Over the past century, the management of Fuchs dystrophy has evolved alongside techniques for corneal surgery. Successful transplantation of a full-thickness cornea for Fuchs dystrophy was first described in 1952, and for decades definitive treatment of Fuchs dystrophy always included transplantation of the corneal endothelium.1 Surgical options were limited to penetrating keratoplasty until endothelial keratoplasty techniques were introduced, beginning in 1998.2
In recent years, an exciting innovation in the management of Fuchs dystrophy has been the advent of Descemet stripping only (DSO), also known as descemetorhexis without endothelial keratoplasty (DWEK). All previous surgical options had the disadvantages of requiring long-term immunosuppressive eye drops and the use of donor corneal tissue. DSO circumvents both of these limitations while maintaining equivalent visual outcomes in a specific patient population.
The basic concept behind DSO is that large guttae are barriers to endothelial cell migration. When the central Descemet membrane (DM) is removed, peripheral endothelial cells can successfully migrate over this area, form a monolayer, and reestablish pump function. This cascade of events results in a clear, compact central cornea.
TECHNIQUE
DSO can be performed as a primary procedure or combined with cataract surgery. A 4-mm circular zone centered on the visual axis is marked on the corneal epithelium. After the anterior chamber is filled with an OVD, 1 mm of DM is scored with a reverse Sinskey hook, and forceps are then used to strip DM to encompass the marked area.
It is important to perform gentle peeling of DM (ie, descemetorhexis) and to avoid engaging the stroma. The diseased DM is then entirely removed, and the remainder is left to heal on its own (Figure 1). All OVD is removed. Neither a peripheral iridotomy nor an air bubble is required.
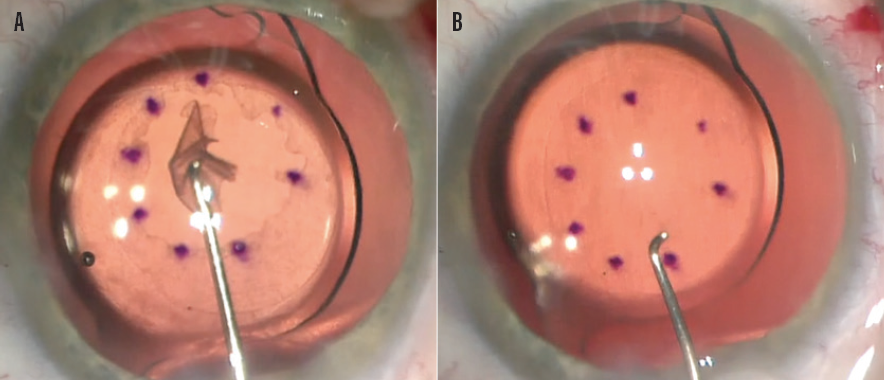
Figure 1. DM is completely separated from the corneal stroma (A). Completed descemetorhexis with DM removed (B).
ADVANTAGES
There are several advantages of DSO compared to Descemet membrane endothelial keratoplasty (DMEK) or Descemet-stripping automated endothelial keratoplasty (DSAEK).
No. 1: A donor cornea is not required. The most notable difference is eliminating the requirement for a donor cornea. Fuchs dystrophy accounts for about 50% of endothelial keratoplasty procedures performed in the United States.3 With DSO, some of these cases can be managed without performing a transplant at all.
No. 2: It can be combined with phacoemulsification for cataract extraction.This saves the patient the costs, inconvenience, and risks associated with a second surgical procedure that requires anesthesia.
No. 3: With no transplanted tissue, long-term immunosuppression is not required. Other current options, like DMEK and DSAEK, require a minimum of 1 year of daily steroid eye drops and regular monitoring for possible rejection.
No. 4: It virtually eliminates adverse events. Although major adverse events from endothelial keratoplasty that lead to an additional procedure are rare, some patients require re-bubbling if the donor tissue is nonadherent, and others develop increased IOP due to the steroid eye drops, which requires treatment with glaucoma medication.
No. 5: Visual outcomes are similar to those after other endothelial keratoplasty techniques. Most important, there is no significant difference in final visual outcomes between DSO and DMEK, as demonstrated in our 2018 study.4 Although the long-term durability of visual outcomes after endothelial keratoplasty is well proven, this is still in question in DSO. The uncertainty is mitigated by reports that endothelial keratoplasty can still be successfully performed after failure of DSO.5-7
DISADVANTAGES
Unlike traditional endothelial keratoplasty, DSO has not been shown to successfully treat severe Fuchs dystrophy with significant guttae out to the peripheral cornea or corneal edema caused by other etiologies.
Additionally, corneal clearance after DSO takes significantly longer than after endothelial keratoplasty. In our study, we found that it took, on average, 7 weeks for DSO patients to achieve better than 20/40 vision compared with just 2 weeks for DMEK patients (Figure 2).4 In the early postoperative period, in fact, patients experienced initial worsening of vision after DSO due to severe central corneal edema before eventual recovery. Therefore, it is important to prepare patients for a prolonged postoperative course.
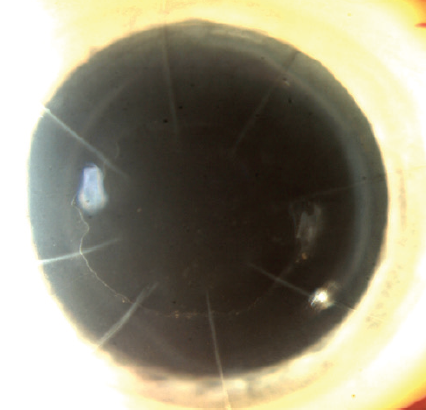
Figure 2. Eight weeks after DSO in a patient with Fuchs dystrophy and previous radial keratotomy, visual acuity in the operated eye was 20/25.
If stromal fibers are inadvertently engaged during DSO, there is a risk of scar formation in the area of engagement and possible delay of endothelial migration as a result.
PATIENT SELECTION AND POSTOPERATIVE MEDICATIONS
Patient selection is paramount for DSO. The ideal patient should have excellent vision in the fellow eye, mild to moderate Fuchs dystrophy with guttae limited to the central cornea, a relatively clear peripheral cornea with a minimum endothelial cell density (ECD) of 1,000 mm,2,8 and a relaxed personality.
It is important to thoroughly counsel patients to expect poor vision in the immediate postoperative period and a prolonged recovery. This procedure should be avoided in patients who may be intolerant of any deterioration of vision in the surgical eye.
Patients who have developed steroid-induced glaucoma in the their first eye after endothelial keratoplasty are particularly good candidates for DSO in their fellow eye if the conditions described here are met. As stated previously, with DSO there is no need for long-term steroid use because no donor corneal tissue is inserted into the eye.
In general, patients receive the same postoperative drops after DSO as they would after cataract surgery. A recent study by Macsai and Shiloach showed that DSO patients treated with a topical Rho kinase inhibitor (ripasudil 0.4%, Glanatec, Kowa) four times daily for 2 months recovered vision more quickly than an observation group that underwent DSO without topical ripasudil.9 The authors also found that the ripasudil group had a higher central ECD at 1 year and less loss of peripheral ECD.
Watch It Now
Dr. Dhaliwal speaks with Neda Shamie, MD, about results with DSO.
SUMMARY
DSO is an alternative to DMEK in patients with Fuchs dystrophy who have central guttae, mild to moderate corneal edema, a peripheral ECD greater that 1,000 mm2, functional vision in the fellow eye, and a relaxed personality. It is important to counsel patients carefully on a prolonged postoperative recovery period and the possibility of requiring endothelial keratoplasty as a rescue procedure if corneal edema does not clear.
Careful surgical technique with proper centration of the descemetorhexis over the pupil is important, as is the avoidance of engaging stromal fibers. So far in early experience with DSO, use of a topical Rho kinase inhibitor in the postoperative period has been shown to improve outcomes.9
1. Stocker FW. Successful corneal graft in a case of endothelial and epithelial dystrophy. Am J Ophthalmol. 1952;35(3):349-352.
2. Melles GR, Eggink FA, Lander F, et al. A surgical technique for posterior lamellar keratoplasty. Cornea. 1998;17(6):618-626.
3. Eye Bank Association of America. 2016 Eye Banking Statistical Report. 2017. Available at: http://restoresight.org/wp-content/uploads/2017/04/2016_Statistical_Report-Final-040717.pdf. Accessed May 14, 2019.
4. Huang MJ, Kane S, Dhaliwal DK. Descemetorhexis without endothelial keratoplasty versus DMEK for treatment of Fuchs endothelial corneal dystrophy. Cornea. 2018;37(12):1479-1483.
5. Bleyen I, Saelens IEY, van Dooren BTH, van Rij G. Spontaneous corneal clearing after Descemet’s stripping. Ophthalmology. 2013;120(1):215.
6. Koenig SB. Planned descemetorhexis without endothelial keratoplasty in eyes with Fuchs corneal endothelial dystrophy. Cornea. 2015;34(9):1149-1151.
7. Borkar DS, Veldman P, Colby KA. Treatment of Fuchs endothelial dystrophy by Descemet stripping without endothelial keratoplasty. Cornea. 2016;35(10):1267-1273.
8. Moloney G, Petsoglou C, Kerdraon Y, et al. Descemetorhexis without grafting for Fuchs endothelial dystrophy–supplementation with topical ripasudil. Cornea. 2017;36(6):642-648.
9. Macsai M, Shiloach M. Use of topical rho kinase inhibitors in the treatment of Fuchs dystrophy after Descement stripping only. Cornea. 2019;38(5):529-534.






