By Uday Devgan, MD, FRCS

Get a group of patients who value emmetropia so much that they have all paid thousands of dollars out of pocket in order to achieve it, and break the news to them that the IOL power calculations for their upcoming cataract surgery are less accurate. That is a recipe for patients’ disappointment. Prior corneal refractive surgery, including radial keratotomy (RK), PRK, conductive keratoplasty, and LASIK, makes it less likely that cataract surgeons will achieve the desired postoperative target unless they adjust their calculations.
VARIABLES
Many variables affect IOL power calculations. The three most important ones are corneal power, axial length, and the effective lens position (ELP) of the IOL after surgery. In virgin eyes, surgeons can accurately measure corneal power and axial length, and newer methods of IOL power calculation can give a high degree of accuracy, with 80% or more patients within 0.50 D of the intended target. During initial testing with the artificial intelligence algorithms of the Ladas Super Formula, more than 90% of patients achieved this level of precision (personal correspondence with John Ladas, MD, PhD).
With eyes that have undergone corneal refractive surgery, however, it is difficult to measure the cornea exactly, and the ELP calculation is more challenging (Figure 1).
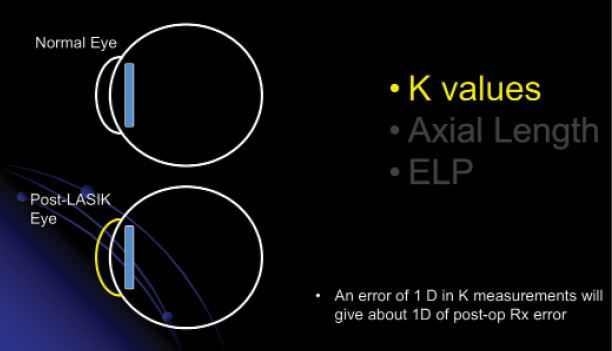
Figure 1. The primary source of error in IOL power calculations in eyes with a history of corneal refractive surgery is the measurement of the corneal power (K values). The axial length and ELP are small sources of error.
VERGENCE AND ELP
Any IOL calculation involves two steps: determining the vergence and then predicting the final ELP (Figure 2). For the simpler third-generation formulae (Holladay 1, SRK/T, Hoffer Q), which use just keratometric (K) power and axial length as input variables, the ELP is based largely on the measured K value. For a virgin eye, a K reading of 43.5 is average, so an average anterior chamber depth and ELP are calculated. After myopic LASIK, however, the same formula will assume that, because the K value is now 38, the anterior chamber is shallower. Surgeons know that the LASIK procedure only changed the K value, not the anterior chamber depth. They should therefore separate the vergence calculation from the ELP determination by using techniques described by Aramberri in his double-K approach, where the measured K reading is used for the vergence and then a fixed K value of 43.5 (or the actual pre-LASIK K value) is used for the ELP part of the equation.1

Figure 2. The two components of an IOL power calculation are the vergence calculation (yellow graph) and the determination of the ELP (blue graph). Together, these eloquently demonstrate the IOL power, as shown in a 3-D Ladas graph (green graph).
TYPE OF CORNEAL REFRACTIVE SURGERY
Some forms of refractive surgery such as RK change both the anterior and posterior surfaces of the cornea, whereas other procedures such as LASIK and PRK only alter the anterior corneal curvature. Because both the anterior and posterior corneal power have an effect on the refractive outcome, knowing which surfaces have been altered is helpful in lens calculations.
Myopic LASIK and PRK produce a larger optical zone, whereas hyperopic LASIK and RK tend to yield smaller optical zones of 3 mm or less. This makes measuring the central cornea more critical, and surgeons should be careful when using older keratometers, which tend to measure at about a 3-mm ring from the central cornea.
The refraction and visual acuity after some procedures such as conductive keratoplasty, RK, and even hyperopic LASIK can change or drift over time. After RK, it can even change daily in a diurnal pattern, making the timing of biometry essential for coordinating with the patient’s daily activities.
TYPE OF POSTOPERATIVE REFRACTIVE SURPRISE
The type of error in an eye with a prior myopic treatment—be it LASIK, PRK, or RK—is an overestimation of the central corneal power. For example, devices will read a central cornea at 38.00 D of power, when the actual central power may be closer to 36.00 D. If the erroneous value is used in standard IOL power calculations, the surgeon will end up with an IOL power that is too low, and instead of achieving the desired emmetropic result, the patient will be uncomfortably hyperopic after cataract surgery.
The principal error in an eye with a prior hyperopic treatment is an underestimation of the central corneal power. In another example, machines may read a central cornea at 46.00 D of power, but the actual central corneal power will be 48.00 D. Using the erroneous value in standard IOL power calculations, the surgeon will end up with an IOL power that is too high, and instead of achieving a plano result, the patient will have a myopic surprise after cataract surgery.
RECOMMENDATIONS FOR IOL CALCULATIONS
There are more than 20 published ways of performing IOL power calculations in eyes with a history of corneal refractive surgery. That likely means that none of them is truly correct and, certainly, that none is perfectly accurate.
Most surgeons use an online calculator such as the one provided by the American Society of Cataract and Refractive Surgery, which performs multiple calculations based on all of the available data entered. It then gives multiple answers, an average of all the answers, and a range of IOL powers. When using this approach, the golden rule is to choose the higher-powered IOL so as to err on the side of slight myopia postoperatively. Myopia is advantageous for achieving better intermediate and near vision, whereas residual hyperopia compromises vision at all distances.
Intraoperative aberrometers, which can measure the aphakic eye after cataract removal but prior to the IOL’s insertion, may help to increase accuracy.
THE FUTURE
My colleagues and I have found that a 3-D graphing and analysis of many of the described methods of IOL power determination show that certain formulae perform better with specific subsets of eyes. We have also crowd-sourced data on many thousands of eyes to refine the complex 3-D graphical shape, and now, we are implementing a neural net machine learning methodology to increase accuracy. Combining this new approach of artificial intelligence with intraoperative aberrometers and guidance systems may end up being the ultimate answer for these tough cases.
At present, however, there is no perfect way of determining IOL power for all eyes, and this is particularly true for eyes that have undergone corneal refractive surgery. Until newer methods become widely available, the best advice is to use all available data with multiple formulae, choose the higher IOL power to err on the side of residual myopia, and set realistic expectations for patients.
1. Aramberri J. Intraocular lens power calculation after corneal refractive surgery: double K method. J Cataract Refract Surg. 2003;29(11):2063-2068.
By Mitchell P. Weikert, MD

The success of modern cataract and refractive surgery has created high expectations. Achieving success in cataract surgery depends upon accurate ocular biometry and IOL power calculations. The process can be quite challenging in patients who have undergone corneal refractive surgery (CRS) such as LASIK, PRK, or radial keratotomy. The two greatest obstacles to hitting refractive targets in these patients are the accurate measurement of corneal power and the prediction of the postoperative effective lens position (ELP).
AT A GLANCE
- Prior corneal refractive surgery makes it less likely that cataract surgeons will achieve the desired postoperative refractive target unless they adjust their calculations.
- The two greatest obstacles to hitting refractive targets in these patients are the accurate measurement of corneal power and the prediction of the postoperative effective lens position
- The authors share advice on how to optimize results.
MEASURING CORNEAL POWER
Typical methods for determining corneal refractive power (manual keratometry, automated keratometry, and topography) measure the radius of curvature of the anterior surface and convert this number to total power using a reduced corneal refractive index. Decreasing the refractive index from 1.376 to 1.3375 (in the United States) or 1.3320 (in Europe and elsewhere) compensates for the negative power of the posterior corneal surface. These adjusted refractive indices are based on a fixed ratio of anterior-to-posterior curvature (the Gulstrand ratio).1 Ablative CRS alters this ratio and introduces error into power estimations that are based solely on anterior curvature. In addition, these methods assume a spherical cornea and only measure small paracentral zones, which may not accurately reflect the cumulative refractive effect across the ablation zone of LASIK/PRK or the optical zone of radial keratotomy.
PREDICTING THE ELP
The ELP represents the effective refractive plane of the IOL after cataract surgery. Although IOL calculation formulas differ in their methods of estimating ELP, most use corneal power at some point in this process. The alterations in corneal power produced by CRS may therefore lead to incorrect estimates of the IOL’s location and its recommended power. For example, a reduced corneal power after myopic ablation may cause many formulas to incorrectly assume that the anterior chamber is shallower and the IOL sits more anteriorly. This will lead to an underestimation of IOL power and a hyperopic result.
In an effort to overcome this problem, Aramberri proposed the Double-K method, which substitutes the pre-CRS average keratometry (K) value for the ELP calculation while using the current, post-CRS K reading for the IOL power calculation in standard vergence-based formulas.2 The technique, which had also been applied by Holladay, was shown to increase the accuracy of IOL power calculation in post-CRS eyes.
REMEDIES
Formulas
Many methods have been developed in an attempt to overcome the aforementioned challenges and increase the accuracy of IOL calculations. Some incorporate the refractive change achieved by CRS (ΔMRCRS) into theoretical models that try to compensate for its effect at the cornea or IOL plane. Others employ a regression approach, using the results of prior cataract surgeries to modify the current, post-CRS average K or to adjust the IOL power calculated from the current Ks. Still others use direct measurement of the anterior curvature, posterior curvature, and corneal thickness to calculate the “true” corneal power. These methods are usually grouped according to the data that they require:
1. Those that use both the pre-CRS K readings and ΔMRCRS
2. Those that use the ΔMRCRS only
3. Those that only use current corneal data
Many of the methods are device specific, having been derived using corneal readings from particular topographers or tomographers.
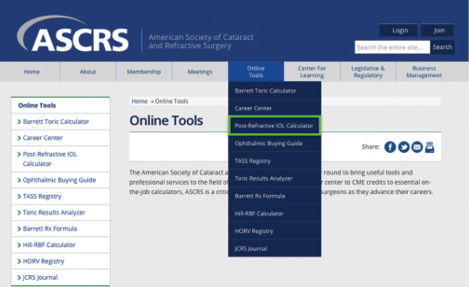
Figure. Selecting the ASCRS Post-Refractive IOL Calculator.
Although it was initially burdensome to perform these individual calculations for each patient, the task has been greatly simplified with the ASCRS Post-Refractive IOL calculator (https://www.ascrs.org). This open-access, web-based tool has assembled a large variety of these methods into a single, easy-to-use location (Figure). The website initially included methods from all three of the aforementioned groups, but the list was reduced to include those from groups 2 and 3 only. The formulas that employed both the pre-CRS K readings and ΔMRCRS, such as the clinical history and corneal bypass methods, were found to be significantly less accurate and were thus removed.3 (See Breakdown of the ASCRS Post-Refractive IOL Calculator.)

After entering all of the biometric, topographic, tomographic, and IOL data at one’s disposal, the ASCRS calculator will provide the recommended IOL power for each method as well as the minimum, maximum, and average IOL powers. These results can often span a considerable range, leading to confusion over which choice is optimal. Of the formulas in group 2, my colleagues and I have found the Masket, Modified Masket, and Barrett True-K formulas to perform the best, with median absolute IOL prediction errors of 0.32, 0.30, and 0.33 D, respectively. We found that the OCT, Haigis-L, and Barrett True-K no history formulas performed the best in group 3, with median absolute IOL prediction errors of 0.35, 0.39, and 0.42 D, respectively. We also look for agreement between different formulas and recommend that surgeons track their personal results to determine which formulas work best for them.
Intraoperative Aberrometry
Intraoperative aberrometry can also be of benefit in post-CRS eyes.4 This device measures the total refractive error after cataract removal and calculates a recommended IOL power to achieve the programmed refractive target. Although the device’s calculations are still vergence based and subject to errors associated with ELP estimation, the aberrometer draws on a large database of user-entered, postoperative refractive data, which it uses to refine its recommendations. Intraoperative aberrometry has been shown to produce results consistent with those of the ASCRS calculator.5
CONCLUSION
The methods described herein have substantially improved the refractive results of cataract surgery in post-CRS eyes. That said, post-CRS IOL calculations still show significant variability when compared to similar surgery in eyes with virgin corneas. Additional procedures may be required to reach outcomes that satisfy this challenging group of patients. For this reason, my colleagues and I recommend addressing these issues during the informed consent process. We also recommend considering patients’ suitability for additional refractive procedures so as to properly set their expectations.
1. Olsen T. Calculation of intraocular lens power: a review. Acta Ophthalmol Scand. 2007;85(5):472-485.
2. Aramberri J. Intraocular lens power calculation after corneal refractive surgery: double-K method. J Cataract Refract Surg. 2003;29(11):2063-2068.
3. Wang L, Hill WE, Koch DD. Evaluation of IOL power prediction methods using the American Society of Cataract and Refractive Surgeons post-keratorefractive IOL power calculator. J Cataract Refract Surg. 2010;36(9):1466-1473.
4. Ianchulev T, Hoffer KJ, Yoo SH, et al. Intraoperative refractive biometry for predicting intraocular lens power calculation after prior myopic refractive surgery. Ophthalmology. 2014;121(1):56-60.
5. Fram NR, Masket S, Wang L. Comparison of intraoperative aberrometry, OCT-based IOL formula, Haigis-L, and Masket formulae for IOL power calculation after laser vision correction. Ophthalmology. 2015;122(6):1096-1101.