CASE PRESENTATION
A 56-year-old man underwent pterygium surgery on both eyes 2 years ago and cataract surgery on his right eye 1 year ago. Unhappy with his hyperopic outcome, the patient referred himself to Duke University School of Medicine in Durham, North Carolina, where cataract surgery was performed and a toric IOL was implanted in his left eye in January 2020. One month later, he underwent successful toric IOL rotation in his left eye. The following refractions are stable: +1.00 -0.25 x 006º = 20/20 OD and plano -0.50 x 125º = 20/20 OS.
The patient greatly desires surgical correction of his hyperopic right eye. The ocular surface of that eye is dry (Figure 1). Additionally, there is a mild recurrence of pterygium with scarring and neovascularization that is not visually or topographically significant at this stage (Figure 2). The posterior capsule is intact and exhibits minimal opacification.
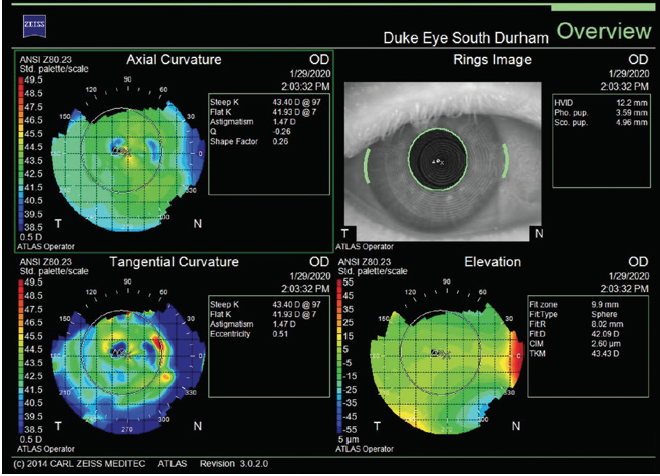
Figure 1. Corneal topography of the right eye shows mild to moderate tear film instability.
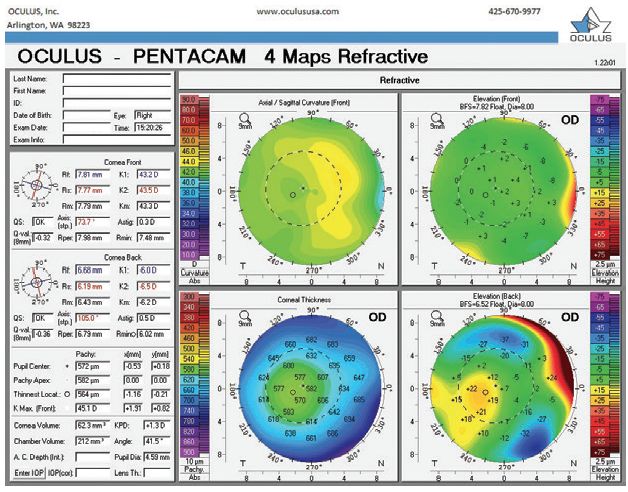
Figure 2. Imaging with the Pentacam (Oculus Optikgeräte) confirms that the corneal scarring and a mild recurrence of pterygium are not visually significant.
Given this patient’s history of ocular surface disease (OSD), pterygium surgery, and corneal neovascularization, how would you approach correcting the residual hyperopia? How would his age and history of chronic OSD influence your decision to perform a laser corneal procedure, an IOL exchange, or a piggyback secondary IOL implantation?
—Case prepared by Alan N. Carlson, MD

MELISSA B. DALUVOY, MD
This patient’s vision is easily corrected with glasses. If he is bothered by the anisometropia and his goal is to be independent of contact lenses and glasses, he has several options. The time that has elapsed since the original cataract procedure may make an IOL exchange challenging, and OSD renders laser refractive surgery a less attractive option than a lens-based procedure. Moreover, pterygium excision made the topography slightly irregular, and signs of dry eye disease (DED) are evident at the slit lamp, although the patient is asymptomatic. For these reasons, a piggyback IOL would be my preference.
The anterior chamber depth measured approximately 4 mm with the Lenstar (Haag-Streit). Roughly 1.50 D of correction is required, but 0.50 D increments often are not available for low power ranges. An argument can be made for implanting a 2.00 D IOL, which would leave this patient slightly myopic. My strategy would be to implant a 2.00 D LI61AO SofPort IOL (Bausch + Lomb) because this silicone lens is available in low powers. I would expect the procedure to be straightforward.

RICK WOLFE, FRACS, FRANZCO
A review of methods of refractive correction after IOL implantation concluded that laser refractive surgery provides more effective and predictable outcomes than other methods.1 I usually perform LASIK rather than PRK for patients such as this one because the former procedure offers faster visual recovery. But there are several drawbacks to LASIK in this case.
First, the extent of the pterygium recurrence and the corneal diameter might prevent adequate centration of a flap of the minimum diameter required for a hyperopic ablation. A thicker flap might be safer to create at the site of previous surgery where there is scarring. Neovascularization can be annoying at the time of surgery, but it is usually manageable. LASIK is not an uncommon procedure after pterygium surgery and graft placement in my practice.
More important, the Placido rings and topography suggest significant OSD of some sort. A diagnosis of possible coexisting epithelial basement membrane dystrophy and DED should be considered and treated. In addition to other DED management strategies, I would perform intense pulsed light treatment because of its efficacy and patients’ relatively fast recovery. Were epithelial basement membrane dystrophy to be found on clinical examination or OCT, I would perform a phototherapeutic keratectomy.
If this patient’s response to DED treatment is not significant, both LASIK and PRK are contraindicated. In that situation, I would implant a secondary sulcus-fixated IOL. I would avoid lenses with sharp edges that could come into contact with the posterior iris, and there would have to be enough room for the lens. IOLs that meet these specifications are not available in all markets. Compared to an IOL exchange, this surgical procedure is easy to perform and does not pose a risk to the zonules, but it is an expensive option.

DISCUSSION: ALAN N. CARLSON, MD
This case typifies the current era of high expectations for surgical outcomes after refractive cataract surgery. The situation is largely of our own doing (or undoing, as it were) when we prepare patients for surgery by offering spectacle-free lens options, frequently for an additional out-of-pocket expense. Patients may believe that we promised more than we delivered.
Only 2 decades ago, this patient would have received bifocal spectacles, and most surgeons would have considered the outcome successful. In contrast, dissatisfied with the mildly hyperopic outcome for his first eye, the patient left his original provider and transferred to another surgeon at another facility for treatment of his second eye as well as correction of the hyperopic outcome of the first eye.
Drs. Daluvoy and Wolfe offer different approaches, each highlighting important strategic features. Although it is hard to argue against the precision of LASIK, a patient who has experienced an early recurrence of pterygium after surgery and who has corneal neovascularization and OSD is far from an ideal LASIK candidate. Preoperative management would be required to maximize the health of the ocular surface, eliminate staining with vital dye (lissamine green or rose bengal), and provide assurance that the peripheral cornea and limbus will not impede suction and flap creation. PRK is less likely to aggravate surface dryness, but it is not an ideal procedure in the setting of corneal neovascularization. When I see neovascularization in the retina, the first thing I think of is ischemia. Neovascularization of the cornea frequently results from mechanisms other than ischemia such as epitheliopathy or an epithelial defect. This patient’s ocular surface places him at risk for progression of the neovascularization after PRK.
An IOL exchange is an option. As Dr. Daluvoy mentions, it would be important to study the healing pattern this long after surgery. If the anterior capsular edge covers the anterior surface of the optic, the bag can usually be opened with a cohesive OVD. If, however, the anterior capsule has fused to the anterior surface of the posterior capsule without visible epithelium in that interface, it may be a much more challenging procedure and riskier for the patient. In that case, implantation of a secondary IOL is an option.
I like the recommendation of a silicone IOL by Dr. Daluvoy because the original IOL is acrylic and the compressive interaction between two acrylic IOLs can reduce refractive predictability. I agree with the suggestion of sulcus fixation based on my experience in a similar case. In that case, I selected the correct secondary IOL power but implanted the lens in the bag. The secondary IOL retropulsed the first IOL, thus reducing the effective IOL power and leaving the patient with persistent hyperopia.
I agree with Dr. Wolfe’s recommendation to avoid sulcus-fixated IOLs that have a sharp or square edge because I have seen iris chafing, pigment dispersion, transillumination iris defects, and even secondary glaucoma when those styles of IOL have been used in a situation such as this one. For example, an MA60MA IOL (Alcon) is available in low powers (-5.00 to +5.00 D) for the capsular bag but also has a relatively thick edge that could be unfriendly to the posterior iris.
Before refractive surgery and refractive cataract surgery, even though outcomes improve every year, it is important to document discussion of possible outcomes, including a refractive margin of error. Preoperative discussion helps to set reasonable expectations, whereas patients may perceive the same points made after surgery as a complication.
1. Sáles CS, Manche EE. Managing residual refractive error after cataract surgery. J Cataract Refract Surg. 2015;41(6):1289-1299.




