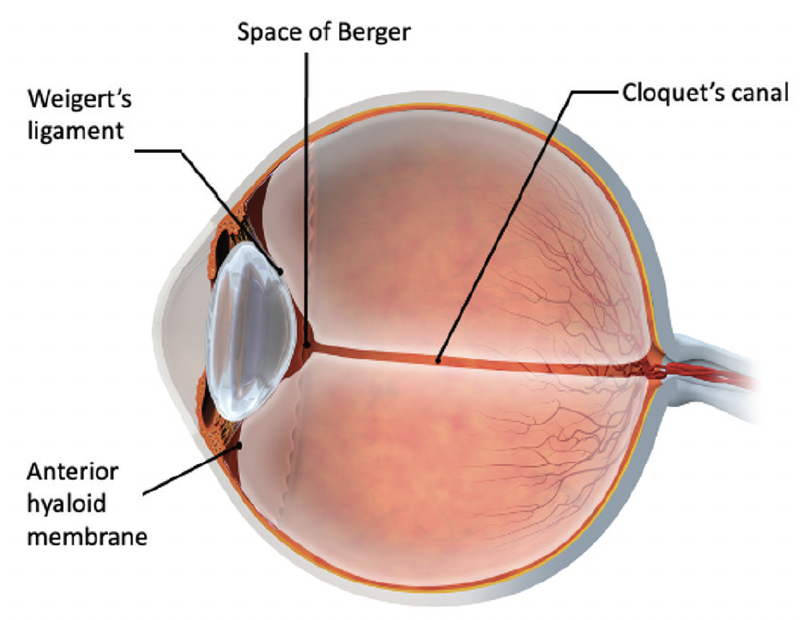
Figure 2. AVF extends behind the posterior lens capsule and encloses the Space of Berger starting from the Weigert’s ligaments.
Dr. Hu: It is natural for high IOP to exert more stress on the AVF, capsule, and zonules. I operate at an IOP level of 20 mmHg and am managing postoperative and post-traumatic eyes without a problem. Recently, I had a case where a different surgeon’s profile was set on the phacoemulsification machine. The sheer inflow of the balanced salt solution (BSS) at 60 mmHg, a common setting for surgeons, on a post-vitrectomized eye was enough to break the capsule and cause complications.
Evolution of technology facilitating lower, more physiological-IOP cataract surgery
Dr. Lubeck: The evolution of phacoemulsification technology provided the foundation for us to rethink IOP during cataract surgery. Each successive generation of systems has lessened the need for supraphysiologic IOP. Now, the surgeon community and industry can prioritize the discussion surrounding lowering IOP during cataract surgery.
Before 2003, we only used longitudinal phacoemulsification, which cores the nucleus. This created chunks of nucleus that caused clogging in the flare tips. I used the tapered 1.1 Kelman tip, which provided great cutting efficiency and hold, but to overcome the clogging, we had to increase our flow and vacuum rates. And because of poor sleeve-to-incision fit, there was a lot of fluid egress that required extremely high infusion pressure to maintain chamber stability.
Dr. Hu: That brings back memories. I trained in the late 1990s on the Legacy system (Alcon), and when I began practicing in 2003, the Infiniti gravity-based fluid infusion system (Alcon) was new. At that time, it was not uncommon to require a second bottle of BSS. In comparison, for long and complex cases we now routinely use less than 50 CCs of BSS, and in most cases, as little as 20 CCs. These passive fluidic systems are not cornea friendly by any means; the more fluid we push through the anterior chamber, just like the more ultrasound energy we consume, the more exogenous environmental factors we expose corneal endothelial cells to.
Dr. Lubeck: This brings us to the first pivotal point in the evolution of technology for low-physiologic IOP in cataract surgery. Infiniti offered a 2.2- to 2.4-mm incision with matched sleeve. The fluid egress was decreased significantly from the primary incision, and the anterior chamber became more stable. In 2008, torsional ultrasound was introduced. This employed a torsional motion instead of longitudinal to shave the nucleus rather than core it. With less clogging of the phaco tip, the likelihood of surge decreased, and chamber stability improved.14
In 2013, the Centurion Vision System (Alcon) was introduced (Figure 3). Active fluidics—pressurized infusion—and the balanced non-tapered phaco tip essentially eliminated the obstacles that led to surge. This allowed us to operate more efficiently at higher flow and vacuum rates with decreased operating room time and fluid use.15
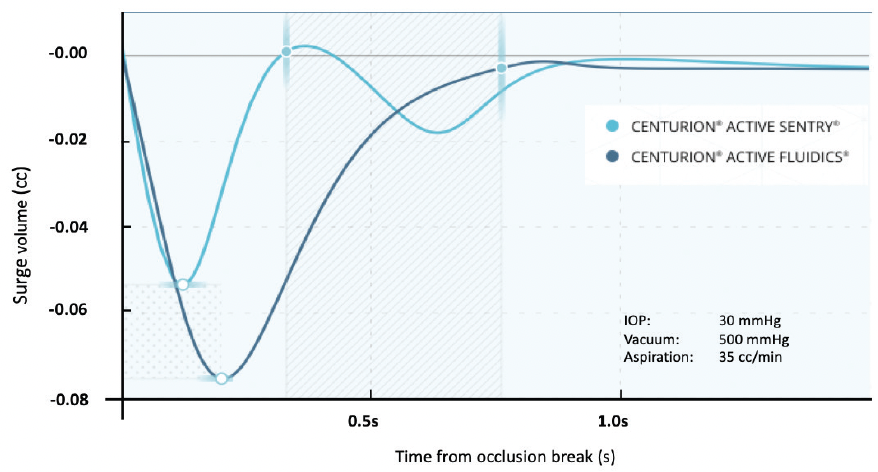
Figure 3. Occlusion break surge comparison. Centurion® Active Sentry® Vision System compensates faster as compared to the Centurion® Vision System with Active Fluidics®.17
Experience performing surgery at a lower, more physiological IOP
Dr. Lubeck: As a community, we did not start talking about lowering our bottle heights until Centurion came along; however, many surgeons stuck to an IOP of 50 to 60 mmHg. Dr. Hu, would you please share your clinical experience and strategy to transition to lower IOP during cataract surgery?
Dr. Hu: When we lower the IOP target in the operating environment, the questions that come to mind are, ‘Will that make the anterior chamber less stable, and will that decrease the efficiency of the surgery?’ Kokubun et al8 did a study comparing surgeries performed at a supraphysiologic level of 50 mmHg versus a more physiologic IOP level of 20 mmHg. Using similar vacuum levels, aspiration flow rates, and ultrasound power settings, they found that there is essentially no difference in the total surgical time or in the utility of ultrasound power as measured by cumulative dissipated energy (CDE).8 This has been my personal experience as well, and it is nice to see that supported by evidence-based findings in the literature.
Another question is whether the stability of the anterior chamber may be affected by low-IOP cataract surgery, particularly if occlusion breaks occur. In a pig-eye study, Suzuki et al compared multiple phaco systems by measuring anterior-chamber fluctuations in low IOP settings (30 mmHg) during phacoemulsification and occlusion break surges (Figure 4).16 The researchers found that the chamber remained stable with the Centurion Vision System with the Active Sentry Handpiece and produced the least variability in the anterior chamber, whereas the gravity-based systems collapsed or shallowed the chamber to a greater extent.16
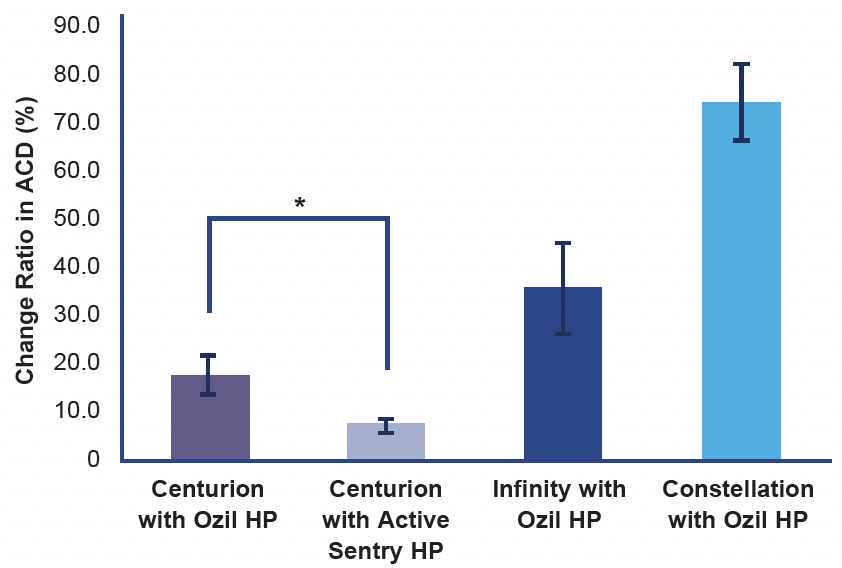
Figure 4. Comparison of Anterior Chamber Depth (ACD) fluctuation in three phacoemulsification systems. The Centurion® Vision System showed the lowest fluctuation in ACD.16
These findings echo my own personal experience. Centurion with the Active Sentry Handpiece is safe and makes my surgeries more efficient. In addition to the IOP variable, I have since changed the other parameters without noticeable difference in the chamber stability and safety. I have increased the aspiration flow rate and vacuum level. I engage phacoemulsification power sparingly to break up pieces from the tip. I describe it as a phaco-assisted high-vacuum system. I can safely and confidently adjust phacoemulsification parameters because the fluid is managed, and the chamber is maintained.
Dr. Al-Mohtaseb: That is an important point for surgeons who are considering lowering the IOP during cataract surgery. The study by Kokubun et al8 shows us that we will not lose efficiency; instead, we gain the benefits from having less fluid through the eye.8
In the past, we worried about changing and managing bottle height based on the type of procedure we did. Once I got comfortable operating at near-physiologic IOP and saw that I was not losing efficiency or compromising safety, I stopped thinking about how to manage bottle height, even in complex cases. I rarely change bottle height at this time.
Dr. Lubeck: At the onset of my own evolution from supraphysiologic to physiologic IOP cataract surgery, these studies did not exist. Initially it was counterintuitive to me, but over time it made increasingly more sense. Now I rarely need to think about IOP management during surgery.
Dr. Hu: This is made possible by the active fluidics management system. When we rely on gravity to push the fluid through the infusion tube into the anterior chamber, we are subject to one variable. Now we have real-time in vivo sensing of the IOP level—the system dynamically compensates for the egress of fluid. The Active Sentry sensor senses the real IOP level, and if it drops below the target level, the pressure plates compress on the BSS bag to increase the inflow of the BSS into the chamber and maintain the chamber stability. The Centurion Vision System with the Active Sentry Handpiece does the job for us, so we can take our mind off IOP and chamber stability and focus on improved efficiency.
Dr. Lubeck: Studies of anterior chamber stability, or surge volume, clearly show that the newest fluidics management systems with sensing and pressurized infusion have minimized surge volumes to such a low level that there is virtually no perceptible change in anterior chamber or capsular bag depth during routine surgery steps. The following video demonstrates that there is no noticeable difference in anterior chamber stability operating at supraphysiologic versus physiologic IOP. In the video clip, I challenge you to determine when the IOP drops from 60 to 22 mmHg. I think you’ll find it very difficult to notice.
What fears, recommendations, or other parameter changes along with decreasing the IOP would you like to address with our colleagues?
Dr. Al-Mohtaseb: Speaking to the concept of fear, the key is to be comfortable in the fact that the technology has come a long way. Now we know that going to near-physiologic IOP does not affect efficiency and has been shown in the literature to be safe.
Dr. Hu: Looking back on my own experience of transitioning, I was exposed to both Centurion and the Active Sentry Handpiece before the commercial launch and through some studies. I quickly embraced them when they were commercially released. I started out at an IOP of 85 mmHg and reduced IOP in 5- to 10-point increments. I did not see any change in performance, and the chamber remained stable. In a matter of days over the course of several dozen cases, I dropped down to an IOP of 20 mmHg, which I continue to operate at now.
All the parameters can be changed, but change one parameter at a time, and do it gradually. Change comes not only from the technologic side but also from the surgeon’s side. I have found that even years after I have settled in a certain setting, it is good to challenge myself and test certain parameters to further improve efficiency and safety.
Dr. Sheybani: I agree. After you get comfortable at lower pressures, you can set it and forget it because the technology takes over and maintains stability. I have also ramped up my vacuum settings because I know that chamber pressure will be maintained, which means I use less phacoemulsification power.
Dr. Lubeck: I recommend keeping your original program in the system and setting up a second program for a physiologic IOP. Start with your original settings in that second program and change your settings gradually. As a general recommendation, do five cases at each change. With the two programs, you can at any point return to your original setting to compare.
I hope that we all continue evolving as surgeons and encourage our colleagues to do the same. Part of that evolution is rethinking, questioning, and asking ourselves if we can do something better and whether we should be doing things differently.
The Patient Experience
Dr. Lubeck: Perhaps most compelling is the improved patient experience when cataract surgery is performed at physiologic IOP. How have your patients’ perspectives been impacted by these parameter adjustments?
Dr. Sheybani: We discussed earlier that patients can feel the rise in pressure. We have evidence showing that the highest pain scores occur during phacoemulsification.18 We also know that nearly half of our patients experience pain during irrigation.19 The biggest changes in IOP occur between irrigations and phacoemulsification steps, which can be jarring for a patient, especially high myopes.
Dr. Al-Mohtaseb: Patients have raised the bar high for us, and especially so in our premium cases. They want to see better and feel better, and they want not only great vision, but they want great vision faster. Comfort during surgery is a big deal, and they expect good vision from day 1. By operating at physiological IOP, we can help meet their expectations.
Dr. Hu: The tangible benefits of physiologic-IOP cataract surgery are less corneal edema after surgery.5-8 The intangible benefits are more confidence in our referring and comanaging doctors, which is a prerequisite for cataract surgery to truly evolve into lens-based refractive surgeries.
Summary
Dr. Lubeck: For the many advantages and benefits discussed, I believe we all would want our own cataract surgery to be performed at near-physiologic IOP. To conclude our roundtable, what final words would you like to leave with our colleagues?
Dr. Al-Mohtaseb: I know that I want my own cataract surgery performed at as low pressure as possible that still maintains the chamber and ensures safety. We are fortunate as surgeons to work so closely with industry to bring forward new technologies. It is important for us to have an open mind and evaluate and use new technologies if they are better for our patients. With respect to education and working with residents, do not make sudden changes—slowly and steadily decrease IOP. Start by decreasing IOP by 5 mmHg—notice the stability and efficiency is unchanged. After you feel comfortable performing surgery at this pressure, drop it a little bit more and continue the cycle.
Dr. Sheybani: Looking at this from the glaucoma side of things, this just makes sense. Why would you want to raise the pressure higher than what is needed? I certainly want to be comfortable while having cataract surgery. For me, the main benefit of operating at physiologic IOP is that it takes away some of the risk variables for our more complex cases.
Dr. Hu: Good word of mouth is still the best practice builder and will bring us more reward than we can imagine. When we have less corneal edema, faster visual recovery, and better vision on day 1, that brings us happy patients and comanaging doctors who feel more confident referring patients to us. The industry has brought us new and better technology platforms, but ultimately, it is up to us as surgeons to incorporate them, to customize them, and to optimize them.
Dr. Lubeck: All these benefits lead to patients who understand and feel that they have had a superior procedure done. Cataract surgery performed at physiologic IOP gives the patient a positive experience and a positive perception of their outcome. I encourage all surgeons to have a mindset that allows evolution, growth, and progress in the way we think, so that going forward we become better surgeons.
© 2023 Alcon Inc. 05/23 US-CNT-2300014
1. Popovic M, Campos-Möller X, Schlenker MB, Ahmed II. Efficacy and safety of femtosecond laser-assisted cataract surgery compared with manual cataract surgery: a meta-analysis of 14,567 eyes. Ophthalmology. 2016;123(10):2113-26. doi: 10.1016/j.ophtha.2016.07.005. Epub 2016 Aug 15. PMID: 27538796.
2. Machiele R, Motlagh M, Patel BC. Intraocular Pressure. [Updated 2022 Jul 11]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK532237/https://www.ncbi.nlm.nih.gov/books/NBK532237/
3. 2020–2021 BCSC Basic and Clinical Science Course https://www.aao.org/bcscsnippetdetail.aspx?id=ec9e931c-3636-4b69-9736-dd22981445eb
4. Pardianto G. Recent awareness and consideration of intraocular pressure fluctuation during eye surgery. J Cataract Refract Surg. 2015;41(3):695.
5. Suzuki H, Oki K, Shiwa T, Oharazawa H, Takahashi H. Effect of bottle height on the corneal endothelium during phacoemulsification. J Cataract Refract Surg. 2009;35(11):2014-7. doi: 10.1016/j.jcrs.2009.05.057. PMID: 19878837.
6. Vasavada AR, Praveen MR, Vasavada VA, et al. Impact of high and low aspiration parameters on postoperative outcomes of phacoemulsification: randomized clinical trial. J Cataract Refract Surg. 2010;36(4):588-93. doi: 10.1016/j.jcrs.2009.11.009. PMID: 20362850.
7. Vasavada V, Raj SM, Praveen MR, Vasavada AR, Henderson BA, Asnani PK. Real-time dynamic intraocular pressure fluctuations during microcoaxial phacoemulsification using different aspiration flow rates and their impact on early postoperative outcomes: a randomized clinical trial. J Refract Surg. 2014;30(8):534-40. doi: 10.3928/1081597X-20140711-06. PMID: 25325894.
8. Kokubun T, et al. Verification for the usefulness of normal tension cataract surgery. Presented at: The 126th Annual Meeting of the Japanese Ophthalmological Society (JOS); Apr 14-17, 2022; Osaka, Japan.
9. Baradaran-Rafii A, Rahmati-Kamel M, Eslani M, Kiavash V, Karimian F. Effect of hydrodynamic parameters on corneal endothelial cell loss after phacoemulsification. J Cataract Refract Surg. 2009;35(4):732-7. doi: 10.1016/j.jcrs.2008.12.017. PMID: 19304097.
10. Takhtaev YV, Kiseleva TN, Shliakman RB. The effect of preset intraoperative intraocular pressure during phacoemulsification on the blood flow velocity in the central retinal artery. Ophthalmol J. 2019;(40):5-12. https://doi.org/10.17816/OV17802.
11. Chen D, Zhu J, Li J, Ding XX, Lu F, Zhao YE. Effect of simulated dynamic intraocular pressure on retinal thickness measured by optical coherence tomography after cataract surgery. Int J Ophthalmol. 2012;5(6):687-93. doi: 10.3980/j.issn.2222-3959.2012.06.07. Epub 2012 Dec 18. PMID: 23275902; PMCID: PMC3530809.
12. Yang HK, Park SJ, Byun SJ, Park KH, Hwang JM. Risk of Nonarteritic Anterior Ischemic Optic Neuropathy After Cataract Surgery. Am J Ophthalmol. 2019;207:343-350. doi: 10.1016/j.ajo.2019.08.001. Epub 2019 Aug 12. PMID: 31415735.
13. Vasavada V, Srivastava S, Vasavada V, et al. Impact of fluidic parameters during phacoemulsification on the anterior vitreous face behavior: Experimental study. Indian J Ophthalmol. 2019;67(10):1634-1637. doi: 10.4103/ijo.IJO_465_19. PMID: 31546498; PMCID: PMC6786211.
14. Han YK, Miller KM. Comparison of vacuum rise time, vacuum limitaccuracy, and occlusion break surge of 3 new phacoemulsification systems. J Cataract Refract Surg. 2009;35(8):1424–1429.
15. Malik PK, Dewan T, Patidar AK, Sain E. Effect of IOP based infusion system with and without balanced phaco tip on cumulative dissipated energy and estimated fluid usage in comparison to gravity fed infusion in torsional phacoemulsification. Eye (Lond). 2017;4(1). doi: 10.1186/s40662-017-0087-5
16. Suzuki H, Igarashi T, Takahashi H. Effect of a new phacoemulsification and aspiration handpiece on anterior chamber stability. J Cataract Refract Surg. 2023;49(1):91-96. doi: 10.1097/j.jcrs.0000000000001071. PMID: 36201661.
17. Alcon data on file.
18. O’Brien PD, Fulcher T, Wallace D, Power W. Patient pain during different stages of phacoemulsification using topical anesthesia. J Cataract Refract Surg. 2001;27(6):880-3. doi: 10.1016/s0886-3350(00)00757-4. PMID: 11408135.
19. Hou CH, Lee JS, Chen KJ, Lin KK. The sources of pain during phacoemulsification using topical anesthesia. Eye (Lond). 2012;26(5):749-50. doi: 10.1038/eye.2012.29. Epub 2012 Feb 24. PMID: 22361848; PMCID: PMC3351060.
CENTURION® Vision System Important Product Information
Caution: Federal (USA) law restricts this device to sale by, or on the order of, a physician.
As part of a properly maintained surgical environment, it is recommended that a backup IOL injector be made available in the event the AutoSert® IOL Injector Handpiece does not perform as expected.
Indication: The CENTURION® Vision system is indicated for emulsification, separation, irrigation, and aspiration of cataracts, residual cortical material and lens epithelial cells, vitreous aspiration and cutting associated with anterior vitrectomy, bipolar coagulation, and intraocular lens injection. The AutoSert® IOL Injector Handpiece is intended to deliver qualified AcrySof®intraocular lenses into the eye following cataract removal.
The AutoSert® IOL Injector Handpiece achieves the functionality of injection of intraocular lenses. The AutoSert® IOL Injector Handpiece is indicated for use with the AcrySof® lenses SN6OWF, SN6AD1, SN6AT3 through SN6AT9, as well as approved AcrySof®lenses that are specifically indicated for use with this inserter, as indicated in the approved labeling of those lenses.
Warnings: Appropriate use of CENTURION® Vision System parameters and accessories is important for successful procedures. Use of low vacuum limits, low flow rates, low bottle heights, high power settings, extended power usage, power usage during occlusion conditions (beeping tones), failure to sufficiently aspirate viscoelastic prior to using power, excessively tight incisions, and combinations of the above actions may result in significant temperature increases at incision site and inside the eye, and lead to severe thermal eye tissue damage.
Good clinical practice dictates the testing for adequate irrigation and aspiration flow prior to entering the eye. Ensure that tubings are not occluded or pinched during any phase of operation.
The consumables used in conjunction with ALCON® instrument products constitute a complete surgical system. Use of consumables and handpieces other than those manufactured by Alcon may affect system performance and create potential hazards.
AEs/Complications: Inadvertent actuation of Prime or Tune while a handpiece is in the eye can create a hazardous condition that may result in patient injury. During any ultrasonic procedure, metal particles may result from inadvertent touching of the ultrasonic tip with a second instrument. Another potential source of metal particles resulting from any ultrasonic handpiece may be the result of ultrasonic energy causing micro abrasion of the ultrasonic tip.
ATTENTION: Refer to the Directions for Use for the accessories/consumables and Operator’s Manual for a complete listing of indications, warnings, cautions and notes.







