CASE PRESENTATION
In the past year, the question I have heard from doctors most often has gone something like this: “I have a postrefractive surgery patient on whom I performed cataract surgery, and now I have a refractive surprise. What can I do to fix it?” I have therefore devoted this and my previous two installments of this series to answering this question with the help of several respected experts on the subject. The goal is prophylaxis. How can ophthalmologists fine-tune cataract surgery outcomes in patients with a history of refractive surgery, and how can surgeons prevent postoperative problems with quality of vision? In each case, the presumption is that no refractive information is available prior to the intervention.
A 73-year-old man presented for evaluation of decreased vision in his right eye. The patient had undergone hyperopic LASIK in 2001 for a manifest refraction of +1.50 +0.50 x 090º.
An examination revealed +2 nuclear sclerotic changes with +1 cortical changes. UCVA was 20/50 OD. He had a manifest refraction of -0.50 +0.50 x 180º = 20/30. Glare testing produced a visual acuity of 20/50. No imaging with the Pentacam (Oculus Optikgeräte) was available.
The patient underwent cataract surgery on his right eye (Figures 1 and 2). Based on intraoperative aberrometry using the ORA System (Alcon), a 22.00 D AcrySof IQ monofocal IOL (model SA60AT, Alcon) was implanted. Six months after surgery, UCVA was 20/25. BCVA was 20/20, and the manifest refraction was +0.50 D.
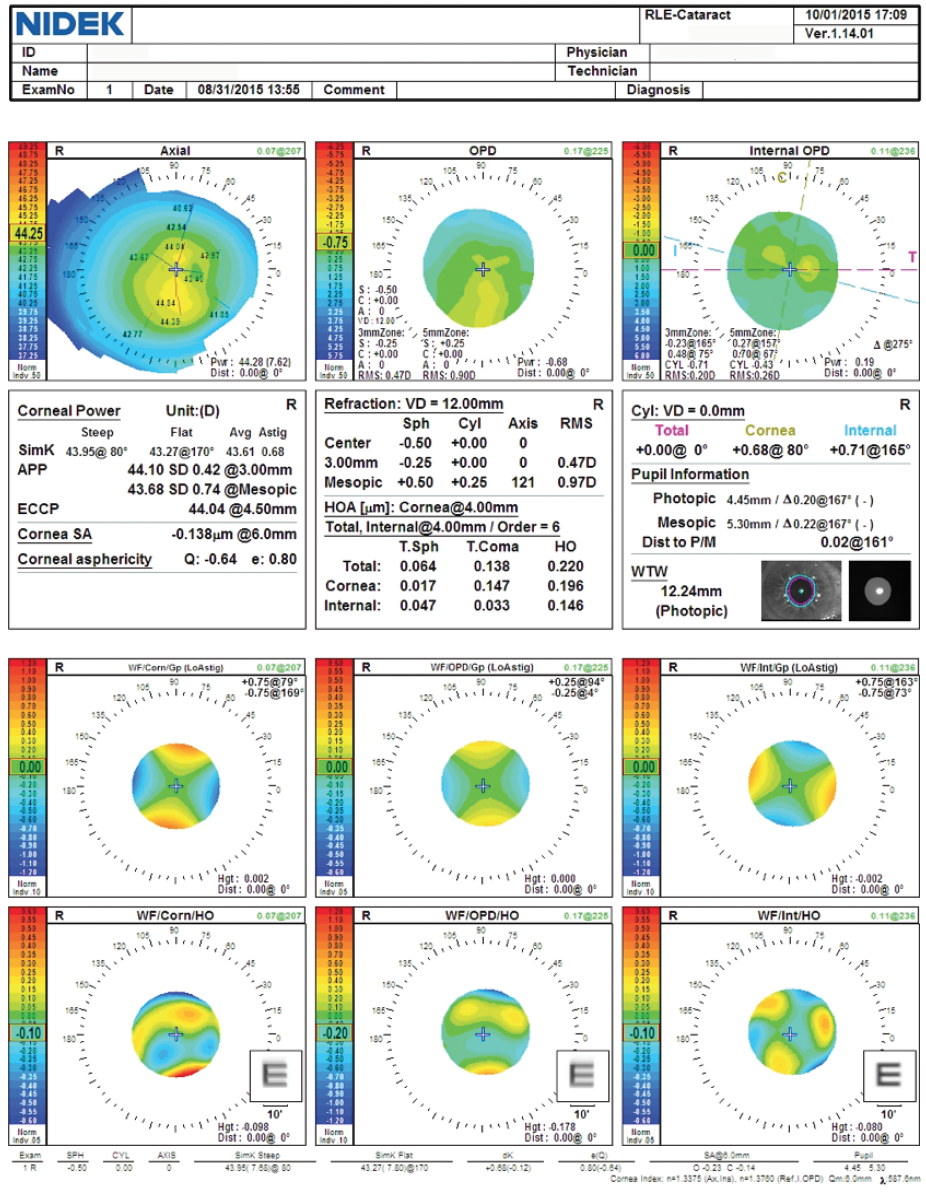
Figure 1. Preoperative computed topography and wavefront data using the OPD-Scan III (Nidek).
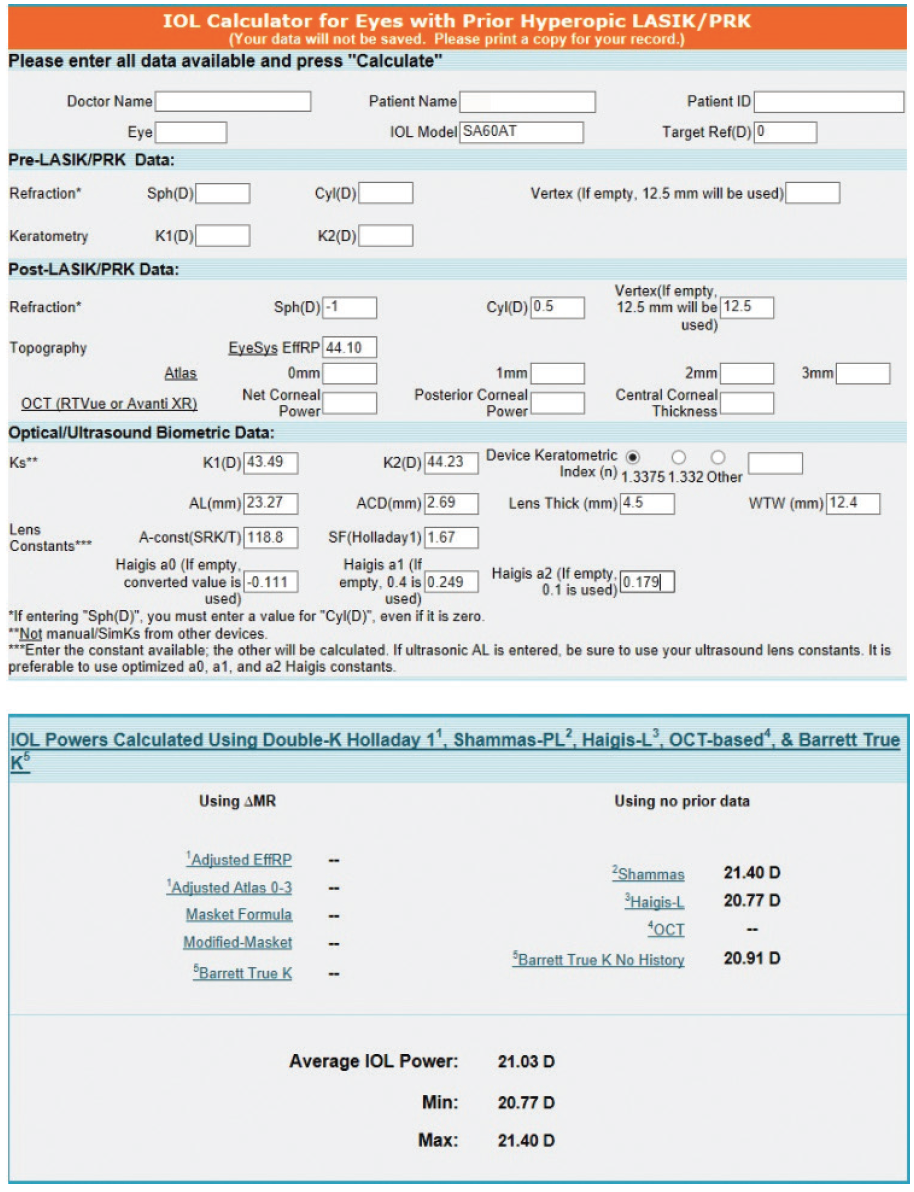
Figure 2. Preoperative calculations using the ASCRS IOL power calculator for prior hyperopic LASIK/PRK.
How would you proceed?
—Case prepared by Karl G. Stonecipher, MD

WARREN E. HILL, MD, FACS
This patient had early-generation hyperopic LASIK. Unlike patients who receive myopic LASIK, those undergoing hyperopic LASIK typically have a modest spherical equivalent, and relatively minimal treatment is undertaken. This patient is in his early 70s. His postoperative distance UCVA is 20/25, improving to 20/20 with 0.50 D sphere.
At a minimum, the preoperative evaluation of a patient with a history of hyperopic LASIK should involve obtaining a topographic axial curvature map, aberration profile, and standard optical biometry measurements that would include automated keratometry. A 6-mm–aperture anterior corneal aberration profile will help drive the selection of the IOL type. Spherical IOLs are typically used after LASIK correction for large amounts of hyperopia, which shifts the anterior corneal spherical aberration to negative values. It is more common for the anterior corneal spherical aberration value simply to be reduced to something close to zero, and, in that scenario, an aberration-neutral IOL is often placed. For this patient, we have only a 4-mm spherical aberration value, which is not standard but is a low value and still positive.
For determining IOL power, I now look most closely at calculations using the Barrett True K method, which I find generally gives the best overall results for a series of patients. In this case, using the recommendation of the Barrett True K method would have resulted in a low hyperopic outcome. The ORA System recommended an IOL power within 0.50 D of plano. This more accurate calculation by the ORA System is atypical because I consider the Barrett True K method to be one of the most accurate postrefractive tools we have to work with.
The take-home lesson from this case is that each and every one of these calculations is an estimate and not an exact calculation. A recurring problem is that all keratorefractive procedures alter the Gullstrand ratio in different ways in different areas of the cornea, resulting in different errors in different places. Add to that the facts that many ablations are not centered on the corneal vertex and that formulas can be misled by iatrogenic steepening or flattening of the cornea. Variable refractive results are to be anticipated, and there can be no guarantee of an exact refractive outcome. For experienced surgeons, the ±0.50 D accuracy for a series of patients after LASIK is generally between 65% and 75%.
This patient achieved a result within 0.50 D of plano, so no further treatment is necessary.

DAVID L. COOKE, MD
I always try to use aspheric IOLs in eyes with a history of myopic laser refractive surgery and spherical IOLs in eyes with a history of hyperopic laser correction. The latter subset of patients causes me less grief overall. Hyperopic treatments are small. I can use the numbers straight from the biometer in these eyes and typically be very close to the correct prediction. The target was plano in this case, as shown in Figure 2. I would instead have targeted -0.50 D, which would have increased the IOL power by about 0.70 D. The three predictions would have averaged 21.72 D. I would likely (incorrectly in this case) have rounded down to 21.50 D for the IOL implant.

BRET L. FISHER, MD
Cataract surgery after hyperopic LASIK can be challenging. Although a spherical lens may offer an optical advantage in some cases, I have used aspheric IOLs, including aspheric multifocal IOLs, in these patients for many years without problems. In published research of IOL implantation after LASIK, two-thirds of the study group had a history of myopic LASIK, and one-third had a history of hyperopic LASIK.1 Although the latter subset was smaller in number, there appeared to be no overall difference in the distance visual acuities achieved using a low-add aspheric multifocal IOL compared to a monofocal IOL in eyes that had undergone hyperopic LASIK.
In this case, the IOL power calculation obtained with intraoperative aberrometry was used and turned out to be correct. In terms of calculating IOL power, I have found that my personally optimized results with the ORA System give me the highest level of accuracy in patients who have undergone laser refractive surgery. In the event of a conflict with my preoperative estimate based on the Barrett True K formula, I tend to trust the ORA System’s recommendation if I believe that the intraoperative conditions for measurement are good and the data used for the calculation are therefore also good.

UDAY DEVGAN, MD, FACS, FRCS
The tough part in these eyes is the small central corneal optical zone created by hyperopic LASIK. This patient’s right eye also has significant negative corneal spherical aberration from laser refractive surgery, so the choice of a monofocal IOL with positive spherical aberration was excellent because it resulted in an optical system with a total spherical aberration of close to zero. Could this patient have done well with an aspheric IOL that induced negative spherical aberration? In my opinion, yes. I find that lower-order aberrations (sphere and cylinder) are more important than spherical aberration (Z4,0 term) and that going from -0.138 to -0.300 D of spherical aberration does not have a tremendous effect on the visual outcome.

WHAT I DID: KARL G. STONECIPHER, MD
No additional intervention was indicated for this patient.
1. Fisher B, Potvin R. Clinical outcomes with distance-dominant multifocal and monofocal intraocular lenses in post-LASIK cataract surgery planned using an intraoperative aberrometer. Clin Exp Ophthalmol. 2018;46(6):630-636.




