



CXL is a first-line intervention to stabilize the cornea and prevent disease progression in eyes with keratoconus-type disorders and ectasia. Debate continues regarding whether the epithelium should be removed for the procedure.
ON OR OFF? THE EPITHELIAL DEBATE
Epithelium-off (epi-off) CXL was approved by the FDA in 2016 for the treatment of progressive keratoconus and ectasia. The procedure remains the only FDA-approved version of CXL. The volume of epi-off CXL performed, however, has been limited by three main factors.
No. 1: The indications for epi-off CXL limit the scope of intervention and have significant implications for timing. Epi-off CXL is approved only for progressive keratoconus. Multiple visits are therefore required to establish proof of progression, even though it may be evident at the first visit that a patient has keratoconus and needs treatment. During the waiting period, the disease may continue to progress, and the patient may lose vision.
No. 2: An epi-off approach to CXL has a higher incidence of adverse events compared to an epithelium-on (epi-on) approach. Adverse events include corneal haze and scarring, infectious keratitis, sterile infiltrates, and delayed epithelial healing.1,2
No. 3: A longer postoperative healing period compared to after epi-on CXL may require patients to miss a significant amount of school and/or work. This concern is amplified by the required staging of treatment of the second eye.1,2
In contrast, epi-on CXL can be performed on both eyes simultaneously. Compared to epi-off CXL, epi-on CXL involves less discomfort because epithelial healing is faster, and patients are able to resume normal everyday function more quickly.1 Critics of epi-on CXL, however, argue that less disease stability after treatment could increase the need for a second CXL procedure.3,4 The reduced efficacy is thought to be multifactorial in nature.
No. 1: Riboflavin has a difficult time penetrating an intact epithelium. Tight junctions between individual cells in the corneal epithelium impede efficient absorption.
No. 2: UV-A light reacts with riboflavin. If the riboflavin is on or in the corneal epithelium, then the UV-A light penetrates the corneal stroma less efficiently.
No. 3: UV-A light is responsible for photooxidation and can destroy riboflavin. This reduces its effective concentration.
INTERVENTIONAL STUDY
Objective. We collaborated on a prospective, randomized, controlled, open-label, multicenter phase 2 clinical trial that evaluated epi-on CXL with a fixed-combination drug consisting of 0.5% riboflavin and 0.015% sodium iodide (RiboStat, CXL Ophthalmics) that is designed to increase epithelial penetration and decrease riboflavin breakdown.1
Methodology. Study participants were at least 8 years old when they were diagnosed with keratoconus or an ectactic disorder, and they had a minimum pachymetry reading of at least 375 µm. The study period was 6 months with an optional 1-year follow-up visit. If appropriate, treatment was bilateral. A total of 2,258 patients were randomly assigned to one of three UV-A light treatment protocols (see the accompanying sidebar below).
UV-A Light Treatment Protocols
4 mW/cm2 for 20 minutes in 15-second intervals
6 mW/cm2 for 20 minutes in 15-second intervals
4 mW/cm2 for 30 minutes in 15-second intervals
Of the 2,258 patients enrolled, 1,922 had been diagnosed with keratoconus. A total of 173 patients were treated at our center for keratoconus, pellucid marginal degeneration, ectasia in the setting of previous refractive surgery, and diurnal fluctuations in the setting of previous radial keratotomy.
The preoperative examination included noting the patient’s date of birth, diagnosis, history of eye rubbing or sleep apnea, uncorrected distance visual acuity (UDVA), best corrected distance visual acuity (CDVA), manifest refraction, IOP, and maximum keratometry reading (Kmax). No patient in our center’s trial had a history of eye rubbing; only one had a history of sleep apnea.
Patients were seen 1 day, 1 week, and 1, 3, 6, and 12 months after CXL. Follow-up information included UDVA, CDVA, manifest refraction, IOP, and Kmax. Primary endpoints included change in CDVA at 6 and 12 months compared to baseline. Secondary endpoints were the changes in UDVA and Kmax at 6 and 12 months compared to baseline.
Results. The full results of the study are available here. Following is a summary of our center’s results. The average change in Kmax observed was -0.45 D (n = 171). The Figure shows the pre- and postoperative CDVA percentages. The average pre- and postoperative IOP readings were 14.38 and 15.36 mm Hg, respectively. No adverse events, including loss of CDVA, were reported.
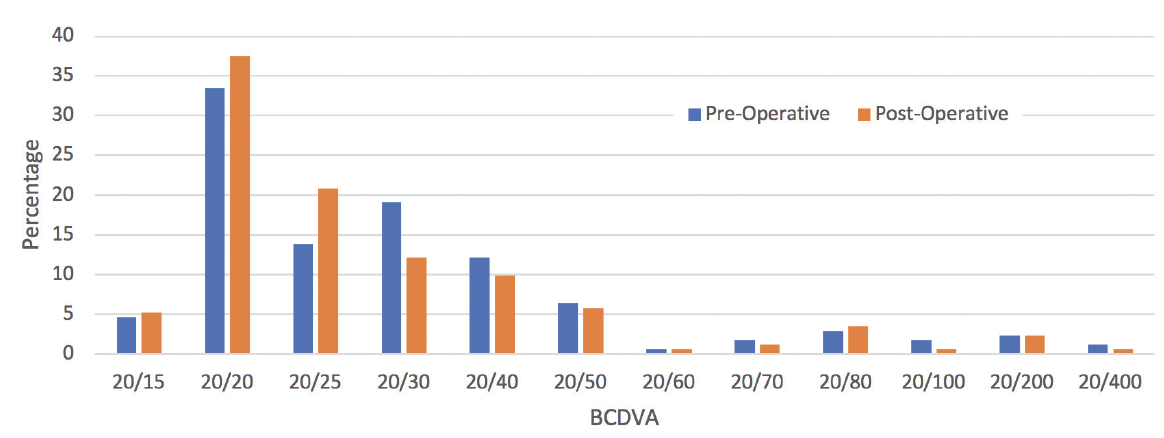
Figure. CDVA before and after epi-on CXL in Cleveland Eye Clinic’s trial. (Abbreviation: BCDVA, best corrected distance visual acuity)
Conclusion
Overall, our data were in line with those for the complete clinical trial. The results indicate that epi-on CXL is a viable alternative for preventing disease progression in eyes with keratoconus-type disorders and ectasia. An improvement in CDVA and unremarkable changes in both Kmax and IOP readings were demonstrated.
1. Rubinfeld RS. Results of a phase 2 trial of epi-on cross-linking for keratoconus. CXL Ophthalmics. Accessed March 14, 2022. https://www.cxlophthalmics.com/aao_14nov2021
2. Rubinfeld RS, Caruso C, Ostacolo C. Corneal cross-linking: the science beyond the myths and misconceptions. Cornea. 2019;38(6):780-790.
3. Gatzioufas Z, Raiskup F, O’Brart D, Spoerl E, Panos GD, Hafezi F. Transepithelial corneal cross-linking using an enhanced riboflavin solution. J Refract Surg. 2016;32(6):372-377.
4. Caporossi A, Mazzotta C, Paradiso AL, Baiocchi S, Marigliani D, Caporossi T. Transepithelial corneal collagen crosslinking for progressive keratoconus: 24-month clinical results. J Cataract Refract Surg. 2013;39(8):1157-1163.




