The Mechanism and Presentation of DED
Brandon D. Ayres, MD: We have gathered for a roundtable discussion about the etiology of dry eye disease (DED) and how new data may redefine our understanding of its process and our clinical treatment strategies. In 2023, the American Academy of Ophthalmology updated its DED preferred practice guidelines to highlight increased tear evaporation as a core mechanism of tear film instability.1 We were taught that there are two primary forms of DED: evaporative dry eye and aqueous tear deficiency dry eye. We have since learned that evaporative DED is more prominent, with about 90% of our patients having either evaporative or mixed-mechanism disease.2 I see this preponderance of evaporative DED in my practice. What do you all see in your practices?
Damon Dierker, OD, FAAO: I agree; I think DED presents on a spectrum, and its diagnosis and treatment should be considered in this context. What I predominantly see in my practice might be called mixed-mechanism DED, but it’s all tear film instability, and it doesn’t exist in a silo.
Preeya K. Gupta, MD: Trying to tease out evaporative versus aqueous-deficient pathology can be complex, but we have assessment tools to help us make treatment decisions. I think it’s important to recognize evaporation and aqueous deficiency as important foundational categories of DED, but we need to address all signs and symptoms of the disease.
Jessica Steen, OD, FAAO: I find that most of my patients have predominantly evaporative DED. Treating these patients effectively begins with an accurate assessment and diagnosis, an understanding of the patient’s past experiences—both their successes and challenges with treatment—and understanding of the underlying disease processes in order to determine the best treatment options in a truly individualized way.
Dr. Ayres: DED is unlike other diseases in eye care because there is no single source of its etiology. What we see on the ocular surface is the end expression of multiple factors (Figure 1). It can be subdivided into different mechanisms and etiologies—meibomian gland dysfunction (MGD), environmental influences, nutritional deficiencies, etc. Even when we confirm excessive tear evaporation, underneath it, there’s still an inflammatory component (Figure 2). We must determine whether the inflammation is chronic or acute in order to prescribe the appropriate treatment.

Figure 1. DED is a vicious cycle with many possible entry points. 1. Bron AJ, et al. Ocul Surf. 2017;15(3):438-510. 2. Galor A, et al. Ocul Surf. 2023;28:262-303.
3. Pflugfelder SC, et al. Ophthalmology. 2017;124(11S):S4-S13. 4. Zhang R, et al. Ocul Surf. 2021;21:145-159.

Figure 2. Regardless of etiology, excess evaporation leads to desiccation stress and damage.
Adapted from Perez VL, et al. Exp Eye Res. 2020;201:108294 and from Pflugfelder SC, de Paiva S. Ophthalmology. 2017;124(11S):S4-S13.
The first ocular surface anti-inflammatory, cyclosporine, was approved in 2003.3 Since then, all anti-inflammatories (including XIIDRA® [lifitegrast ophthalmic solution 5.0%], the first pharmaceutical in the LFA-1 antagonist class, approved for the treatment of the signs and symptoms of DED in 2016), and immunomodulators have targeted inflammation, while tear stimulants have addressed aqueous deficiency. Thus, until recently, evaporative dry eye has not been specifically addressed by a prescription medication. We now have MIEBO (perfluorohexyloctane ophthalmic solution; Bausch + Lomb). MIEBO is the first FDA-approved medication to treat the signs and symptoms of DED that uniquely targets tear evaporation.4 Every patient in the MIEBO FDA clinical trial was selected for clinical signs of MGD, which I believe was a first. Study subjects had to have a history of DED and clinical signs of MGD, a key consequence of which is tear evaporation.

Expanding DED Treatment Awareness and Access
Dr. Ayres: DED is a problem among the US population. There are estimates that 38 million patients have DED,5,6 but only 1.2 million have a prescription medication,7 which means we have millions of potential patients who are self-diagnosing and are visiting their local pharmacy to choose a topical drop from the sea of OTC dry eye medications. Those who are not getting relief from ocular dryness with OTC drops should see an eye care professional. This is a huge opportunity for us to get patients into our offices for real, lasting treatment. Left untreated, DED can progress, with symptoms worsening over time. We need to educate our patients that they don’t need to suffer with these symptoms—we have the tools to help them.
Dr. Dierker: We must screen for DED in all patients, referred and routine, and we need to agree on a protocol for diagnosing and treating DED based on signs and symptoms. How can I counsel my cataract surgery team if I don’t know what a patient’s ocular surface looks like? Likewise for glaucoma, the decision to choose an eye drop, or a laser, or a MIGS procedure will be partially based on the health of their ocular surface. In my population in Indiana, DED is more common than not. I think there is a huge opportunity to educate the public about DED and offer effective treatment options. The patients who I put on topical prescription DED drops are so thankful to get relief. It feels great whenever I can help a patient who’s tried and failed other approaches.
Dr. Gupta: I frequently encounter patients who are frustrated by their chronic symptoms, and they tell me they’ve seen three doctors or that they’ve tried every eye drop available at their local pharmacy. So, there’s a little bit of a disconnect as to why these therapies we now have are not reaching those patients. I think we should start patients on a prescription topical therapy for DED more regularly. MIEBO and XIIDRA each have a favorable safety profile, and I trust in the FDA’s evaluation and approval process.
Dr. Steen: In my practice where most patients are using chronic topical ocular therapeutics related to the management of glaucoma, I find that addressing the ocular surface and the resulting improvement in the signs and symptoms of DED are key drivers to improve adherence in patients with both DED and glaucoma.
MIEBO Efficacy and Practice Patterns
Dr. Ayres: MIEBO® (perfluorohexyloctane ophthalmic solution) has changed my practice pattern. I used to automatically prescribe an anti-inflammatory medication for any signs of DED. Now, if I see any signs of excessive tear evaporation, I prescribe MIEBO. It is the only FDA-approved DED medication that directly targets ocular surface evaporation. How do you each approach DED patients, and how might using MIEBO change your practice patterns?
Dr. Steen: Having the first and only approved medication to directly target tear evaporation has been a real call to action for eye care practitioners.
Dr. Dierker: There’s no doubt that anti-inflammatory therapies will continue to be a mainstay in DED management. Yet, having a topical drop that can reduce the rate of evaporation from the ocular surface could potentially qualify a greater number of candidates for treatment. With MIEBO now available, I would advise colleagues to avoid waiting until a patient’s DED worsens to the point of corneal staining before prescribing a medication. For me, visually significant corneal staining is a low bar; often, the frequent use of artificial tears is enough for me to suggest a prescription medication.
Dr. Gupta: Having a pharmacological therapy that targets evaporation is a game-changer for how I approach patients. A treatment that can restrict tear evaporation is, to me, an important component in the DED treatment paradigm.
Dr. Dierker: The patient population recruited in the clinical trials for MIEBO is generally representative of what we see in our practices. Clinically, I see symptomatic patients who have MGD, obstruction in the meibomian glands, reduced tear break-up time (TBUT), and a compromised tear volume. Having a therapeutic that can address the signs I find is very helpful.
Dr. Gupta: Because no two dry eye patients come in with the same disease process, having treatments that target different pathways is so helpful to us as clinicians. Additionally, our treatments need to be effective and tolerable for patients. In its clinical trials, MIEBO demonstrated a clinically significant improvement in both the signs and symptoms of DED in as little as 2 weeks and through day 57.
In two phase 3 clinical studies, researchers evaluated the safety and efficacy of MIEBO for the treatment of DED. The GOBI8 and MOJAVE9 studies demonstrated that MIEBO works within 2 weeks, as noted above: there was a change in total corneal fluorescein staining from baseline at days 15 (secondary endpoint) and 57 (primary endpoint) (Figure 3). Similarly, MIEBO demonstrated a change from baseline in visual analog scale (VAS) dryness score at Days 15 (secondary endpoint) and 57 (primary endpoint) (Figure 4).

Figure 3. In its phase 3 clinical trials (GOBI1 and MOJAVE2), MIEBO demonstrated improvement in total corneal fluorescein staining (tCFS) as early as day 15 and sustained through day 57. In the pooled dataset, the mean baseline tCFS for both MIEBO (n=614) and the control (saline; n=603) was 6.9. MIEBO recipients gained twice the improvement in corneal staining as the control by day 57 (the primary endpoint). (1. Tauber J, et al. Ophthalmology. 2023;130(5):516-524. 2. Sheppard JD, et al. Am J Ophthalmol. 2023;252:265-274.)
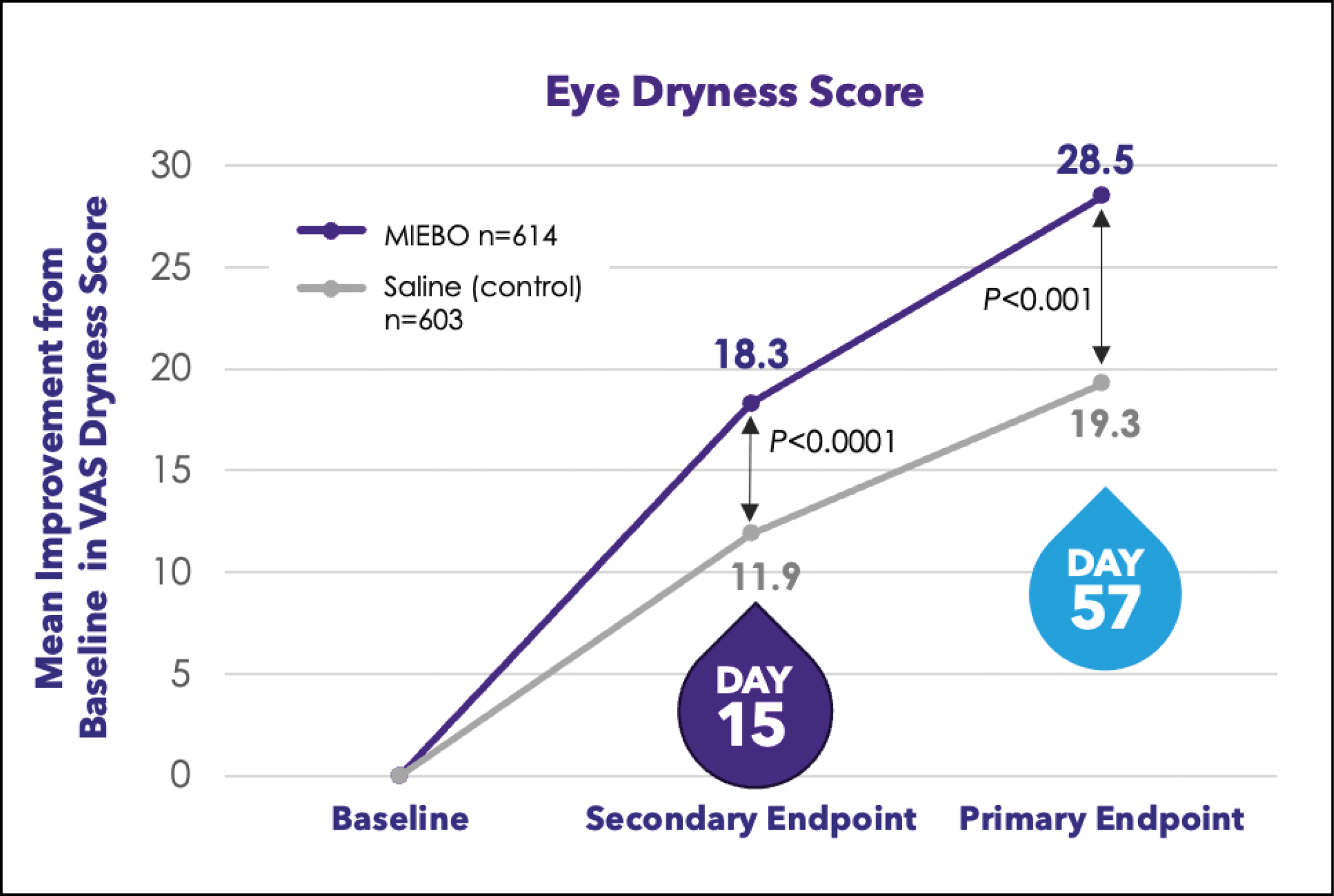
Figure 4. In its phase 3 clinical trials (GOBI1 and MOJAVE2), MIEBO demonstrated relief from ocular dryness as early as day 15 and sustained through day 57. In the pooled dataset, the mean baseline eye dryness score on the Visual Analog Scale (VAS) was 65.6 for MIEBO (n=614) and 65.5 for saline (n=603). By day 57 (the primary endpoint), MIEBO recipients showed an improvement in ocular dryness that was 1.5 times greater than the saline control. (1. Tauber J, et al. Ophthalmology. 2023;130(5):516-524. 2. Sheppard JD, et al. Am J Ophthalmol. 2023;252:265-274.)
Furthermore, MIEBO proved to be user-friendly: patients in the studies reported that it was comfortable to use, and the most common adverse events were blurred vision upon instillation, which was mild and transient, and conjunctival redness which occurred at an incidence of 1-3%.4
Dr. Ayres: It’s also important to remember that inflammation underlies DED. Fortunately, we now have two topical treatments: MIEBO® (perfluorohexyloctane ophthalmic solution) to address tear evaporation, and XIIDRA® (lifitegrast ophthalmic solution) 5.0% to address chronic inflammation. These therapies allow us to treat our DED patients in a more targeted fashion.
The Inflammatory Component of DED
Dr. Ayres: Inflammation of the ocular surface has been the topic of DED for years. Early-generation anti-inflammatories were approved only to improve tear production, not necessarily to address the signs and symptoms of DED. Since 2016, we have had XIIDRA, which was uniquely designed as a topical anti-inflammatory to treat signs and symptoms of DED. While the exact mechanism of action is unknown, it is the only prescription therapy for signs and symptoms of DED that addresses active and inactive T-cells.10,11 Why is this impactful for treating DED?
Dr. Dierker: Our patients want symptomatic relief quickly. Thus, XIIDRA was game-changing—suddenly, I could say to my patients, “This drug designed specifically to treat dry eye disease has been approved for symptoms.” XIIDRA changed the conversation for me.
Unchecked tear film instability will eventually lead to inflammatory DED. When I identify patients who have inflammation, they often have conjunctival hyperemia and elevated levels of the inflammatory marker MMP-9. When I examine these patients’ lid margins, the need to address the inflammatory component is clear. XIIDRA remains the only topical nonsteroidal anti-inflammatory that can improve symptoms in as little as 2 weeks, as seen in 2 out of 4 clinical studies.10
Dr. Steen: The mechanism of action of XIIDRA is unique. It is able to disrupt the pathophysiology of the disease process on the immunomodulation level, and at a specific location, to impact not just the inactive T-cells but also the previously activated T-cells. I know XIIDRA is the only non-steroid prescription that can do that.
Dr. Gupta: I think the mechanism of action of XIIDRA enables it to work fast—in 2 out of 4 clinical trials, it was shown to be effective in as early as 2 weeks.10 Also, XIIDRA can inhibit migration of T-cells back to the ocular surface, thereby suppressing the inflammatory reaction early, and I think that’s what really makes XIIDRA unique.
Dr. Ayres: I, too, like how fast XIIDRA can work. It is the only non steroid anti-inflammatory that has shown the ability to provide symptom relief in as little as 2 weeks in pivotal trials.10 And, while the exact mechanism of action is not known, it works on both active and inactive T-cells to decrease inflammation: its ability to block the interaction of lymphocyte function-associated antigen 1 (LFA-1) and intercellular adhesion molecule 1 (ICAM-1) not only suppresses the immune system from activating T-cells that are already circulating, it also suppresses already active T-cells from being drawn into the inflammatory site.10

The Mechanism of Action of MIEBO and XIIDRA
Dr. Gupta: Here, we are going to talk about the mechanism of action of both MIEBO and XIIDRA. Dr. Ayres, how does MIEBO work?
Dr. Ayres: I am impressed by MIEBO. MIEBO is a single-ingredient medication, perfluorohexyloctane ophthalmic solution, that protects the tear film from evaporation. With this treatment, within 2 weeks we see improvement in the signs and symptoms of DED.
Dr. Steen: What makes MIEBO so unique is the molecule itself. It has two different components that make the molecule align in a specific way. The fluorinated component orients itself out into the environment, and the other component of the molecule inserts itself into the lipid layer. This creates a monolayer on the ocular surface that helps reduce evaporation, and so can help to improve tear film homeostasis and thus protect the ocular surface (Figure 5).

Figure 5. MIEBO forms a monolayer where the ocular tears meet the air to prevent surface evaporation.
Dr. Dierker: MIEBO® (perfluorohexyloctane ophthalmic solution) created a new category of medication for signs and symptoms of DED. We know that excessive evaporation is almost universal in such patients, and now we have this single-ingredient, nonsteroidal, unpreserved, comfortable drop that targets evaporation.
Dr. Gupta: When we think about evaporation, we should consider the function of the meibum, which is to protect the ocular surface from tear evaporation. Something Dr. Steen said is very important to understand: perfluorohexyloctane, or PFHO, is a very different molecule than we’re used to. It spreads across the ocular surface and helps to limit evaporation by forming a long-lasting monolayer. As I learned about the mechanism of MIEBO, it challenged me to think of other things we can do that help to limit tear evaporation. Has anyone else experienced an “aha” moment of understanding the tear film or how we approach it?
Dr. Dierker: MIEBO works differently than any other topical medication we’ve had. Other topical molecules must penetrate the tear film to reach the cornea and conjunctiva to work effectively. MIEBO inserts in the tear film, into the lipid layer, to provide that evaporative barrier and help prevent moisture from getting out. It’s a different way to look at its mechanism of action.
Dr. Steen: From a mechanistic standpoint, we usually take one of two approaches to the management of any disease process: we find the area of pathology and disrupt or alter that pathway in some way; or, as with MIEBO, we mimic the anti-evaporative function of natural meibum.
Dr. Gupta: The durability of MIEBO is really meaningful to me as a clinician, and I think it resonates with patients. MIEBO remains present on the ocular surface for at least 6 hours in PK rabbit studies, whereas artificial tears typically dissipate within a few minutes. The clinical significance of this is unknown.
Dr. Dierker: I use that same talking point with my patients, and it usually gets them on board with MIEBO. I explain that OTC lubricant eyedrops tend to only provide temporary relief.
Dr. Steen: Once the patient has started MIEBO therapy and then returns for that first follow-up visit, I always ask how their use of artificial tears has changed (I use the SPEED questionnaire to capture this subjective information). To me, a reduction in use of artificial tears is one indicator of the success of the treatment. For those who continue to use artificial tears, I counsel them not to instill artificial tears immediately after MIEBO, because they could potentially impact the effectiveness of MIEBO. It’s also important to note that MIEBO should not be administered while wearing contact lenses. Contact lenses should be removed before use and for at least 30 minutes after administration of MIEBO.
XIIDRA
Dr. Gupta: Let’s now talk about XIIDRA® (lifitegrast ophthalmic solution) 5.0%, an LFA-1 antagonist. Dr. Dierker, please tell us how XIIDRA works.
Dr. Dierker: XIIDRA is an anti-inflammatory immunomodulator that targets a very specific interaction in patients with DED. In the presence of T-cell–mediated inflammation, intercellular adhesion molecule-1 (ICAM-1) is overexpressed on the ocular surface. On these T-cells, the lymphocyte function-associated antigen-1 (LFA-1) binds with ICAM-1, causing a cytokine storm. The molecule in XIIDRA is an antagonist to LFA-1 that blocks that interaction (Figure 6). Also, it impacts both active and inactive T-cells. XIIDRA is FDA-approved to treat the signs and symptoms of DED, and importantly, it gave some patients symptom relief within 2 weeks as seen in 2 out of 4 studies. It is the first immunomodulator to target a very specific part of the inflammatory pathway, as well as T-cells that are both active and inactive.

Figure 6. How XIIDRA blocks the cytokine storm caused by the interaction of ICAM-1 and LFA-1 in DED. The exact MOA is unknown.
1. XIIDRA prescribing information 2. Gao et al. Exp Eye Res. 2004;78(4):823-835. 510.
Dr. Gupta: There are many immunomodulators for us clinicians to choose from to treat DED. How do you know what to choose for your patient?
Dr. Steen: For me, it comes back to the clinical data to support each therapeutic option. In patients with clear signs of inflammation, the clinical data to support the onset of symptom relief in as little as 2 weeks, combined with an appropriate tolerability profile (Figure 7), is something that sways patients toward XIIDRA especially for those who have tried multiple products and seen multiple doctors.

Figure 7. XIIDRA was well-tolerated across five clinical studies.
1. XIIDRA prescribing information. 2. Nichols KK, et al. Eur J Ophthalmol. 2019;29(4):394-401.
Dr. Ayres: Because there are multiple mechanisms of action contributing to DED, I like to conduct thorough preoperative testing—including tear film osmolarity, MMP-9, evaluating the meibomian glands and TBUT—to see if I can pinpoint where the dysregulation is. These data help me discern whether the patient needs help with the evaporative component or the inflammatory side of DED, or both. I find that exploring the ocular surface for this information is an exciting new frontier in the ocular surface world.
Dr. Dierker: I agree with Dr. Ayres about our approach to DED management, and I don’t think we have to overthink it. MIEBO® (perfluorohexyloctane ophthalmic solution) and XIIDRA® (lifitegrast ophthalmic solution) 5.0%, work differently; their mechanisms of action are targeting ocular surface evaporation and inflammation, respectively. Yet, they’re both labeled to treat the signs and symptoms of DED, and either one is an appropriate therapy for many of our patients.
I think we can simply ask ourselves: are this patient’s eyes clearly inflamed, or does the patient have a pro-inflammatory systemic disease that would indicate XIIDRA as the first-line therapy? Or, is this a patient who is in front of a computer all day, or who wears contact lenses, or experiences some other circumstance that will trigger evaporative stress in the ocular surface? They may not have a lot of systemic inflammation contributing to their ocular surface disease. We can treat them now with MIEBO. I think there’s clearly room for both products, thanks to their labeling.
Dr. Gupta: Absolutely, there’s room for both medications. When it comes to selecting which immunomodulator I like to use, I am compelled by the symptom data. The reason I reach for lifitegrast often in my practice is because it works on both active and inactive T-cells. T-cells that are circulating in the blood have a 90-day life cycle. Traditional molecules like cyclosporine only work on the production of new T-cells, which is why patients often take a long time to respond to these older medications, because they still have circulating T-cells. XIIDRA works on T-cells that are in circulation, and it also slows down the activation of new T-cells. I think that is why we see symptom improvement as early as 2 weeks in some patients with XIIDRA.

Clinical Considerations for Managing DED Patients
Dr. Dierker: Let’s discuss the nuts and bolts of addressing DED in the clinic. How do we approach the dry eye patient? What does that workup look like? How do we choose between or combine our topical therapeutics?
Dr. Ayres, we’ll start with you. Our practice settings may differ, but we’re seeing patients with DED all day, every day. How are you screening for signs and symptoms of DED in your patient population?
Dr. Ayres: When a patient presents to our office, we begin by giving them a questionnaire (modified from validated studies) that asks them about fluctuations in their vision and other such symptoms. When it comes time for the physical examination, our technicians are trained to look for certain key signs, such as irregularity on a patient’s topographic maps, that require my assessment.
The patients who have not received a previous diagnosis of DED are usually surprised when we tell them we will prioritize improving those symptoms before we address the issue they originally came in for.
Dr. Dierker: Dr. Steen, with your glaucoma-based population, how are you assessing signs and symptoms and making the differential diagnosis of DED? What does your screening process look like?
Dr. Steen: Most of my patients present for ongoing care related to a glaucoma diagnosis, or are referred for assessment for suspicion of glaucoma, or are seeking a second opinion related to treatment or management of glaucoma. However, whether the patient is already on topical glaucoma therapy or needs to begin it, my team and I ensure that their ocular surface is optimized to allow for healthy ongoing topical therapy with maximal adherence.
Similar to Dr. Ayres, I use a validated questionnaire, the SPEED questionnaire. I also include fluorescein corneal staining, lissamine green conjunctival staining, and a careful evaluation of the eyelids and tear meniscus height.

Dr. Gupta: My routine protocols are very similar. I think the key is being consistent in how we approach these patients. If I could only pick two things to screen for DED, I’d ask the patient if they were using an artificial tear, because a lot of patients don’t consider this a form of treatment. In my opinion, if somebody is going out of their way to use drops, then they have real disease.
Secondly, using a 10-cent fluorescein strip gives me a wealth of information. I can assess the TBUT, look for corneal staining (Figure 8), examine the pattern of the dye across the surface, and check if there are any contributing lid abnormalities. I use the strip the same way every single time for all types of patients who come into my practice, and it has helped me to find the disease quite often.

Figure 8. Dr. Gupta uses fluorescein staining to assess the
health of the ocualr surface.
Courtesy of Preeya Gupta, MD
Dr. Dierker: I agree with Dr. Gupta that we all need an efficient, reproducible protocol to screen for DED in each patient we see. It sounds like we all use targeted questions and look for symptoms, because DED is a symptomatic disease. We also look for loss of corneal homeostasis, whether that’s increased tear osmolarity or putting fluorescein in the eye and looking at the TBUT or ocular surface staining.
Dr. Steen, you mentioned that DED often starts from abnormalities in the eyelid, whether it’s inflammation, obstruction, or both. How do you assess eyelids in your clinic?
Dr. Steen: I have a methodical approach to assessing the meibomian glands, specifically. It starts with looking at the lid itself. Is the eyelid positioned where I expect it to be? Is there a significant entropion, ectropion, or eyelid laxity? Looking at the tear meniscus height is something I find truly valuable, as well. Then, I push on the lower lid, starting just nasal to the center of the eyelid with firm pressure on the tarsal plate to see if there is meibomian gland secretion and to assess the quality of the secretion. Does it look healthy, is it turbid, thickened, or absent?
Dr. Ayres: Expressing the meibomian glands gives us so much information about the patient’s ocular surface health. At Will’s Eye Hospital, we’re all trained to use cotton tip applicators, even if someone doesn’t have a single symptom of DED. For every single patient, I put a little pressure on the tarsal plate and note the meibomian gland secretions.
Dr. Gupta: Dr. Ayres and I were both on the Cornea Clinical Committee for the ASCRS, and we came up with a little mnemonic device—LLPP—to evaluate the ocular surface efficiently. LLPP stands for: look at the lids; lift the lids to look for any corneal pathology or superior conjunctival pathology; and then pull and push. (Pull the upper lid looking for laxity. Push refers to pushing the meibomian glands to look at their secretion). The mnemonic device helps us remember to include it in each examination.
Dr. Dierker: I like that: look down, look up, press—those steps may only take 5 or 10 seconds. To ignore the eyelids as part of a comprehensive examination is to ignore one of the most common reasons patients schedule time to see us: because they have sequela related to an eyelid problem.
Dr. Ayres: As I alluded to previously, I often diagnose DED in a patient while I’m evaluating them for another reason. Now the patient has two diagnoses, and my priority is to rapidly rehabilitate the ocular surface. If their DED is primarily inflammatory, I will reach for XIIDRA® (lifitegrast ophthalmic solution) 5.0%. If I think it’s primarily evaporative, I will reach for MIEBO® (perfluorohexyloctane ophthalmic solution). That patient will begin these treatments and then transition back to their primary eye care team for follow-up until their ocular surface is healthy enough for surgery. If they don’t continue that therapy, we won’t see improvement. How do each of you communicate the need for ongoing DED management to your referring physicians?
Dr. Gupta: I think it starts with the patient. I always try to impress on the patient all the things that are causing their blurred vision and the need for ongoing treatment. The patient’s buy-in is primary.
With referring physicians, I believe in maintaining open lines of communication. All of my referring doctors have my cell phone number and our office number if there’s a question or problem. Yet, I still see patients who have discontinued their therapy.
Dr. Dierker: I think you’ve hit on it: it’s consistency. We have to make patients’ ocular health a priority. I think there’s an opportunity for both optometrists and ophthalmologists to do a better job with that.
CASE STUDY
Dr. Steen: We’ve had an opportunity to talk about DED, how each of us diagnoses the disease process, and our approaches to treatment strategies. Now, we get to bring it home and talk about specific patient cases. Dr. Ayres, tell us about a particular case that you treated recently.
Severe OSD Before Cataract Surgery
Dr. Ayres: This is a patient whom my team and I have been treating for a long time, and her presentation will sound familiar to many of you. She was interested in gaining better vision through cataract surgery, but she presented with myriad ocular surface problems, starting with severe anterior basement membrane corneal dystrophy (Figure 1).

Figure 1. The preoperative clinical examination revealed a high degree of corneal staining.
This patient’s medical history included previous keratectomies to remove the surface irregularities, which uncovered significant OSD, primarily evaporative (Figure 2). We told this individual that we’d have to delay her cataract surgery until her ocular surface was healthy enough.

Figure 2. The patient’s preoperative topography showed high levels of irregular astigmatism due to OSD.
She exhibited true DED—not just punctate epithelial erosions along the inferior cornea, but irregular corneal topographies right up into the visual axis. When this patient began her journey, we didn’t have MIEBO® (perfluorohexyloctane ophthalmic solution), so I originally prescribed a typical course of anti-inflammatory medication, but her TBUT remained at approximately 5 seconds with corneal staining. I switched her to MIEBO once it became available, and I saw a significant improvement within just a few weeks, not only in her symptoms, but importantly, in her clinical signs. I was finally able to say that this patient was a candidate for cataract surgery. In cases of evaporative DED like this, I am now using MIEBO as an early intervention. It really helps improve the ocular surface so that patients like this one can move toward their ultimate goal.
Dr. Dierker: Clearly, you had identified this patient as having visually significant OSD, which is an important distinction. Topographic changes due to DED may impact surgical planning. Could this patient have been a candidate for XIIDRA® (lifitegrast ophthalmic solution) 5.0%, as well? XIIDRA has been shown to improve biometric and keratometric measurements in as little as 4 weeks. I’m interested why you chose MIEBO in this case, because I think you could make the argument for either medication.
Dr. Ayres: This patient had already been on immunomodulators in the past, and she had not experienced enough of an improvement. I think that using an anti-inflammatory in this patient was a necessary step, but it wasn’t sufficient to get her across the finish line. Thus, I switched to MIEBO to reach that goal.
Dr. Steen: How is this patient going to be managed postoperatively, long-term?
Dr. Ayres: This patient did well after her surgery (Figure 3). She gained a significant improvement in her vision, but we cannot back off on the surface treatment, or else her corneas may regress. She already knows she has DED that requires ongoing management, but we try to mitigate symptoms as much as possible.

Figure 3. One month after the patient initiated MIEBO drops, her corneal topography was much improved.
Treatment Strategies
Dr. Dierker: Let’s say we have a patient who has signs and symptoms of DED in the presence of MGD. How do you determine your treatment strategy between evaporative therapy and inflammatory therapy?
Dr. Gupta: First, we must determine whether the patient is currently using any treatments or not. Then, corneal staining is key, to me—it means that they have moderate-level disease or worse. My team and I don’t accept corneal staining in our perioperative patients, and we shouldn’t accept corneal staining in our routine care, either, because we know it’s going to compromise patients’ vision.
When I see corneal staining, I anticipate an excessive amount of inflammation on the ocular surface. If this patient is not currently using any therapeutic, I’ll tell them that we’ll start with one topical medication and keep adding drops until the corneal staining resolves. But then, there’s also the symptomatic component.
If there are a significant number of inflammatory signs—redness, breakdown of the corneal epithelium, staining of the conjunctiva—these are among the early findings of inflammatory DED. For those patients, I tend to lean toward the immunomodulator pathway as my initial therapy. Yet, many patients need more than one therapy, and you can’t ignore the evaporative component, as that may be an equal contributor to their disease process.

Dr. Dierker: Dr. Steen, what are the things in the patient’s history, treatment goals, or clinical presentation that would make you choose MIEBO® (perfluorohexyloctane ophthalmic solution) as their first treatment?
Dr. Steen: As Dr. Gupta mentioned, the patient’s history is important, and I also think about it from a systemic perspective. What systemic medications are they taking? What systemic conditions do they have that may have an impact on the immune system and may exacerbate signs and symptoms of dry eye disease? For example, diabetes mellitus is important to recognize, because of its potential impact on the ocular surface.
I ask about fluctuation in vision, especially toward the end of the day, to indicate too much evaporation. I also clinically assess for meibomian gland dysfunction, reduced TBUT, and I pay attention to the pattern of the tear break-up.
One of the highlights of the clinical trials assessing the safety and efficacy of MIEBO was its tolerability profile (Figure 9). For patients who may have stinging and burning upon instillation of other eye drops,12 including artificial tears, I find that MIEBO is a medication that is comfortable.

Figure 9. In two pivotal clinical trials (phase 3 GOBI1 and phase 3 MOJAVE2), a total of 1217 patients were randomized 1:1 to receive either MIEBO (n=614) or hypotonic (0.6%) saline (n=603) QID for 8 weeks. Most ocular AEs were considered mild (most common was blurred vision), and discontinuation rates due to AEs for MIEBO were comparable to the control (pooled: 0.2% vs 0.5%, respectively). In total, one patient in the MIEBO arm (out of 614) discontinued due to eye irritation. (1. Tauber J, et al. Ophthalmology. 2023;130(5):516-524. 2. Sheppard JD, et al. Am J Ophthalmol. 2023;252:265-274. 3. Protzko EE, et al. Cornea. 2024;43(9):1100-1107.).
Dr. Dierker: I agree that fluctuating vision has been a gap in our patients whose symptoms worsen later in the day. This symptom is common with evaporative dry eye. I also agree that MIEBO is very well tolerated, both in the clinical studies and in my patient population. It works quickly, too—within a couple of weeks.
Dr. Ayres, what does a candidate for XIIDRA® (lifitegrast ophthalmic solution) 5.0% look like for you and your practice?
Dr. Ayres: Inflammation is the core of DED. When we clinicians are trying to decide whether to treat the inflammation or the evaporation first, for me, it comes down to clinical testing and the physical examination. The inflamed eye may have superficial punctate keratitis. Remember, a patient with an epitheliopathy anywhere on the cornea deserves an anti-inflammatory. That was the clinical rule I learned in training and still follow.
The question of what to start patients on is becoming a bigger issue. Very few patients these days are on a single medication, and polypharmacy in DED is very common. This may include a prescription anti-inflammatory and something like MIEBO® (perfluorohexyloctane ophthalmic solution), or maybe it’s an autologous serum plus amniotic cytokine extract drops. I am very quick to prescribe an immunomodulator such as XIIDRA® (lifitegrast ophthalmic solution) 5.0%. XIIDRA works well, and it’s been proven in multiple studies. If inflammation is the key part of a patient’s DED, I’m going to start with XIIDRA.
Dr. Dierker: Dr. Gupta, how do XIIDRA and MIEBO mesh with other DED therapies patients may be using: nutritional supplements, in-office therapies with light or thermal applications, etc.?
Dr. Gupta: My staff and I do a lot of interventional procedures in the office for MGD, and I think they are complementary for the appropriate patients. I love doing those procedures to jumpstart the function of the glands and support the anti-evaporative effects of MIEBO. For patients who have mild disease, a topical therapy may be all they need. Some patients need the combination of a pharmaceutical and an adjunctive procedure, and some patients need all three categories of treatments you mentioned. Managing DED is a journey we take with the patient over time.
Dr. Ayres: Until we had MIEBO, we had no prescription eye drop that addressed the excessive tear evaporation caused by MGD. Interventional dry eye therapy is still a big part of my practice, as is using MIEBO. They are not mutually exclusive.
Dr. Dierker: It’s the same for me. And, if I’ve started a patient on an in-office DED treatment and I still see inflammation or poor-quality meibomian gland secretions on follow-up, it’s very easy to add XIIDRA or MIEBO. Although there is no clinical study on the simultaneous use of MIEBO and XIIDRA, they are not contraindicated.
© 2025 Bausch + Lomb MBO.0706.USA.24
1. Amescua G, Ahmad S, Cheuyng AY, et al; American Academy of Ophthalmology Preferred Practice Pattern Cornea/External Disease Committee. Dy eye syndrome PPP 2023. Accessed July 30, 2024. https://www.aao.org/education/preferred-practice-pattern/dry-eye-syndrome-ppp-2023.
2. Lemp MA, Crews LA, Bron AJ, et al. Cornea. 2012;31:472-478.
3. Restasis approval package. Center for Drug Evalaution and Research.
Accessed July 30, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2003/21-023_Restasis_Approv.PDF.
4. MIEBO. Prescribing Information. Bausch & Lomb, Inc; 2023. Accessed August 27, 2024. https://pi.bausch.com/globalassets/pdf/packageinserts/pharma/miebo-package-insert.pdf
5. Paulsen AJ, Cruickshanks KJ, Fischer ME, et al. Dry eye in the beaver dam offspring study: prevalence, risk factors, and health-related quality of life. Am J Ophthalmol. 2014;157(4):799-806.
6. Farrand KF, Fridman M, Stillman IÖ, Schaumberg DA. Prevalence of diagnosed dry eye disease in the United States among adults aged 18 years and older. Am J Ophthalmol. 2017;182:90-98.
7. Karpecki PM, Nichols KK. Addressing excessive evaporation: an unmet need in dry eye disease. Supplements and featured presentations. Am J Manag Care. 2023; 29:S239-S247. Accessed August 27, 2024. https://doi.org/10.37765/ajmc.2023.89448
8. Tauber J, Berdy GJ, Wirta DL, et al. NOV03 for dry eye disease associated with meibomian gland dysfunction: Results of the randomized phase 3 GOBI study. Ophthalmology. 2023;130(5):516-524.
9. Sheppard J, Kurata F, Epitropoulos AT, et al. NOV03 for signs and symptoms of dry eye disease associated with meibomian gland dysfunction: The randomized phase 3 Mojave study. Am J Ophthalmology. 2023;252:265-274.
10. XIIDRA. Prescribing information. Bausch + Lomb, Inc.; 2020. Accessed August 27, 2024. https://www.novartis.com/us-en/sites/novartis_us/files/xiidra.pdf
11. Perez VL, Pflugfelder SC, Zhang S, et al. Lifitegrast, a novel integrin antagonist for treatment of dry eye disease. Ocul Surf. 2016;14(2):207-215.
12. de Paiva CS, Pflugfelder SC, Ng SM, et al. Editor: Cochrane Eyes and Vision Group. Topical cyclosporine A therapy for dry eye syndrome. Cochrane Database Syst Rev. 2019(9):CD010051.
INDICATION
Xiidra® (lifitegrast ophthalmic solution) 5% is indicated for the treatment of signs and symptoms of dry eye disease (DED).
IMPORTANT SAFETY INFORMATION
• Xiidra is contraindicated in patients with known hypersensitivity to lifitegrast or to any of the other ingredients.
• In clinical trials, the most common adverse reactions reported in 5-25% of patients were instillation site irritation, dysgeusia and reduced visual acuity. Other adverse reactions reported in 1% to 5% of the patients were blurred vision, conjunctival hyperemia, eye irritation, headache, increased lacrimation, eye discharge, eye discomfort, eye pruritus and sinusitis.
• To avoid the potential for eye injury or contamination of the solution, patients should not touch the tip of the single-use container to their eye or to any surface.
• Contact lenses should be removed prior to the administration of Xiidra and may be reinserted 15 minutes following administration.
• Safety and efficacy in pediatric patients below the age of 17 years have not been established.
Please see accompanying full Prescribing Information for XIIDRA
MIEBO® (perfluorohexyloctane ophthalmic solution)
INDICATION
MIEBO® (perfluorohexyloctane ophthalmic solution) is indicated for the treatment of the signs and symptoms of dry eye disease.
IMPORTANT SAFETY INFORMATION
- MIEBO is contraindicated in patients with known hypersensitivity to perfluorohexyloctane
- MIEBO should not be administered while wearing contact lenses. Contact lenses should be removed before use and for at least 30 minutes after administration of MIEBO
- Instruct patients to instill one drop of MIEBO into each eye four times daily
- The safety and efficacy in pediatric patients below the age of 18 have not been established
- In pivotal trials, the most common ocular adverse reaction was blurred vision (1% to 3% of patients reported blurred vision and conjunctival redness)
Click here for full Prescribing Information for MIEBO.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit MedWatch: FDA Safety Information & Adverse Event Reporting Program or call 1-800-FDA-1088.



