CASE PRESENTATION
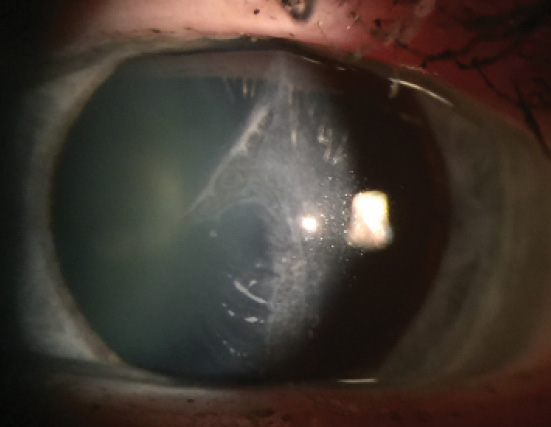
Figure 1. Slit-lamp view of a traumatic LASIK flap injury in the
patient’s left eye.
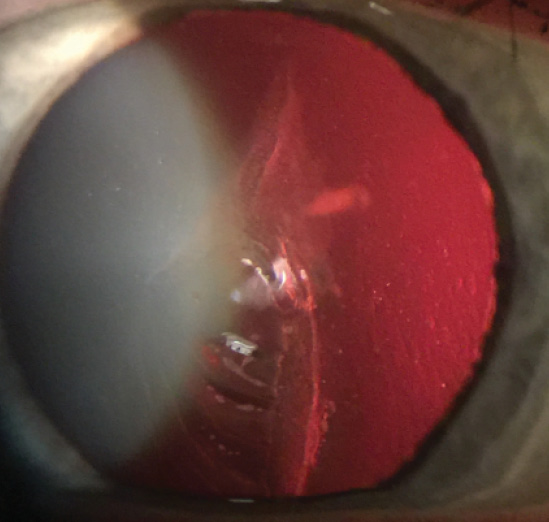
Figure 2. Red reflex slit-lamp photograph of the torn,
dislocated LASIK flap.
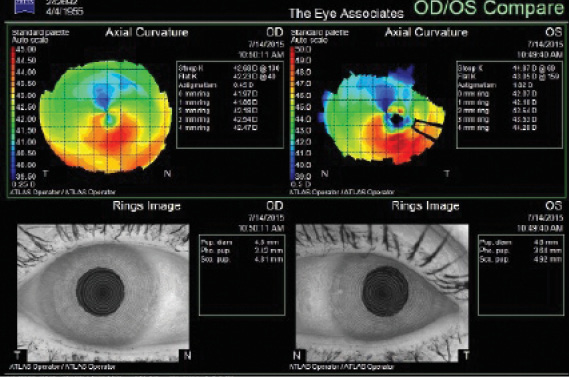
Figure 3. Topography of the patient’s injured left eye.

PREEYA K. GUPTA, MD
In these situations, it is important to understand the patient’s visual goals. In this case, the patient has 20/20-1 BCVA in the traumatized eye, but presumably, the quality of vision is poor due to glare and distortion. The simplest first option would be to conduct a hard contact lens trial to see if the visual aberrations improved. Given the central scar, I would not expect the contact lens to completely resolve the patient’s problem.
Epithelial ingrowth is the next major issue. When ingrowth is limited, it is acceptable to watch the cells to see if they advance. Considering the trauma to the flap and multiple entry points for epithelial cells to grow into the interface, however, I would favor intervention, especially if there are signs of a flap melt. My preference would be to amputate the flap, remove the epithelial ingrowth, and apply mitomycin C (MMC). The centrally triangulated tear will make it very difficult to lift the flap safely for epithelial debridement alone. Often, even gentle manipulation can further tear a traumatized flap, and gluing or suturing it is not feasible because of the tissue’s fragility.
The cornea can take months to stabilize. At that point, if the patient still desired better UCVA, one could consider a PRK enhancement for residual refractive error but with informed consent about the possibility of worsening scar/haze formation and an unpredictable refractive outcome. The key would be to wait for refractive stability before considering a refractive enhancement.

MITCHELL A. JACKSON, MD
Traumatic LASIK flap injuries are extremely difficult for the refractive surgeon to treat, especially if he or she is the original LASIK surgeon. Despite the trauma, patients typically retain the same high expectations they had prior to LASIK. From a medical-legal perspective, then, it is imperative to ensure that this patient has reasonable expectations through a true underpromise-overdeliver approach.
She has fairly good quantitative distance BCVA in the traumatized eye. Based on the slit-lamp appearance of the cornea, however, her qualitative vision suffers from irregular astigmatism and severe corneal aberrations. I would take a stepwise approach to repairing the cornea. I would strive to keep it simple by initially trying to lift the flap and clean up the epithelial ingrowth as much as possible. The judicious use of topical steroids and topical cyclosporine will probably be necessary to minimize permanent corneal scarring. After 3 to 6 months of refractive and corneal topographic stability, topography-guided PRK with MMC will likely be needed to treat any residual refractive error.
If all else fails, it may be necessary to amputate the flap. Again, after 3 to 6 months of refractive and corneal topographic stability, topography-guided PRK with MMC may be needed for any correctable refractive effect. The good news is that the patient was myopic in this eye at the initial referral visit, which would allow some room for PRK treatment in the future if needed.

WILLIAM F. WILEY, MD
It appears that the tear has allowed epithelial ingrowth underneath the flap, resulting in a media opacity and, likely, irregular astigmatism. I would counsel the patient that the trauma has triggered a healing response that is currently interfering with her vision and that will likely progress, causing further vision loss. The options include surgical treatment or observation. The latter does not come without risks. Nevertheless, surgical correction does not guarantee success, and multiple procedures may be required to achieve an acceptable visual acuity.
For a surgical correction, I would remove the LASIK flap along with the irregular epithelium. This strategy would probably solve the majority of shape irregularity and media opacity, because the scar tissue/media opacity appears to be limited to within the flap and or epithelial layer. The traditional next approach would entail allowing the eye to heal after flap removal and then treating the residual refractive error with a separate secondary procedure months later.
I have had success with flap removal and excimer laser ablation in one surgical procedure through the use of intraoperative aberrometry. To potentially treat the patient’s refractive error and solve the media opacity, I would follow these steps:
1. Amputate the flap and remove the irregular epithelium under an OR microscope.
2. Perform intraoperative aberrometry to assess the new refractive state of the eye. (Phenylephrine might be required for pupillary dilation, and I might use a plano bandage contact lens to allow for a uniform anterior corneal surface.)
3. Transfer the patient to the excimer laser. Program the desired correction/outcome based on the intraoperative aberrometry reading. Apply MMC to prevent the formation of corneal haze.
4. Conduct follow-up and postoperative care as I would after standard surface ablation. n
Section Editor Alan N. Carlson, MD
• professor of ophthalmology and chief, corneal and refractive surgery,
Duke University Eye Center, Durham, North Carolina
Section Editor Stephen Coleman, MD
• director of Coleman Vision, Albuquerque, New Mexico
Section Editor Cathleen M. McCabe, MD
• cataract and refractive specialist, The Eye Associates in Bradenton
and Sarasota, Florida
• (941) 792-2020; cmccabe13@hotmail.com
Section Editor Karl G. Stonecipher, MD
• clinical associate professor of ophthalmology, University of North
Carolina, Chapel Hill
• director of refractive surgery, TLC in Greensboro, North Carolina
Preeya K. Gupta, MD
• assistant professor of ophthalmology, Duke Eye Center, Durham,
North Carolina
• (919) 660-5071;
Mitchell A. Jackson, MD
• founder and CEO, Jacksoneye, Lake Villa, Illinois
• mjlaserdoc@msn.com; Twitter @djmjspin
William F. Wiley, MD
• private practice at the Cleveland Eye Clinic, Cleveland, Ohio
• assistant clinical professor of ophthalmology, University Hospitals/
Case Western University, Cleveland, Ohio
• (440) 526-1974;


