In this roundtable discussion held during the American Academy of Ophthalmology (AAO) 2017 in New Orleans, ophthalmologists working in glaucoma, retina, and cataract and refractive surgery talked with Inder Paul Singh, MD, about prospective clinical study data, hands-on experiences, and how their profession’s view of vitreolysis is evolving alongside growing data and the introduction of the new Reflex Technology platform.
Inder Paul Singh, MD: A great number of doctors are now performing laser vitreolysis, and it’s starting to grow in popularity, but there are still some misperceptions and concerns about this procedure.
I’ve been performing Laser Floater Removal (LFR) for about 4 years now—more than 3,000 cases—and I can tell you, compared to just about everything I do in my practice, this procedure has probably made the biggest impact on my practice in terms of patient satisfaction, staff buy-in, and outcomes. What still surprises me today is the impact floaters have on patients’ daily life and quality of vision. Until I started performing this procedure, I didn’t know how much of an impact vitreous floaters have on patients’ daily functioning and quality of life.
EARLY EXPERIENCES
I want to start by asking why you began using the procedure, and what you have learned in the course of your first few cases. What impact has it made on your patients’ daily life?
Chirag P. Shah, MD, MPH: I treated patients with YAG vitreolysis through the clinical trial that we conducted a few years ago,1 and for some patients it can be really profound. One reason is that for the first time in many of these patients’ lives, someone is actually listening to them and acknowledging that they do have floaters that are visually significant and affect their quality of life. Most of the time, doctors tend to dismiss symptoms of floaters and just move on to something else. It was quite powerful to see how much respect patients gave me just because I acknowledged their symptoms. After undergoing vitreolysis, some patients were thrilled. It was like the old days when I used to do cataract surgery and see that really powerful “wow factor” after surgery.
Dr. Singh: Absolutely. Dr. Tibbetts, tell us how you got involved and what your experiences have been so far.
Michael D. Tibbetts, MD: I started doing laser vitreolysis after learning the results of Dr. Shah’s study.1 I was initially very skeptical. I’d heard about the procedure being commercialized in many markets, and that it was the primary focus of some practices. I remained very skeptical until Dr. Shah’s early data convinced me that it was a worthwhile procedure in selected patients.
For years, I had been dismissive with many patients who had floaters, just as Dr. Shah described. Once I started listening to their symptoms, I realized that floaters were impacting their quality of life, so I decided to try vitreolysis. I initially treated a few patients and was quite impressed by their response to treatment. My first patient was pseudophakic with an amblyopic eye. She was impressed with the symptomatic improvement in her first eye, so I treated her other eye, and then I continued to gain experience with the procedure over the last few years.
Dr. Singh: It’s amazing. After treating only a few patients, the impact these floaters have on our patients’ daily functioning is quite apparent. These first few cases help solidify the need for a treatment option for these patients (Figure 1). Dr. Stonecipher, tell us about your experience and how you got involved.
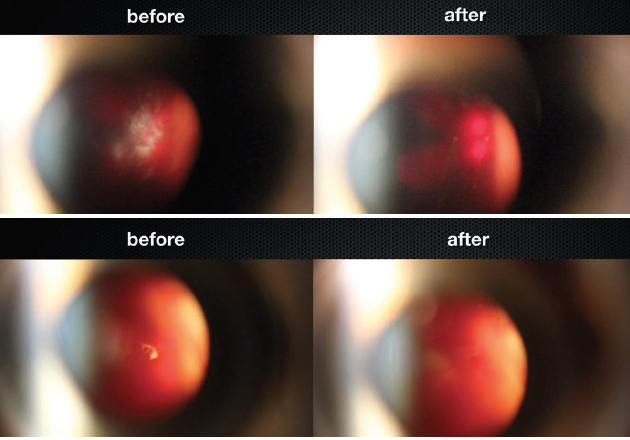
Figure 1. Patients before and after laser vitreolysis.
Courtesy of Inder Paul Singh, MD
Karl Stonecipher, MD: Dr. Singh and I have been friends for quite some time, and I started doing this about a year after he did. I’ve probably done a little more than half the number of cases that he has done. When we first discussed it, I didn’t have the right laser technology. I tried it with an older YAG model, but I wasn’t satisfied with the procedure. Once I got the Ultra Q Reflex (Ellex), I felt comfortable treating patients with laser vitreolysis.
I’d always felt that I didn’t want to say, “These people with floaters don’t have a problem,” because I’d heard that with LASIK. People would be minus 7 or 8, and doctors would tell them, “There’s nothing wrong with you.” It was the same situation with floaters. These people came in with complaints, and when I started looking at some of the ancillary data, we could diagnose the specific problem and understand what patients were complaining about.
I agree that it’s best to start out with the easy cases, like pseudophakic patients. I was able to stand on the shoulders of giants, including John Karickhoff, MD, and, for me, Dr. Singh. He gave me many pearls. For example, making sure the retina is not in focus and starting with pseudophakic patients with large Weiss rings. I think it certainly helps to have somebody help you get there.
Dr. Singh: Very good point. I think those doctors who are interested should try to reach out to those who have been performing laser vitreolysis for a while. There is a learning curve, and those pearls can make a big difference. Dr. Mac, you were telling me about some of your early treatments and the impact they had on patients’ quality of life. Tell us a about your first patient and how you began.
Ivan Mac, MD, MBA: I do a fair bit of premium lens cataract surgery. We run into those patients every once in a while where we’ve treated them and everything looks perfect—the retina looks great, the ocular surface has been optimized, we’ve used the YAG laser to clear a posterior capsule opacification—yet the patients are still a little dissatisfied with the results. They’re happy, but they’re not ecstatic, and I want ecstatic patients.
My very first LFR patient was a gentleman that had multifocal IOLs. He was happy with his surgery but not ecstatic. I noticed that there was a little amorphous cloud in the vitreous. We went ahead and evaluated the Ultra Q Reflex, and I talked with both Dr. Singh and Dr. Stonecipher. I was very impressed with how easy the technology was to adopt and how gentle it was on the patient, so we decided to get it.
I treated that very first patient for that amorphous cloud. We did two treatments, making kind of a grid pattern in the posterior vitreous, staying away from the retina and making sure we could visualize everything. The patient came back 1 week after his second treatment and said, “Doc, I don’t know what you did, but it’s made a world of difference.” He offered to provide a testimonial for us, and he did a review for us on Facebook. He was my very first patient, and he was evangelical about the procedure. I was hooked.
Dr. Singh: Multifocal lens patients with amorphous clouds in the middle of the vitreous are some of the most symptomatic patients. Due to the multifocality and the aberrations caused by the floaters, patients are incredibly happy after floater treatment. In fact, our group presented a paper at ASCRS 2016 demonstrating the improvement in higher-order aberration (HOA) after vitreolysis using the iTrace Ray Tracing Aberrometer/Topographer (Tracey Technologies).2 The improvement was seen in both monofocal and multifocal lens patients.
Dr. Stonecipher: I’ll take that one step further. We’ve all had patients who look left and right as we’re doing their cataract surgery, and we put their lens in and see a big floater at the back that we didn’t see before, because the cataract was significant. I start talking to those patients on the table. I say, “You’re going to see better and be happy after surgery, but I see this floater, and if it bothers you, we’ve got something that we can do to help.”
Dr. Mac: We also have to make sure to tell patients preoperatively that they have a floater that may become more bothersome to them after cataract surgery. If we tell them after surgery, then, in their minds, the cataract surgery has caused the floater.
Dr. Singh: Yes, in the preoperative assessment, it is important to inform the patients that they have a vitreous floater. Even if they do not have symptoms before cataract surgery, we find symptoms can start or worsen after cataract surgery. We often tell patients, “By the way, you might notice floaters after cataract surgery. If that does occur, we have something we can use in the office to help you.” That way, they do not get surprised or blame the surgeon for causing the floater.
Dr. Stonecipher: I tell my patients that they wouldn’t really notice the floaters now, but once we remove their cataract and put on a lens, they will see it clearly. From experience, I’ve often seen patients start to notice their floaters again. Some people will learn to adapt, but some people won’t.
Dr. Mac: We need to do something for these patients, and having this technology has made me very excited that we can really help them.
Dr. Singh: What I’m hearing from everybody is that quality of life for our patients is becoming more and more important. We never really thought about the need to treat floaters, but a study in 2011 showed that patients with floaters had a higher risk tolerance than people who had symptomatic HIV, diabetes, and hypertension.3 There’s a need out there, and now we have something that can fill that need.

A RETINAL SPECIALIST’S PERSPECTIVE
Dr. Singh: Historically, I think a lot of us didn’t have much information on laser vitreolysis. It wasn’t taught in schools, and there was some pushback. What do you think are the procedure’s limitations? Why are some doctors still not quite as friendly as we are to this procedure today?
Dr. Stonecipher: A lot of retina colleagues are saying LFR is not a good thing. It’s an “if it isn’t broken, don’t fix it” kind of mentality. One great retina surgeon in my area sends me patients with symptomatic floaters, but he still says, “I think this is a risky procedure.” I really appreciate Dr. Shah putting his study together,1 because now I can look at these friends of mine and say, “Here is a study that actually proves its safety and benefits.” And partly as a result of that study, some retinal surgeons are actually starting to do the procedure themselves.
Dr. Mac: I just got stopped in the exhibit hall by two of my retinal colleagues, who asked, “Does this laser floater thing really work?” I said, “Yes. You guys need to start doing this. And if you can’t get the results to where you need it to be, then you can always still do a vitrectomy.” They both said, “Oh no, no! I don’t want to do it. I’m going to send the patients over to you.”
Dr. Singh: I think one of the limiting factors that’s leading to this disconnect was that not enough data were available about YAG vitreolysis, and the data that did exist were using laser technology that was not optimized for the procedure. We’ll talk more about what’s new with the technology we use now, but it is important to note that the illumination system used in previous studies was very different than what we are using now. The illumination systems in standard YAG lasers do not allow the user to visualize far enough behind the posterior capsule to identify many of the symptomatic floaters, let alone provide the necessary spatial context to understand how far the floater is from the retina and/or lens. Also, the energy settings used in those previous studies were much lower, and surgeons used fewer shots, so the outcomes were not quite the same as we see now. Dr. Shah, you really changed all that when you performed the first placebo-controlled prospective clinical trial of YAG vitreolysis with this new technology.1 Tell us about why you did your study and what kind of outcomes you saw in your practice.
Dr. Shah: Just to speak to Dr. Stonecipher’s point, as the person who represents the retinal surgeons on this panel, I think there’s a lot of skepticism in the field, because there were no data for vitreolysis. I see that skepticism breaking down now that we have data. We have a few studies showing that there is moderate efficacy, with a good safety profile.1,4 Our study has, fortunately, inspired other similar studies that are using our protocol right now around the world. As we get more and more data over the next few years and can better understand the best types of floaters to treat, the true rates of adverse effects, and which patients have the best satisfaction, I think that we will find YAG vitreolysis might fill the niche between observation and vitrectomy surgery.
Several years ago, one of my optometry friends asked me to give a lecture at MIT about floaters. I was not excited. I wanted to talk about a “real retinal disease” like macular degeneration or retinal detachment, so I reluctantly agreed to do this lecture. In the process of putting together my slide deck, I realized that there were limited good quality data in the literature. There was a glaring unmet need for a proper clinical trial. I’m at an institution where we do many clinical trials, and we have the machinery to do it pretty quickly and easily. With the help of my colleagues, we designed a trial to look at YAG vitreolysis.
We selected patients with Weiss ring floaters, the easiest floater to treat based on some of the experiences that you’ve had. We compared patients treated with a single session of YAG vitreolysis versus sham laser. We did a small pilot study, not a large, multicenter trial with years of follow-up. It was a small single-center trial with 52 patients, whom we followed over a 6-month period after treatment with YAG vitreolysis (Figure 2).1
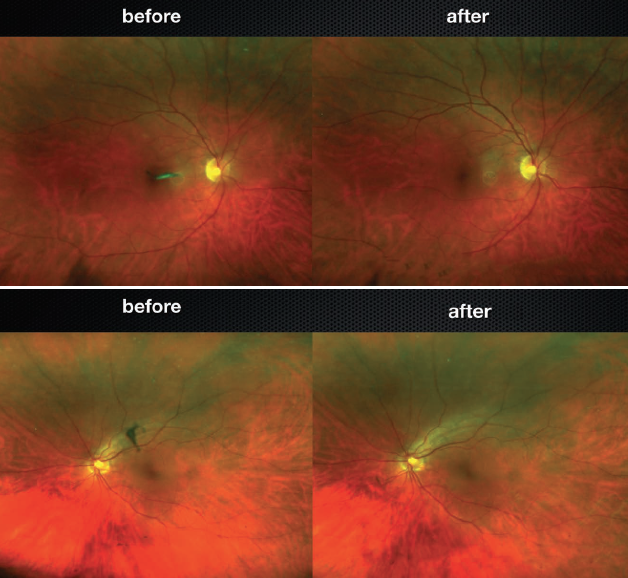
Figure 2. Patients before and after laser vitreolysis.
Courtesy of Chirag P. Shah, MD, MPH
Our study was designed for safety. We specifically measured the distance of the Weiss ring from the retina and from the posterior capsule in phakic patients, selecting patients with buffers of 3 mm or greater for the retina and of 5 mm or greater from the posterior capsule. The first patients I treated were all pseudophakic. I’d never done the procedure before, so I thought starting with easier cases would allow me to learn. After I talked to you, Dr. Singh, I realized that my powers may have been a little bit too low, and I increased the power to get true vaporization rather than fractionation.
We found that patients experienced moderate improvement in symptoms—54% mean subjective change over a broad range of 0% to 100% improvement. An objective masked grader looked at before and after ocular photographs, and saw 94% improvement. That is a pretty big disconnect between the objective interpretation of the outcome after YAG vitreolysis and the patient’s subjective experience. We tried to determine why some patients have a robust response while others do not, despite both having a good objective response. I think that’s an important area for future research.
One other aspect of the study I should note is that patients only had one YAG treatment session, because if I treated control patients with two sham lasers, they would realize that they were in the sham group and be unmasked. I think that the success rate in our study might have been even more robust if I could have: (1) offered more than one session; (2) selected patients with just classic Weiss rings; (3) and selected patients who were, perhaps, a bit easier to please.
Dr. Singh: That’s great information. Data are so important, and I do think your study has helped us truly evaluate this newer YAG laser technology.1 I think this will help ease the minds of those who are still a little skeptical. Keeping in mind that safety has always been a main concern for our colleagues, can you tell us more about adverse events you saw in your 6-month study?
Dr. Shah: I hit and pitted an IOL lens peripherally, but it was not significant. That was the only adverse event in the treatment arm. There were no retinal detachments, retinal tears, retinal damage, or elevation of IOP. We measured IOP 30 minutes after the procedure, as well as at 1 week, 3 months, and 6 months, and saw no elevation.
PATIENT SELECTION
Dr. Singh: Patient selection is a vitally important consideration. If a surgeon who is new to this procedure said to you, “I want to start doing vitreolysis,” what characteristics would you tell them to look for in his or her first patients?
Dr. Mac: The slam-dunks are pseudophakic patients who have Weiss rings. In one session, you’re going to make a huge improvement in their quality of life.
Dr. Stonecipher: My perfect patient is just as you described: pseudophakic with a nice little vitreous floater that’s easy to see and to bring into focus (Figure 1). Those patients tend to be satisfied and do not keep coming back with problems. I always tell patients before the procedure that there will be more than one session, which I think makes them more understanding afterward if they have some floaters left over. Now, more challenging patients present as well (Figure 3). With these patients, I will let them know that it may take more than one session to resolve their issues.
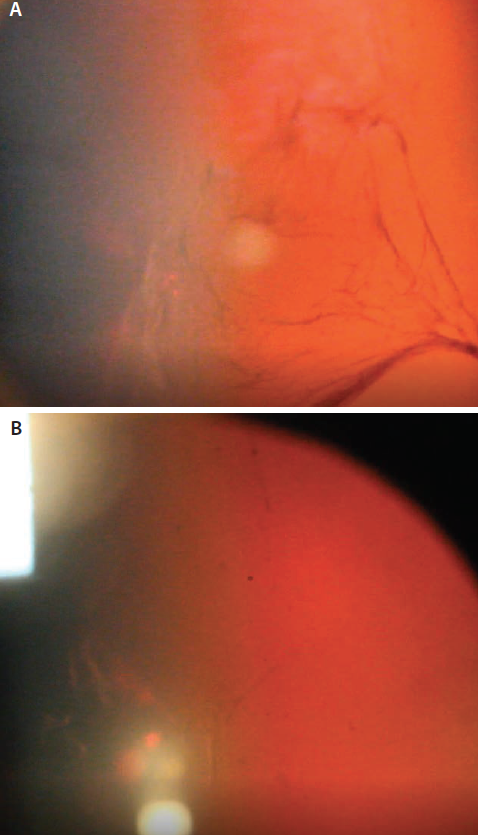
Figure 3. Notice the membrane in this patient prior to treatment, which is fairly extensive (A). Notice the reduced size of the membrane after the procedure (B). Some of these larger floaters may require an additional intervention.
Courtesy of Karl Stonecipher, MD
Dr. Singh: We have the same experience. We presented a study of our first 362 patients last year,5 and we found that Weiss ring patients averaged only 1.3 sessions. Most of those patients are satisfied with one session, and with fewer shots needed than the number of shots required for treatment of a larger, amorphous cloud. Those with amorphous clouds needed an average of three sessions to achieve 90% satisfaction. The reason is that vaporization from the plasma spark is limited to just a few microns.
The energy delivery with Reflex Technology is very efficient. When you increase the energy on the laser, the increase in dispersion in the eye (the convergence zone) increases in a nonlinear fashion. For instance, at 1 mJ, the convergence zone is 110 μm, and when increased to 10 mJ, the convergence zone increases to 210 μm (a zone increase of less than 50% with a 10X increase in laser power). This is what ensures safety using higher energy levels. For larger amorphous clouds, however, we may not be able to vaporize or sever enough of the opacity in just one or two sessions. This is why patient education on the front end is so important.
Another advantage of treating Weiss rings is that they have a good correlation with symptoms. There is a solitary opacity in the vitreous, we have a clear way of understanding that this is the patient’s problem, and we can see the ring break up and vaporize when we’re performing the procedure. This is why we always recommend Weiss patients as good candidates for starter cases. I also recommend not performing YAG vitreolysis on patients with new symptoms. I like to wait at least 4 to 6 months to see if they will neuroadapt, and I also recheck the retina for defects. I have seen three patients with retinal tears at their second follow-up visit after initially seeing them for a new posterior vitreous detachment. I was thankful I did not treat those patients when I first saw them, because they may have thought the laser caused the tear.
Dr. Stonecipher: If the posterior hyaloid has separated and a vitreous floater is present, does it have an attachment to the vitreous base anymore? Or, if it does not and you’re lasering a free floater, does that make a difference?
Dr. Singh: I think you’re severing vitreous strands and opacities when you vaporize, not creating true traction and pulling like you do with a vitrector. Is that fair to say?
Dr. Shah: That’s an intriguing question. I think you’re correct, and this is where more data with longer follow-up would be helpful.
Dr. Singh: That’s a very good point: we do need more data on this subject. I do think some surgeons look to data sets on capsulotomy and retinal detachment rates that show a slight increase with a YAG capsulotomy, and then they relate that to vitreolysis. It is important to note the forces at work for a YAG capsulotomy and vitreolysis are different. In a YAG capsulotomy, you are breaking the posterior capsule. The posterior capsule is attached to zonules, which are attached to the vitreous base, theoretically resulting in a direct transfer of energy to the vitreous base. In vitreolysis, although there is a collagen matrix, we do not have that same connection to the vitreous base.
Now I want to understand how we get to a place where doctors understand this procedure is safe and effective overall. One of the historical limitations was visualizing the vitreous. If you look at standardized lasers that we’ve had for many years, we couldn’t see very far behind the posterior capsule because the illumination systems were not really designed for that use. I think a lot of our colleagues think, “Well, I use the YAG laser for laser peripheral iridotomy and capsulotomy. How can I use it for anything behind that? It doesn’t have the efficacy.”
NEW TECHNOLOGY
Dr. Singh: What has changed in our YAG lasers? How has Reflex Technology—the Ultra Q Reflex and the Tango Reflex (Ellex)—with True Coaxial Illumination (TCI) changed our ability to visualize the vitreous and get the spatial context needed for laser vitreolysis?
Dr. Stonecipher: Visualization is key for this procedure. We see some patients whose floaters are not visible in the clinic, but when I get to my laser, I can see floaters very easily. We look up and look down, and as one floats by, we laser it. Those patients are not common, but they’re a challenge for me, and they seem to be a lot more symptomatic than patients whose floaters are fixed.
Dr. Singh: To me, the visualization system with this new Reflex Technology is really fascinating. Historically, we couldn’t see where we were in the vitreous. The crucial safety concern for me is the possibility of hitting a lens or the retina. TCI is the heart and soul of this laser. It is what makes this system different and enables us to perform YAG vitreolysis with confidence. We can fire the laser with the slit lamp in any position, titrating the illumination. With TCI, we now have much better depth perception, which also allows us to understand the floater’s location, whether that is right behind the lens or near the retina. This system provides spatial context that enables us to know where we are in the vitreous, crucial capabilities for a safe and efficacious treatment.
TCI also allows the light source, the aiming beam, and our oculars to be on the same optical pathway. In contrast, with standards lasers, the illumination comes from below on a different pathway. The standard laser and the illumination meet at the posterior capsule, which is what we want for laser peripheral iridotomies and capsulotomies. But when we’re trying to identify the floaters in the middle or posterior vitreous (where most of the symptomatic floaters reside), the illumination and the standard laser beams cross, and we cannot see the floater. Because TCI puts the laser, illumination, and ocular view on the same optical pathway, we see the floater and we can appreciate how far it is from the retina. I think this technology is the reason behind the positive data we’re seeing from Dr. Chirag’s study.1 We now have a way to visualize floaters in a way that makes this procedure safe and effective.
Dr. Mac: In my experience, the mid-vitreous lens to visualize floaters is more effective in this laser as well.
Dr. Singh: Yes. To do this procedure safely with good efficacy, we need laser technology that maximizes visualization of the vitreous, as well as lenses designed to view the entire vitreous. A YAG capsulotomy lens, like the Abraham lens, will not give you the focal length you need, but we do have special lenses available to us. What lenses are you using?
Dr. Mac: I think it’s important to emphasize that when physicians are starting out with this procedure, they may not be using lenses if they’re not glaucoma specialists who look at angles every day. Some might use a lens for YAG capsulotomy, but others, including me, do not. With this new laser, I treated a case of angle closure the other day with an iridectomy lens. I popped a hole in four or five shots, and I was done. I’ve been through tryouts with several different lasers, and I think the Ellex laser is the right technology for all these various procedures, including YAG vitreolysis. The energy doesn’t come back at us, so we don’t have to worry about that energy being projected toward the retina.
Dr. Singh: There are two companies that make vitreolysis lenses: Volk and Ocular Instruments. I use both, but have tended more toward the Volk mid-vitreous lenses.
As you mentioned, Dr. Mac, energy is a big concern for all of us. When I started performing YAG vitreolysis, I was worried about energy levels. We use 20 to 30 shots at 1-2 mJ to complete a capsulotomy. What’s different about YAG vitreolysis, or LFR, is that we use higher energy, oftentimes 5 or 6 mJ or even higher in some cases. As a result, I think it’s important to understand that our historic understanding of energy delivery, including what we’re now training in our residency programs, revolves around YAG capsulotomy. Sit down, focus on that capsule, fire when you see a clear shot, and you’re done.
That was our training for lasers. In this case, our understanding of energy delivery is even more important. Reflex Technology has a truncated energy beam for very efficient energy delivery, so less energy is needed to cause optical breakdown. As we discussed earlier, when we increase the laser’s energy setting, the relationship with the amount of dispersion, or the convergent zone, is not linear. It was an eye-opening moment for me to realize that if I go up to a higher energy level, I am not linearly increasing dispersion in the eye. This is why we feel comfortable going up to higher power levels for many of our patients.
Dr. Stonecipher: A lot of ophthalmologists don’t understand the laser to that level of depth. So when we say we’re changing the dynamics of these pieces of material in the vitreous, they think that we’re breaking them up into smaller parts. Instead, we’re seeing gas bubbles as the tissue is vaporized away. I don’t think that this concept has been emphasized enough. We’re actually getting rid of a floater.
Dr. Singh: That’s a great point. I’m glad you brought that up because when you are treating a Weiss ring, for example, you do see it break up, but you also are seeing it vaporize. You’re not left with a thousand pieces at the end. Let’s talk about how that works.
Dr. Tibbetts: You titrate up the power and start to see gas bubbles form. When you hit the opacity with the laser, it doesn’t just deflect back—you see it break up and vaporize. We know from experience that repeated pulses of the laser can effectively vaporize the opacity.
Initially, visualization was the hardest part of this procedure for me to learn. I tell my patients, “I’m going to do my best once I get you under the microscope, but if I can’t clearly see what I’m treating, we will not be able to do the treatment.”
Dr. Stonecipher: I’ve done a large number of these procedures, and I still get nervous in a phakic patient. I’m constantly looking at the retina, looking at the lens, looking at the retina, looking at the lens, trying to figure out where I am in the vitreous. I’d also note that it’s important to have somebody hold the patient’s head or strap the head in place, because this is a procedure where you do not want the patient moving around, creating small discrepancies that could result in you hitting a lens or retinal tissue.
Dr. Tibbetts: Another small point: double-check your settings. It’s important to not have an offset, which would obviously make you more prone to errors.
Dr. Singh: I want to break all of this down because I know a lot of our colleagues are thinking, “How do I decipher all of this great information?” You brought up how with vaporization, you see the floater break up into pieces, but you also see those gas bubbles. That is plasma occurring, right? You’re at 10,000° F in a small-micron area, with a 3- or 4-nanosecond pulse. Because of that short pulse duration, right after the plasma spark is formed, the heat dissipates before the next shot is fired. That’s why we don’t see that heat building up in the eye, and we don’t see the inflammation we would expect if it exerted a lot of energy into the eye. That’s also why we don’t need to stop at 20 shots.
People say to me, “Dr. Singh, why are you doing 150 or 200 shots for a Weiss ring? That’s a lot more than a YAG capsulotomy.” My explanation is just as you said, with the breakup and vaporization. I fire and break floaters into pieces, and then keep going until they are all vaporized and gone. This is one reason that previous attempts did not have the same efficacy we see today. People are justifiably afraid to fire many shots, so they stop at 40 to 50. If we stop with that few shots, we will leave behind multiple smaller pieces. It takes some time to feel comfortable firing 200 to 300 shots in one sitting, but in time, it begins to feel safe to keep going until the smaller pieces are vaporized.
Dr. Mac: I tell my patients I’m playing Asteroids, the old video game. They get it.
Dr. Tibbetts: Are you guys using a single pulse or a triple pulse?
Dr. Stonecipher: I use both. I prefer a single because I’ve had patients move. If a floater is big and I want to move things along, I’ll use a triple.
PATIENT EDUCATION AND POSTOPERATIVE REGIMEN
Dr. Singh: For surgeons who do not understand how YAG vitreolysis works, it can be a surprise to realize that it may take more than one session. Let’s talk about treating the same patient multiple times. When patients come back for their postoperative visit, how do you decide if a patient needs more treatment? What does that treatment plan look like?
Dr. Tibbetts: I check IOP about 20 minutes after the procedure, examine the fundus with an indirect ophthalmoscope, and then schedule the follow-up examination for a week later. We set the expectation at the outset that it may take more than one session for treatment. When patients return, we gauge their response to treatment. We ask, “From zero to 100, how much better are your floater symptoms?” And just as we do preoperatively, we correlate the anatomy that we visualize at the microscope to the patients’ subjective symptoms.
If patients say there’s no improvement, yet I no longer see the opacity, then I have to talk them through that. But more often than not, patients report a meaningful improvement in their symptoms. If they’re above 70% or 80% symptomatic improvement, I suggest we give it another month and I see them back. If their improvement is below that threshold, usually there is still an opacity there, and I re-treat.
Dr. Stonecipher: I give patients a steroid postoperatively for 5 days, much like what I do with YAG capsulotomy, but that choice is probably more for my peace of mind. I’ve never had a case of cystoid macular edema. We probably do not need topical drops, which I know some of you do not use, and we might not even need to see some of our patients in a week.
Dr. Shah: In our study, we didn’t treat with any drops postoperatively.1 We followed patients very closely, and there was no inflammation 30 minutes later or at 1 week or 1 month.
Dr. Singh: That’s a surprise to a lot of our colleagues, right? They say, “Oh no. You’re directing all this energy into the eye, so you’re causing inflammation,” but we actually are not. I don’t treat with a nonsteroidal anti-inflammatory drug (NSAID) or steroids postoperatively, but I don’t do anything from a postoperative inflammation standpoint with my YAG capsulotomy, either. I always say to colleagues, “If you’re used to using an NSAID or a steroid for your YAG capsulotomy, it’s not going to hurt to just keep doing that same regimen for vitreolysis. I don’t change my regimen for postoperative inflammation (using no drops), and I don’t see any inflammation.”
We’ve talked about patient selection in terms of overall diagnosis, but let me ask you about your preoperative assessment. When you’re in the room with your patients, what are you looking for? What characteristics do you want to see in a candidate for whom treatment is likely to succeed?
Dr. Mac: I want to get a good look at the posterior segment, examining the peripheral retina to make sure I don’t see any retinal pathology. If patients have a tear or a hole, we rule out vitreolysis and recommend our retina colleagues take over from there. If the retina appears healthy, I want to see the floater itself. The floater’s location to the retina and to the lens is very important to patient selection and planning where I need to treat during the procedure. I often bring patients back for a final look before they are treated if I feel uncomfortable with the location of the floater.
Dr. Stonecipher: I have a low threshold for performing optical coherence tomography (OCT), so I do an OCT on pretty much every patient. If the general rule is that an early epiretinal membrane exists in about 8% to 10% of the normal population, it makes sense to me to look at that for everybody. The other test I want to see is the Corneal Analyzer OPD-Scan III (Nidek), which gives me a lot of information and has software that allows me to show the patient the problem from a visual quality standpoint, much like we do with the iTrace system. It also allows me to say, “This is a normal cornea, and this is a normal lens. Now this is your eye….” Many times, we can see the shadows cast from the floater, which I think is important. By showing patients that they have a problem, it solidifies their understanding and desire for treatment. And I should mention that I have no vested interest in either of these devices.
Dr. Shah: In our study, we used wide-angle color photography, which was very helpful.1 It’s amazing how well you can see the floaters on the color photographs. The patient can point right at it and say, “That’s what’s causing me my trouble.”
Dr. Tibbetts: For the initial examination, it’s sometimes surprising what we see by taking a close look from the anterior to posterior vitreous. A patient may have an anterior vitreous opacity, perhaps a residual piece of the posterior capsule from a previous capsulotomy floating in and out. It’s important to know where that is as well as to make sure that symptoms match what you’re seeing. To Dr. Shah’s point about patients with higher potential for dissatisfaction, in my experience, those are the patients who more often have anterior amorphous opacities. If you’re just treating the Weiss ring in the mid-vitreous and not treating the anterior vitreous, which I think can be more challenging, your patient may not be as satisfied after surgery. It’s important to do a thorough examination of the vitreous so you can plan your treatment and counsel the patient accordingly.
Dr. Singh: I’m hearing that patient education and expectations are keys to success, and part of that process is the ability to show patients the problem. That’s my experience as well. I’ve had wonderful OCTs preoperatively and postoperatively demonstrating the resolution of the floater as well as the shadow cast on the retina. We have had patients referred to us who had MRIs and visual fields due to complaints of a scotoma, and no one thought, “Hey, maybe the vitreous floater is causing it.” OCT, fundus photography, and other modalities can help identify the floaters and the shadows they cast.
Dr. Stonecipher: One additional step I’ve taken is to ask patients to draw the problem. That helps me, patients love it, and it’s documentation that goes in the chart. It’s especially helpful for an enhancement or second procedure. I had a professional table tennis player who was very particular. He’s the only person I’ve ever had to treat three times. He kept saying, “It’s right here!” I couldn’t quite get to it because it was floating in and out of vision, so I said, “Let’s draw it.” Luckily, I was able to find out which floater was really giving him fits, and we treated it.
Dr. Singh: Listen to your patients. They tell you, “Hey doc, it always comes from up here,” and we can direct our attention to the inferior vitreous. By just listening to them and having them draw out their symptoms, you can better isolate where the opacity might be coming from. That might save you some time and keep you from lasering floaters that are not the main cause of symptoms.
Dr. Mac: I’m always amazed how symptomatic patients are from floaters. I ignored them in the past, and it’s just been amazing to me to learn how debilitating they can be for certain patients.
Dr. Singh: I was inspired to start doing LFR by a colleague of mine, a primary care doctor. He had come to me for years with a large floater, and I kept saying, “See you later. Bye-bye.” He went to a retina colleague of mine who said, “You’re phakic, you have a Weiss ring, and you’re a physician? Uh-uh. I’m not doing a vitrectomy. I’m not going to deal with the adverse event potential.” Instead, the doctor had vitreolysis performed by Dr. Karickhoff, who is one of the fathers of YAG vitreolysis. My colleague came back to see me and said, “Check out my floater.” It was gone. It took that kind of patient to make me realize, “Wow, this is something we can actually do to help our patients.”
When I do a preoperative workup, I want to look at the floater and its position, making sure that it’s not too close to the lens or the retina. I determine if the position correlates with the symptoms. I look for size density as well. If patients have a large floater that also has a relatively hard density, then I tell them ahead of time that it might take more than one session. I want to set the right expectations ahead of time, just like any patient having cataract or other surgery. Saying, “I see a large floater, and it may take two or three sessions to get you to our goal,” helps a great deal.
Dr. Mac: I think setting expectations is important. You have to tell patients, “I’m going to improve your symptoms 80%, but I’m never going to make you 100% symptom-free.” That leads to better patient acceptance of the results.
This is all relatively new to us. My vitreous class in school was about 2 hours long. Dr. Karickhoff’s book was exceptional,6 but it was new. We were taught to avoid the vitreous, and now we are learning that we can help.
Dr. Singh: Now we’ve taken his knowledge from a historical perspective and applied it through new technology. In the context of that new technology, I think it’s important to understand flow because this procedure is not a quick 5-second YAG capsulotomy. It may take 5, 10, or 15 minutes depending on the type of floater and your comfort level with visualization. We might be doing 200 or 300 shots or even more.
With a standard YAG laser, if you do 100 shots in a row, it overheats and goes “Beep!” and resets. Now with the Ultra Q Reflex and Tango Reflex, which have an active cooling cavity that allows you to do many shots at one time without resetting, the flow for the YAG vitreolysis procedure is much smoother. What’s your perspective on flow in your practice? Do you do vitreolysis the same day as your consult, have patients come back on certain laser days, or do you take another approach?
Dr. Tibbetts: I explain the treatment options, and then I always give patients time to think about it. I wait at least 4 to 5 months after the patient reports a symptomatic floater to treat it. I do a scleral depressed examination to ensure there’s no peripheral pathology, but I don’t rush into treating the floater.
Dr. Mac: I do the same thing, with about a 6-month wait.
Dr. Stonecipher: You have to give them time to think about it.
Dr. Tibbetts: Some patients are a little frustrated by the wait. They expected to get something done sooner. I advise my patients that we need to ensure that the posterior vitreous detachment is complete and no peripheral pathology is identified before treatment. Many patients will also have symptomatic improvement over time, which is another benefit of waiting.
Dr. Stonecipher: We take the opposite approach. We see a lot of patients, so we usually treat them the same day. They typically have a floater in each eye, so we do one eye and bring them back in 2 weeks to treat the other eye. Next, we either refer them back to their primary care physician or see them again in 3 months. That works from a billing perspective because my patients using insurance have a 90-day period after which we can recharge to perform an enhancement, if needed.

SAFETY
Dr. Singh: Lastly, I want to talk more about safety, the most important consideration and a big fear that all of us have with any new procedure. Dr. Shah mentioned the safety outcomes of his study, which were excellent.1 Have you all had similar experiences? Have you had any adverse events and, if so, how have you managed them?
Dr. Mac: I’ve learned that the type of floaters and the amount of energy that it takes to break up and vaporize them can influence postoperative IOP. In patients who have diffuse strands in the vitreous that require 800 or 900 shots, you have to watch for postoperative IOP spikes. Now I put those patients on dorzolamide/timolol combination drops (Cosopt; Merck) ahead of time to prevent a pressure spike, and we’ve never had a problem.
Dr. Shah: The Research and Safety in Therapeutics Committee at the American Society of Retina Specialists has been collecting cases of adverse events after YAG vitreolysis. Paul Han, MD, published a paper in July 2017 about the data.7 There wasn’t a denominator, but the paper noted that there were cases of glaucoma, retinal tear, retinal detachment, lens damage, and retinal hemorrhaging after YAG vitreolysis. Hopefully, as we get more data, we’ll be able to pull a lot of these studies together and get true rates of adverse effects.
Dr. Singh: There’s always a possibility of adverse events with any procedure and a risk/benefit analysis we go through. Does the procedure’s benefit outweigh the risk? The data set to which you are referring included data submitted from surgeons around the country. There were no specific criteria of which I am aware. Cases submitted were at the discretion of the surgeons. We do not know what laser technology they were using, what lenses, power setting, what type of floater was treated, and, in some cases, if there was previous pathology that placed the patients at a higher risk of a retinal defect. I do think that with the proper technology and the right protocol, we minimize adverse events.
My colleagues and I presented a paper at ASCRS 2017, a retrospective analysis of over 1,200 procedures that we performed with the Ultra Q Reflex, Reflex Technology.4 We found no retinal defects of any kind, no inflammation in the anterior chamber or vitreous, and no exacerbation of diabetic retinopathy or epiretinal membranes. We did have two phakic lenses that were hit, occurring within the first 20 cases. That was before we understood how to maximize visualization behind the lens by titrating the illumination. The most common adverse event was an IOP spike, affecting seven patients out of the 1,200+ procedures. Patients with IOP spikes tended to have floaters right behind the posterior capsule, post-YAG capsulotomy, and over 500 shots performed. Just as you said, Dr. Mac, I minimize not the power, but rather the number of shots for these patients because I think a higher number of shots raises the risk of an IOP spike for them.
If you feel uncomfortable with glaucoma patients, maybe have their IOP checked not only immediately after surgery, but also a few hours later or the following week, just to be safe. I always tell doctors who are just getting started that there’s no right or wrong. If you want to see patients a week later, you can see them a week later. Do whatever you like until you feel comfortable and understand the outcomes in your population.
CONCLUSION
Dr. Singh: I want to thank all of you for your insights. It’s a pleasure to be here with you, and I think it’s exciting for all of us to be part of this new paradigm shift in our profession. And with multiple studies ongoing as we speak, hopefully the emergence of new data will help more of our colleagues feel comfortable and start getting involved.
1. Shah CP, Heier JS. YAG laser vitreolysis vs sham YAG vitreolysis for symptomatic vitreous floaters: a randomized clinical trial. JAMA Ophthalmol. 2017;135(9):918-923.
2. Singh IP. Treating vitreous floaters: patient satisfaction and complications of modern YAG vitreolysis. Paper presented at: ASCRS; May 6-10, 2016; New Orleans.
3. Wagle AM, Lim WY, Yap TP, et al. Utility values associated with vitreous floaters. Am J Ophthalmol. 2011;152:60-65.
4. Singh IP. Neodymium: YAG laser vitreolysis: retrospective safety study. Paper presented at: ASCRS; May 5-9, 2017; Los Angeles.
5. Singh IP. Floaters: an underappreciated and undertreated problem. Cataract & Refractive Surgery Today. October 2016 (suppl).
6. Karickhoff JR. Laser treatment of eye floaters. Washington Medical Publishing. 2017.
7. Hahn P, Schneider EW, Tabandeh H, et al; American Society of Retina Specialists Research and Safety in Therapeutics (ASRS ReST) Committee. Reported complications following laser vitreolysis. JAMA Ophthalmol. 2017; 135(9):973-976.










