A common scenario in most busy cataract practices is the mature cataract in the setting of potential zonulopathy. During surgery, the lens must be freed from the capsular bag, disassembled with little to no traction placed on the zonules, and emulsified with minimal ultrasonic energy and vacuum to reduce the risk of trauma to the corneal endothelium and posterior capsule, respectively.
Successful hydrodissection allows the surgeon to separate and spin the lens within the capsular bag. When hydrodissection fails, some ophthalmologists move on to hydrodelineation, but skipping hydrodissection can make spinning the lens more difficult and render subsequent cortical cleanup cumbersome.
Why does hydrodissection fail? Fluid always follows the path of least resistance. If the cannula tip is too close to the edge of the capsulorhexis or if the cannula is elevated, it can create a potential space and cause the fluid wave to reflux anteriorly rather than posteriorly. The key is to position the cannula so that the path of least resistance is not anterior but posterior. I designed the flat, right-angled Kim Capsular Fornix Hydrodissection Cannula (Katena) to be placed deep in the capsular fornix and call the technique “capsular fornix hydrodissection.” The instrument has a blunt tip that is 1.8 mm long, and its placement is similar to that of a chopper for horizontal chopping. The cannula’s tip is rotated downward to face the optic nerve. After positioning the instrument, it is important not to exert any pressure on the lens, capsule, or zonules. Otherwise, the surgeon will encounter resistance when trying to push the fluid wave. I recommend subtly wiggling the cannula back and forth within the iris plane, which will ensure that the tip is between the capsule and the lens and not pushed up against the capsular bag or within the lenticular material. When the plunger is pushed, an easy-flowing wave of hydrodissection should result without significant resistance.
With propagation and completion of the hydrodissection fluid wave, the capsular bag can fill with balanced salt solution to create a high-pressure compartment. The trapped balanced salt solution will cause a positive pressure effect in the space between the posterior capsule and the lens. Pushing on the lens with the cannula will decompress the bag and release this fluid pressure, helping to break any adhesions between the capsule and cortex. I have found that the maneuver improves mobilization of the lens. Specifically, I turn the cannula’s tip to the left and sweep it along the left anterior capsular rim while I gently inject balanced salt solution; I can thus use both mechanical and fluid dissection forces. After the right side of the capsular rim is freed in a similar fashion, the lens should spin freely within the capsular bag.
Disassembling a brunescent lens in the presence of zonulopathy is challenging: the nucleus is difficult to fracture, and poor zonular support can make manipulating the lens complex. When surgery does not proceed smoothly, the ophthalmologist may feel inclined to use more ultrasonic energy and vacuum, but doing so increases the risk of corneal endothelial and zonular trauma, including posterior capsular rupture, vitreous loss, and a dropped nucleus. I favor two lens disassembly techniques in this situation that reduce the use of ultrasonic energy and bypass the need to rotate the lens within the bag, thereby minimizing stress on the zonules.
The first, which I call “double-chop,” is founded on the principle of mechanical fracturing.1 It requires an understanding of vector forces, a chopper, and a phaco handpiece. I start by placing the chopper (Kim Double Chopper, Katena) in the capsular fornix, similar to horizontal chop, but then I tilt the instrument so that its tip is below the lens. Instead of impaling the lens, the phaco tip is tilted vertically under irrigation alone; no ultrasonic energy or vacuum is applied. Next, I raise the chopper while pushing down with the phaco tip so that the two instruments meet in the center. Double-chop uses centripetal and diagonal vector forces (Figure). Most importantly, the technique allows the two instruments to crush the central endonucleus, which is the densest portion of the lens.
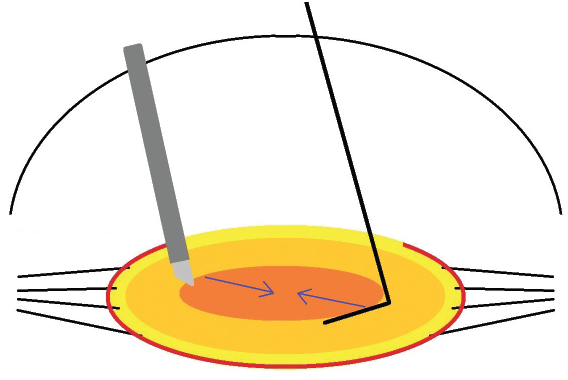
Figure. The surgeon places the double chopper into the equator and rotates the instrument vertically such that its tip is underneath the endonucleus. The phaco tip is also tilted vertically on top of the endonucleus to improve mechanical leverage. Bringing both instruments to the center with centripetal and diagonal vector forces crushes the lens into two heminuclei. The maneuver uses no ultrasonic energy or vacuum and places no traction on the zonules.
I subsequently use the cross-chop technique, which I perform as follows.2 I place the chopper around the contralateral equator while holding the phaco tip between the two heminuclei. Pulling the chopper centrally chops the heminucleus. In a matter of seconds, I have disassembled the lens into three pieces without ultrasonic energy or vacuum. By not spinning the lens, I have avoided placing torsional stress on the zonules, and because all the vector forces were directed toward the center, I kept the zonules free from traction.
Watch it Now
CASE EXAMPLE
An 84-year-old white woman was referred to me for cataract evaluation. A slit-lamp examination showed pseudoexfoliative material on the anterior lens capsule in each eye but more so in the left eye. The patient had 2+ to 3+ nuclear sclerosis with a 2+ cortical cataract in her right eye and 3+ nuclear sclerosis with a 1+ to 2+ cortical cataract in her left eye. There was no obvious phacodonesis, iris atrophy, or history of trauma.
I used the techniques of capsular fornix hydrodissection, double chop, and cross-chop described earlier, and surgery proceeded uneventfully. I freed the lens from the capsular bag without difficulty. Centripetal, mechanical vector forces crushed the endonucleus, disassembling the lens effectively without causing zonular traction. The cumulative dissipated energy was 2.32%-seconds in the right eye and 1.79%-seconds in the left eye.
One day after surgery, the corneas were clear, and anterior chamber reaction was minimal. One week after surgery, the patient had a BCVA of 20/20 OU.
Take-Home Points
- The hydrodissection wave always follows the path of least resistance.
- Capsular fornix hydrodissection enables the surgeon to achieve a highly reproducible wave because deep placement of the cannula’s tip prevents anterior fluid reflux.
- A gentle wiggle of the cannula ensures that its tip is not pushed against the capsular bag or into lenticular material. The tip should be placed between the capsule and lens, which will improve the effectiveness of capsular fornix hydrodissection.
- Disassembling the endonucleus with centripetal mechanical vector forces reduces zonular stress and minimizes the use of vacuum and ultrasonic energy.
- These techniques can be used in a wide range of challenging situations, including dense lenses, soft nuclei, weak zonules, and small pupils.
1. Kim DB. Double-chop: modified-chop technique eliminating ultrasonic energy and vacuum for lens fragmentation. J Cataract Refract Surg. 2016;42(10):1402-1407.
2. Kim DB. Cross chop: modified rotationless horizontal chop technique for weak zonules. J Cataract Refract Surg. 2009;35(8):1335-1337.

