CASE PRESENTATION
A healthy, active, 80-year-old man presents with a complaint of seeing halos in his left eye, particularly around lights, when in a darkened room or driving at night. The patient has no complaint about his daytime vision or about the vision in his right eye. He acknowledges that he sees well but says that the halos are intolerable.
The patient underwent bilateral cataract surgery in 2017. He says that he developed some glare symptoms in his right eye several months after surgery but that they resolved after an Nd:YAG capsulotomy. He reports that he has consulted several other ophthalmologists regarding the halos and has undergone three Nd:YAG laser capsulotomies on the left eye that achieved no improvement in his symptoms.
UCVA is 20/20 OU. An examination reveals a three-piece IOL in the bag and an open capsule in the patient’s right eye (Figure 1). The left eye has a well-centered one-piece clear hydrophobic acrylic IOL and a wide-open posterior capsule (Figure 2).
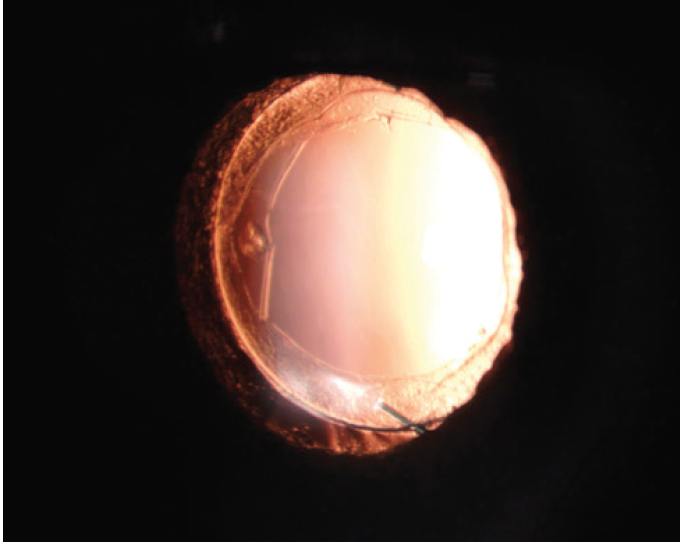
Figure 1. A well-centered three-piece IOL in the right eye.
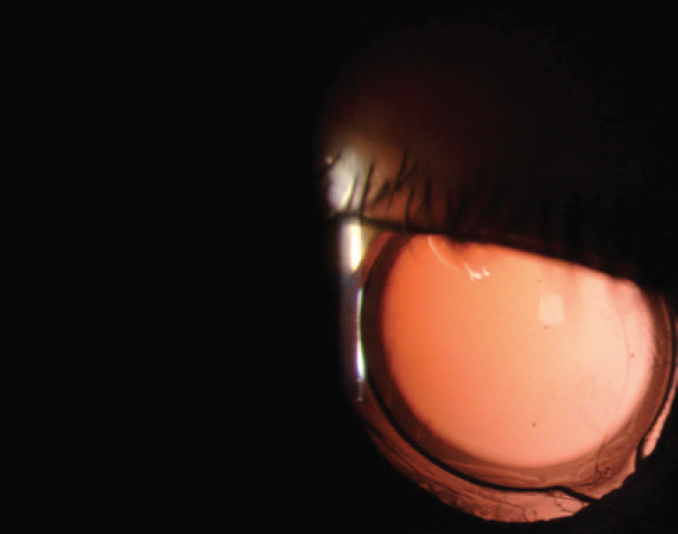
Figure 2. A well-centered one-piece acrylic IOL in the left eye.
The patient says that he would like the quality of vision in his left eye to be the same as that in his right eye. He adds that he is planning to maintain an active lifestyle for at least the next 15 years.
How would you proceed? Would you offer an IOL exchange?
—Case prepared by Audrey R. Talley Rostov, MD

JENNIFER LOH, MD
This patient is experiencing positive dysphotopsia (halos), possibly from an issue with the one-piece IOL in the left eye. Before considering an IOL exchange, I would closely evaluate the cornea for issues in the ocular pathway that could be causing the halos. I would perform a careful slit-lamp examination with fluorescein and check for epithelial basement membrane dystrophy, dry eye disease, and/or Salzmann nodules. I would also obtain topographic measurements to determine if irregular astigmatism—which can produce higher-order aberrations (HOAs) that can cause halos—is present. I would evaluate the size of the dark-adapted pupil in case it is wider than the lens optic, which, although an uncommon situation in elderly patients, can cause halos. Additionally, I would obtain an OCT scan of the macula to identify any foveal irregularities such as an epiretinal membrane or cystoid macular edema. Although UCVA in this eye is 20/20, even subtle foveal abnormalities can cause symptoms. A manifest refraction would also be essential. Again, although the UCVA is 20/20, I would like to know if a small residual refractive error is contributing to this patient’s visual symptoms.
The one-piece IOL appears to be well centered within the capsular bag, behind the anterior capsule. If a thorough evaluation reveals no other pathology, I would prescribe a topical medication to reduce the size of the pupil, which may decrease or eliminate the halos. Options include brimonidine or low-dose pilocarpine. If either of these medications were effective, then the patient might avoid surgery. If not, I would consider exchanging the current IOL for a three-piece monofocal IOL placed in the sulcus.

P. DEE G. STEPHENSON, MD, FACS
Unsatisfactory visual quality continues to be a common complaint among cataract surgery patients. As a surgeon, one of the most difficult decisions I face is choosing the IOL that I believe will give the patient in my hands the best quality of vision and the lowest degree of HOAs. All IOLs have limitations, some minor and some not.
In this case, I would like to know which IOL model is in each eye and why the patient did not receive the same model bilaterally. Second, I would like to know the size of each pupil and which eye is dominant. Third, I would like to know if there is any residual refractive error. Fourth, I would like to know if any lens tilt or decentration has occurred, and I would use the iTrace (Tracey Technologies) to make this determination.
The right eye probably received an aspheric fourth-generation silicone IOL that has few HOAs. Silicone tends to provide a better quality of vision than an acrylic material. My guess is that the right eye has an aspheric LI601AO IOL (Bausch + Lomb), which has an equal power from center to edge, and, based on the ridges visible in Figure 2, that the left eye has a Tecnis One-Piece IOL (Johnson & Johnson Vision). The latter lens can cause HOAs in patients with large pupils.
I would begin by determining if a residual refractive error is present in the patient’s left eye. If so, I would correct the error with glasses to see if his symptoms resolve. Another option would be to have the patient administer low-dose pilocarpine or brimonidine in the left eye at night and report whether the symptoms resolve. The next step—and probably the most definitive one—would be an IOL exchange with placement of a silicone three-piece aspheric IOL, either in the bag or the sulcus, after obtaining informed consent detailing the risks of the intervention. Because of the open capsule after three laser capsulotomies, I doubt that the anterior hyaloid face is intact. I would therefore be extremely careful not to lose vitreous and to preserve whatever capsular support remains while removing the current IOL.
Cases such as this one are challenging, and I expect them to become more common as the practice of mixing IOL models is employed for a growing number of patients.

MARIE-JOSÉ TASSIGNON, MD, PHD, FEBO
When patients complain about poor image quality, the first step is to consider IOL design. Why did this patient receive a different IOL in each eye? Did he undergo cataract procedures by two different surgeons? The case presentation does not address these questions. Nor does it disclose which eye is dominant. Based on the patient’s complaint, the left eye may be dominant.
The right eye received a three-piece IOL, and the patient was happy with the result after undergoing an Nd:YAG laser capsulotomy. The left eye received a one-piece IOL, and its optic presents a peripheral rim. Although the IOL is not identified, I suspect that it is a Tecnis One-Piece lens. The peripheral rim of this IOL can cause glare symptoms if the eye has a large pupil at night or if the optic is relatively posteriorly located or tilted. When a patient complains about glare, I find it helpful to obtain additional measurements such as with aberrometry (iTrace) and a straylight meter (C-Quant, Oculus).
Without more information, it is difficult to draw a conclusion, but I suspect that the patient’s complaints are not caused by the lens haptics. Rather, I think that he has HOAs caused by the profile of the lens optic. If that hypothesis is confirmed to be correct, the problem can be solved only with an IOL exchange. An Nd:YAG laser capsulotomy would not be a viable alternative. I would take advantage of the remaining anterior and posterior capsules and implant a Bag-in-the-Lens (Morcher, not available in the United States). If capsular support were insufficient, then I would place bean-shaped rings (Morcher, not available in the United States) to stabilize the IOL.

WHAT I DID: AUDREY R. TALLEY ROSTOV, MD
The patient had two distinctly different IOLs, but both were well centered. The three-piece silicone IOL in the right eye was not causing symptoms. The one-piece acrylic IOL in the left eye was causing halos, probably from the edge glare and bevel and the IOL’s position. Because this patient had already undergone three Nd:YAG laser capsulotomies, the capsule was wide open, and an IOL exchange would have necessitated a vitrectomy and placement of a scleral-fixated IOL.
I had a long, frank discussion with the patient. I explained that no IOL is perfect. I emphasized that UCVA was 20/20 in each eye and that the only time the halos troubled him was at night. He agreed to a trial of brimonidine drops administered at night and/or when he would be in a darkened area such as a movie theater.
After a 3-month trial, the patient reported some improvement in his perception of halos around lights when he was driving at night. He does not like the idea of administering eye drops at night for the rest of his life, so he plans to reevaluate his situation after another 3 months, during which time daylight hours will be shorter so his time driving at night will increase.




