
Cataract surgery is one of the most safe and effective procedures performed worldwide. The inflammatory response to surgery, however, can trigger a cascade of complications such as retinal edema, the presence of proinflammatory cells in the anterior chamber, and fibrosis.1 Additionally, patients can experience poor surgical outcomes, delayed healing, discomfort, and pain when inflammation is not controlled postoperatively. In extreme cases, postoperative inflammation can cause cystoid macular edema (CME) and even vision loss after cataract surgery.2
The prevention of postoperative inflammation-related complications therefore is a crucial element of successful cataract surgery. By eliminating the potential cascade of complications, we create the opportunity for patients to experience quick visual recovery and a comfortable postoperative course.
OMIDRIA PROVIDES EXTRA REASSURANCE
The addition of antiinflammatories such as steroids and NSAIDs to cataract surgery probably single-handedly ushered the procedure into the 20th century. Now, several intraoperative and sustained-release technologies are available to further increase the effectiveness of inflammation control during cataract surgery.
In my practice, patients generally start using an NSAID 2 days before surgery to saturate the cyclooxygenase (COX)-1 and COX-2 receptors. On the day of surgery, phenylephrine and ketorolac intraocular solution 1% / 0.3% (OMIDRIA®, Rayner) is added to the phacoemulsification irrigating solution to provide continuous intracameral administration. This helps provide extra reassurance that the receptors we spent 2 days saturating with the topical NSAID remain saturated, reducing the inflammatory response to both routine and unexpected surgical manipulations during cataract surgery.
Without OMIDRIA, the COX-1 and COX-2 receptors become desaturated during surgery. If enough receptors are exposed and activated, an inflammatory response is triggered and the risk for complications such as CME increases. Postoperatively, OMIDRIA stays inside the eye for at least 10 hours, keeping those receptors bound and protected.3
THE ROLES AND ADVANTAGES OF OMIDRIA
A 4-mL vial of OMIDRIA is placed in 500 mL of ocular irrigating solution, allowing continual intracameral administration throughout surgery. I begin by injecting OMIDRIA solution at the beginning of the case through my paracentesis prior to initiating any intraocular manipulations. I then inject lidocaine followed by an OVD before entering the eye through the primary incision.
I use OMIDRIA in all of my cataract surgery cases because it facilitates a more successful procedure and decreases the risk for unexpected miosis and other complications such as IFIS and breakthrough iritis.4,5 It also reduces the need for pupil expansion devices and overall makes my surgeries go smoother.6
Intraoperative miosis. Cataract surgeons commonly deal with intraoperative miosis, which can decrease visualization and increase the risk for complications such as iris capture and capsular rupture. Although it is unpredictable, about 25% of patients with no identified risk factors will experience intraoperative miosis.7 OMIDRIA helps me stay ahead of the unexpected. I know that I will have maximal visibility during the procedure because it provides a well-maintained dilated pupil.
The use of intracameral OMIDRIA also reduces the need for a pupil expansion device.6 This is advantageous because these devices can damage the iris and cause increased inflammation.8 Their use also increases the duration of the procedure. Additionally, there is a cost savings to the ambulatory surgery center when a pupil expansion device is not required to dilate the pupil.
Intraoperative complications. Incorporating OMIDRIA into routine cataract surgery also decreases the risk for complications including intraoperative floppy iris syndrome, CME, and breakthrough iritis. My colleague and I conducted a retrospective two-cohort study to assess the effectiveness of OMIDRIA to reduce the incidence of postoperative CME, breakthrough iritis, pain, and photophobia compared to postoperative topical steroids (Figure).9
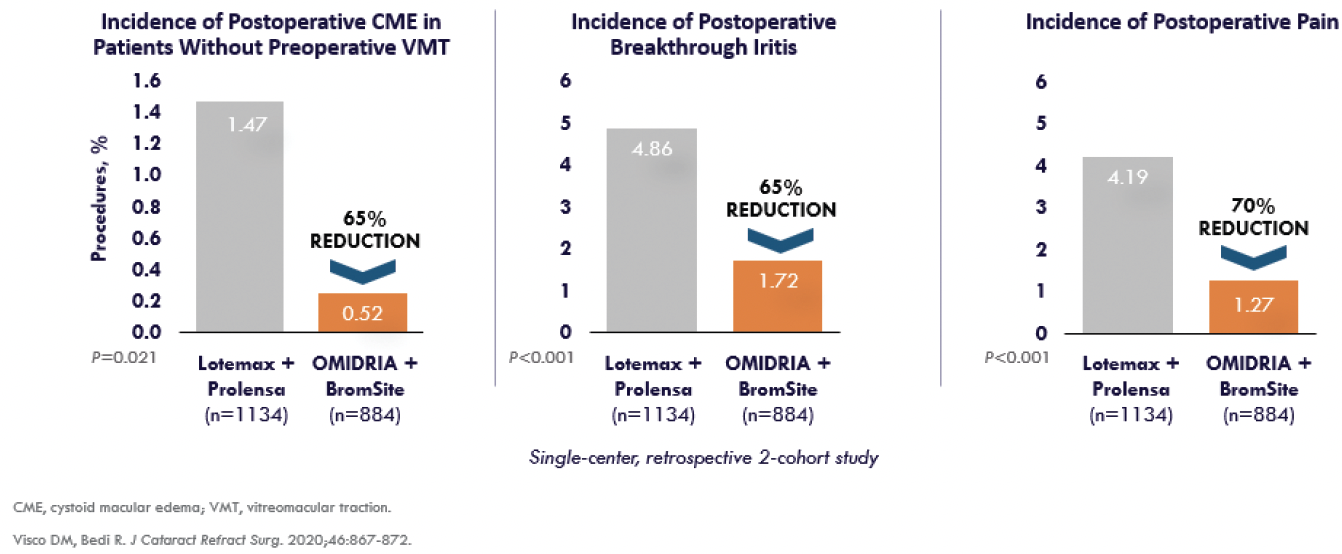
Figure. OMIDRIA reduces postoperative CME, breakthrough iritis, and postoperative pain.
A total of 2,218 eyes were enrolled in the study. Eyes either received OMIDRIA (n = 1,334) or topical loteprednol 0.5% 2 days preoperatively that was tapered postoperatively (n = 884). All eyes received topical NSAIDs pre- and postoperatively. Eyes in the OMIDRIA group had a significantly lower incidence of clinical CME (0.52% vs 1.47%, respectively; P = .021), breakthrough iritis (1.72% vs 4.86%, respectively; P < .001), and pain (1.27% vs 4.19%, respectively; P < .001). Additionally, patients in the OMIDRIA group had a lower incidence of photophobia, but this difference was not statistically significant (0.90% vs 1.13%, respectively; P = .590).
Patient outcomes. In my experience, the use of OMIDRIA provides more positive patient outcomes by reducing the inflammatory response to cataract surgery. Patients’ postoperative day 1 vision is better, and their risk for postoperative pain is much lower than if postoperative steroids and NSAIDs were used alone.10
Most of us don’t like to think that we cause patients pain. But, in fact, they can experience quite a bit of discomfort after cataract surgery. With OMIDRIA, not only is the anterior chamber quieter, but my patients are much more comfortable after surgery.
Patient experience. The use of OMIDRIA enhances the patient experience in several ways. Most importantly, it reduces or eliminates the need for topical steroids in the postoperative period. This may be because OMIDRIA provides continuous delivery of a therapeutic agent and inhibits the COX-1 and COX-2 receptors for up to 10 hours postoperatively.3 When I first started practice, patients were prescribed three drops in tapered dose regimens over the course of 4 postoperative weeks. This regimen was confusing, expensive, and a hassle for patients. In continuity with the ketorolac component of OMIDRIA, our patients simply use bromfenac twice a day beginning 2 days preoperatively and for 1 month after cataract surgery.
No longer prescribing a steroid postoperatively means that patients have fewer drops to buy and fewer drops to take. It’s easy for them, and it is safe. At my practice, the incidence of CME with a topical regimen of steroids and NSAIDs was approximately 1.5%. With OMIDRIA and an NSAID (no topical steroid), it dropped to 0.5%.9
Reimbursement. One thing I appreciate is Rayner’s helpfulness in navigating reimbursement for OMIDRIA. Ultimately, patients receive so much benefit from OMIDRIA that we want to provide it to all of them. Learning the best practices for billing and coding is important for our goal of providing all patients the opportunity to benefit from OMIDRIA.
CONCLUSION
The use of OMIDRIA during cataract surgery has simplified the procedure for me and for my patients. Patients are appreciative of the tangible benefits that OMIDRIA offers to them, including better postoperative day 1 vision and less pain and discomfort. As a surgeon, I appreciate the better visibility from a well-maintained dilated pupil, the lowered risk for postoperative complications, and the reduced need for pupil expansion devices.
I believe that OMIDRIA is the standard of care for cataract surgery. Surgeons who have this product available for patients should use it because the benefits can further help to ensure cataract surgery success.
1. Mohammadpour M, Jafarinasab MR, Javadi MA. Outcomes of acute postoperative inflammation after cataract surgery. Eur J Ophthalmol. 2007;17:20-28.
2. Tzelikis PF, Morato CS, Neves NT, Hida WT, Alves MR. Intraindividual comparison of nepafenac 0.3% for the prevention of macular edema after phacoemulsification. J Cataract Refract Surg. 2018;44:440-446.
3. Waterbury LD. Alternative drug delivery for patients undergoing cataract surgery as demonstrated in a canine model. J Ocular Pharmacol Ther. 2018;34(1-2):154-160.
4. Lindstrom RL, Loden JC, Walters TR, et al. Intracameral phenylephrine and ketorolac injection (OMS302) for maintenance of intraoperative pupil diameter and reduction of postoperative pain in intraocular lens replacement with phacoemulsification. Clin Ophthalmol. 2014;8:1735-1744.
5. Silverstein SM, Rana VK, Stephens R, et al. Effect of phenylephrine 1.0%–ketorolac 0.3% injection on tamsulosin-associated intraoperative floppy-iris syndrome. J Cataract Refract Surg. 2018;44(9):1103-1108.
6. Matossian C. Clinical and economic outcomes in cataract surgery using phenylephrine 1.0%-ketorolac 0.3% in a real-world setting. Paper presented at: ASCRS/ASOA Annual Meeting 2018. April 13-17, 2018; Washington, DC.
7. Chang DF, Campbell JR, Colin J, et al. Prospective masked comparison of intraoperative floppy iris syndrome severity with tamsulosin versus alfuzosin. Ophthalmology. 2014;121(4):829-834.
8. Al-Hashimi S, Donaldson K, Davidson R, et al. Medical and surgical management of the small pupil during cataract surgery. J Cataract Refract Surg. 2018;44(8):1032-1041.
9. Visco DM, Bedi R. Effect of intracameral phenylephrine 1.0%–ketorolac 0.3% on postoperative cystoid macular edema, iritis, pain, and photophobia after cataract surgery. J Cataract Refract Surg. 2020;46:867-872.
10. Rosenberg ED, Nattis AS, Alevi D, et al. Visual outcomes, efficacy, and surgical complications associated with intracameral phenylephrine 1.0%/ketorolac 0.3% administered during cataract surgery. Clin Ophthalmol. 2018;12:21-28.



