Key Takeaways
- Ophthalmologists discuss their approaches to planning cataract surgery on a patient with keratoconus who wants to minimize his dependence on scleral contact lenses.
- A possible approach involves regularizing the corneal surface.
- IOL options discussed include postoperatively adjustable designs, pinhole optics, aspheric and toric monofocal lenses.
CASE PRESENTATION
A 64-year-old White man presents with a history of keratoconus. The patient wears scleral contact lenses and would like to minimize his dependence on them. On presentation, his BCVA in those lenses is 20/40 OD and 20/30 OS. His manifest refraction is -6.25 +7.00 x 160º = 20/60 OD and -4.00 +4.25 x 043º = 20/50 OS.
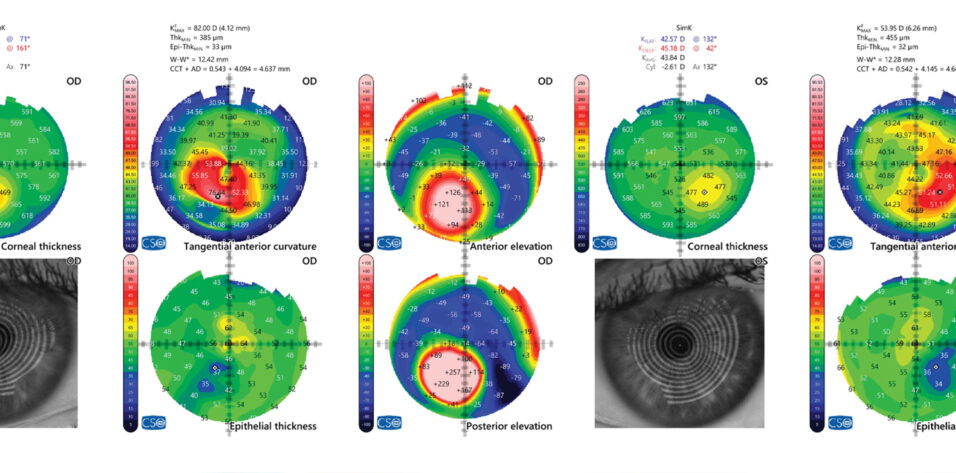
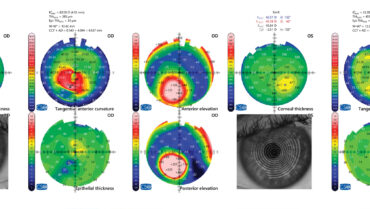
A slit-lamp examination finds keratoconus and Fleischer rings in both eyes and some apical scarring in the right eye. Both eyes have 2+ nuclear sclerotic and 1+ posterior subcapsular cataracts as well as 2+ anterior cortical opacities. OCT of the retina is normal in each eye (Figure 1). Biometry is shown in Figure 2.

Figure 1. Measurements with the MS-39 (CSO Italia).

Figure 2. Biometry (IOLMaster, Carl Zeiss Meditec).
How would you proceed?
— Case presented by Audrey Rostov, MD

DARSHAK PATEL, FRCOPHTH, MSC
I would counsel the patient to expect the postoperative UCVA in his left eye to be functional, but I would stress that the primary aim of surgery would be to improve his BCVA. His level of motivation would be essential to a successful outcome given the time, multiple procedures, and number of visits required.
Reducing the left eye’s need for a scleral contact lens would require regularizing the cornea. Total corneal (front and back surfaces) wavefront-guided transepithelial PRK (transPRK) combined with CXL would avoid inducing cataract-related aberrations in whole eye wavefront analysis. Three to 6 months following corneal treatment, it should be possible to estimate what the corrected distance visual acuity in the patient’s left eye would be after phacoemulsification, while accounting for cataract density, corneal shape, and pinhole acuity. If the eye’s visual potential is good (≤ 3.00 D of corneal cylinder and a low magnitude of higher-order aberrations), a Light Adjustable Lens (LAL; RxSight) would be implanted. Either the Barrett True K (keratoconus setting with measured posterior corneal astigmatism) or Kane-KC formula would be used. After LAL implantation, a hyperopic light adjustment to the lens is less dependent on pupillary dilation, so low hyperopia would be the preferred outcome. Specifically, the refractive target would be -0.50 D in anticipation of an approximately +0.50 D hyperopic shift based on keratoconus severity.
The keratoconus in the right eye is advanced with apical scarring. Neither the placement of corneal allogenic ring segments nor transPRK combined with CXL would be likely to correct the cylinder and aberrations sufficiently to allow the implantation of a toric IOL. The corneal thickness, moreover, would make transPRK combined with CXL problematic. An IC-8 Apthera lens (Bausch + Lomb) would therefore be implanted to improve the patient’s BCVA. Preoperatively, a pinhole test and pilocarpine treatment could allow him to determine the acceptability of this surgical strategy, especially if the apical scar coincides with the visual axis. He would be counselled to expect the BCVA and quality of vision in his right eye to improve after surgery with the trade-off of dimmer vision. The treatment target would be -1.50 D to account for a subsequent hyperopic shift, and the ideal final refraction would be -0.75 D.

FARHAD HAFEZI, MD, PHD, FARVO
Biometry shows an enlarged pupil, consistent with measurement acquisition under mydriasis. The patient’s natural (photopic and mesopic) pupil size is clinically relevant given the multiple optical challenges, including bilateral keratoconus.
Reducing corneal asymmetry and the magnitude of higher-order aberrations would be essential to improving the reliability of IOL power calculations and selection as well as to optimizing his visual acuity after cataract surgery.
First, the corneal surface would be regularized using ELZA-PACE, a second-generation customized epithelium-on CXL technique (currently unpublished) developed at the ELZA Institute, where I practice. To date, more than 300 patients have undergone this treatment. The procedure typically induces localized flattening (approximately 4.00–8.00 D) of the cone. In some instances, irregular astigmatism has been further reduced through a favorable coupling effect. My colleagues and I have found that CXL using the ELZA-PACE technique alone generally improves patients’ BCVA by 2 to 3 lines and substantially reduces vertical coma.
Once the patient’s topography stabilizes (3–6 months after treatment), total corneal wavefront–guided transPRK could be considered to further regularize the cornea by addressing anterior and posterior aberrations. Given his age, it appears that up to 50 µm of stroma could be removed.
After the cornea stabilizes, cataract surgery with an aspheric monofocal or toric monofocal IOL would be performed. IOL calculations would use a standard or keratoconus-specific formula such as the Barrett depending on residual corneal irregularity.

WHAT I DID: AUDREY ROSTOV, MD
The patient and I discussed his goals and expectations for cataract surgery. He was eager to reduce his dependence on contact lenses but understood that he might need to wear them to achieve his optimal BCVA. His corneas had been stable for more than 10 years, and we discussed the options of CXL and topography-guided PRK. We also discussed potential deep anterior lamellar keratoplasty in his right eye, but the patient chose to proceed with cataract surgery alone because he was used to his “imperfect” vision.
The left eye was treated first. IOL calculations were performed with the Barrett True K keratoconus formula. Laser cataract surgery was performed with the Lensar Laser System (Lensar), which enabled customization of the capsulotomy with alignment nubs for a toric IOL. An enVista Toric IOL (Bausch + Lomb) was selected for its aberration-neutral profile. Surgery was uneventful, and the patient’s UCVA on postoperative day 1 was 20/20-1 OS.
The same surgical approach was used for the right eye. The refractive target was -1.50 D. Because the right eye had more astigmatism than the left, I considered implanting an enVista toric IOL in the bag plus an LAL in either the bag or the sulcus to allow postoperative fine-tuning of the sphere and cylinder. I frequently implant the LAL but have found it to be less effective in keratoconic eyes owing to the irregular astigmatism.
At the American-European Congress of Ophthalmic Surgery’s 2025 Summer Symposium, Robert Weinstock, MD, described the layering of toric IOLs in eyes with high astigmatism. I refer to the technique as a Weinstack. In this situation, I used his method of splitting the spherical power between the lens implants and placing the IOL with the higher cylinder power in the bag first, followed by the second IOL in the bag on top of the first one. Some manipulation with Kuglen and Sinskey hooks was required to position the toric IOLs in the same plane. Fortunately, the customized capsulotomy helped with centration. The OVD was carefully irrigated out from between the IOLs. Dr. Weinstock’s off-label approach is innovative and worked well for this patient.
At his most recent postoperative visit 3 months after surgery, his UCVA was 20/50 OD and 20/20- OS. The refraction in the right eye has varied, but he was highly satisfied and much better able to perform the activities of daily living. He plans to be fitted with a scleral lens for occasions when he wants to optimize the BCVA in his right eye. An advantage of scleral contact lenses is that they can be worn after toric IOL implantation.