Over the past decade, cataract surgery has become increasingly refractive in nature, and conversations about IOL calculations and refractive accuracy have therefore dominated the planning phase of cataract surgery. Previous studies have demonstrated several causes of inaccuracies in lens power calculations.1 Sources of error include effective lens positioning (ELP 35%), axial length (27%), and refraction (17%).2 During a recent presentation at the American Academy of Ophthalmology’s annual conference, Douglas Koch, MD, posed the question, “When calculating IOL accuracy, how well do we actually do?” Within the setting of his clinical practice, he is able to achieve approximately 70% accuracy within a half diopter. According to Dr. Koch, accuracy decreases in more complex eyes, and there is room for improvement.
Measuring the Cornea
Today’s cataract surgical landscape continues to evolve, and other factors have emerged to become the focus of improving IOL power calculations. ELP is still the primary goal, but corneal keratometry—both anterior and posterior—is now considered to be the greatest challenge to achieving accurate ELP. Refraction has not changed much, but axial length measurement errors have been greatly reduced by SS-OCT devices that offer superior accuracy of optical biometry. SS-OCT has also provided an improved ability to penetrate dense cataracts previously requiring ultrasound measurements.
IOL power is traditionally calculated using vergence formulas, including Barrett Suite formulas, Holladay 1 and 2, Hoffer Q, SRK/T, and Haigis, as well as other methods like ray tracing and AI. More or less, vergence formulas calculate ELP, a challenge that continues to prove difficult in some cases. Direct OCT measurements and estimation of ELP have been done, but none of these methods are commonly employed.
What is the best practice today? In my practice, the importance of accurate cornea measurements drives what I do. Utilizing multi-zone telecentric keratometry instead of simple LEDs, the IOLMaster 700 (Zeiss) provides consistent K values independent of measurement distance, leading to increased accuracy and speed. This provides not only quantitative information but also qualitative information regarding the accuracy of the measured K’s. With the addition of SS-OCT, corneal thickness can be measured, and the posterior corneal curvature can be determined. Coupling this data with the Barrett Universal II—and now two exclusive new formulas, Barrett TK Universal II and Barrett TK Toric—favorable results in increasing accuracy in refractive outcomes have been shown, even in eyes that have undergone previous corneal refractive surgery.3
Patient Case Study
A 71-year-old female patient presented with classic cataract symptoms. She had blurry vision and nighttime glare OU, making it very difficult for her to drive. She also had some dry eye symptoms, with a SPEED score of 9. The refraction was hyperopic with some against-the-rule astigmatism. Interestingly, on exam, she had 2+ nuclear and cortical cataracts OU and moderate nasal pterygia in OU (1.6 mm OD, 1.8 mm OS). The question of whether the pterygia are affecting keratometry, and therefore visually significant, needed to be addressed. Biometry with the IOLMaster 700 can be very helpful because images of both the eye and the Purkinje reflections/spots used to calculate the K’s can be evaluated, thus minimizing refractive surprises. Additionally, the internal checks of consistency between the spots from the various zones and over time from 15 discreet measurements are highlighted in the report (Figure 1).
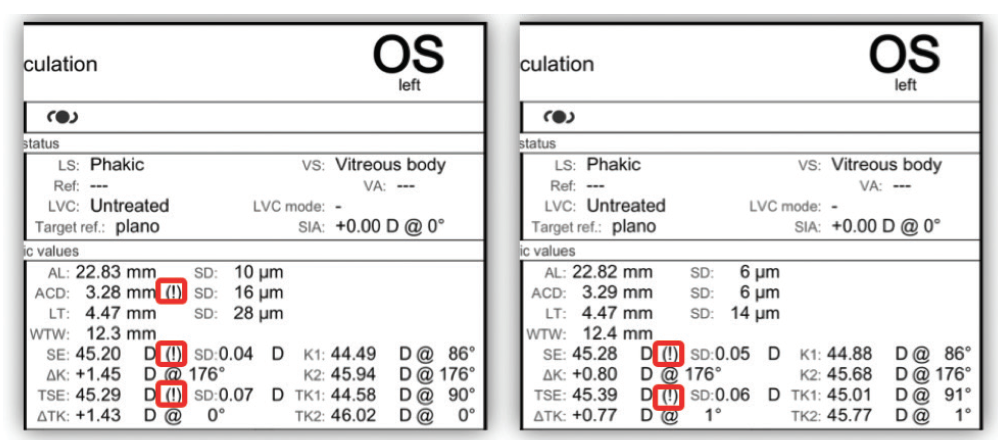
Figure 1. IOLMaster700 highlights areas of concern (!). Repeated measurements also demonstrated a 0.65 D change in astigmatism between two exams.
In this case, even though the standard deviation (SD) metrics were good, the exclamation points (!) on the biometry report suggested that further evaluation was warranted. Corneal topography was performed with the Atlas Topographer (Zeiss), demonstrating irregular mires, nasal flattening, and an unstable ocular surface OS (Figure 2). Interestingly, the topographic map had a horizontal bowtie, suggesting that the pterygium was not contributing to the keratometric nor the refractive astigmatism, but rather exacerbating dry eye. Similar findings were noted OD.
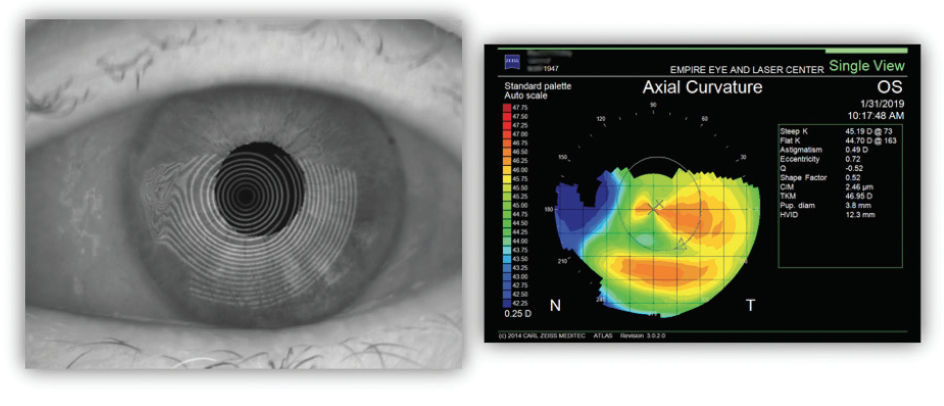
Figure 2. Irregular mires and nasal flattening due to pterygium.
This patient was treated with Vectored Thermal Pulse therapy (LipiFlow, Johnson & Johnson Vision). Three weeks later, we repeated the biometry and noticed no highlighted areas of concern. This implies an improvement to the patient’s dry eye. Repeated biometry measurements also demonstrated a keratometric variability of only 0.05 D, another indication of an improved ocular surface.
It is vital to pay attention to the ocular surface before creating a surgical treatment plan. Ocular surface variability can be a significant source of error, which is why having both quantitative and qualitative analysis can be important. When there are indications that there may be quality concerns with the biometric measurements, additional testing should be performed to identify possible additional pathologies. Having this kind of detailed information is extremely helpful in counseling the patient, discussing options, and devising a surgical plan.
Creating a Patient Plan
Every patient is unique, so it is important to determine how much time and effort is needed to obtain the patient’s desired outcomes. For my patients, I start with their visual history and expectations. I learn about their lifestyle and how they characterize their symptoms in a real-world setting. Some patients don’t mind continuing spectacle wear, so for these patients I simply perform a single measurement. For patients who desire astigmatism and/or presbyopia correction, I get at least two full sets of measurements to determine consistency.
Conclusion
To achieve the best possible refractive outcomes, it is important to develop a comprehensive approach to IOL calculations. Integrating technologies like telecentric keratometry, SS-OCT, and the latest generation formulas can not only help to reduce the risk of refractive surprises, but also foster better patient education, stronger surgical strategies, and improved patient outcomes.
1. Lee AC, Qazi MA, Pepose JS. Biometry and intraocular lens power calculation. Curr Opin Ophthalmol. 2008;19(1):13-17.
2. Norrby S. Sources of error in intraocular lens power calculation. J Cataract Refract Surg. 2008;34(3):368-376.
3. Refractive outcomes in post-myopic LVC eyes–Barrett True K formula with Total Keratometry (TK). zeiss.com https://www.zeiss.com/meditec/int/resource-center/clinical-insights/articles/ophthalmic-diagnostics/refractive-outcomes-in-post-myopic-lvc-eyes.html. Published September 9, 2019. Accessed November 20, 2019.




