A 68-year-old woman is referred by her retina doctor for cataract surgery on the right eye. The patient underwent a pars plana vitrectomy (PPV) and membrane peel 6 weeks prior to presentation. She complains of poor vision in her right eye and can only see her hand if she holds it very close to that eye.
On examination, the BCVA is count fingers at 3 feet in the right eye, with a 2+ nuclear sclerotic, 2+ cortical, and 3+ posterior subcapsular cataract (PSC) and a possible defect in the posterior capsule centrally (Figure). A vitreous hemorrhage is present inferiorly. Macular edema is evident on examination and with optical coherence tomography (OCT). The fellow eye has a BCVA of 20/20, and the examination is unremarkable.
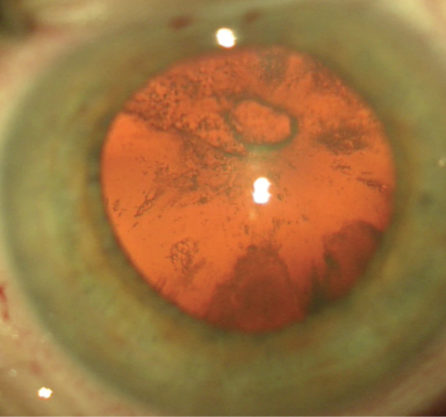
Figure. An apparent capsular defect is visible with red reflex in the right eye.
The documented immediate postoperative note from the retinal surgery says that the “lens developed increasing PSC opacity during [the] case.” On discussion, the retina surgeon expresses suspicion that there may be a defect in the capsule or damage caused by flow from the infusion during the PPV.
The surgical plan includes waiting 1 month for capsular fibrosis around the apparent defect and a preoperative slit-lamp examination on the day of surgery to look for evidence of a true break (white cataract formation) versus trauma in order to decide on the best surgical approach.
Would you obtain any other preoperative testing? What would your surgical approach be? What contingency plans would you make? If fragments of the cataract fall through a posterior capsular defect, what will your next step be?
—Case prepared by Cathleen M. McCabe, MD.

ZAINA AL-MOHTASEB, MD
The description of increasing PSC opacity during the vitrectomy is worrisome, suggesting a violation of the posterior capsule. Although ultrasound biomicroscopy preoperatively would help me to evaluate the integrity of the posterior capsule, during surgery, I would still proceed as if this were a posterior polar cataract. I would perform a small capsulorhexis in case I needed to capture the lens optic. Then, I would carefully perform hydrodelineation without hydrodissection to prevent hydrostatic pressure from extending a posterior capsular tear.
I would use a Nagahara chopper to chop the endonucleus into pieces without rotating the nucleus. Before removing any instrument, I would inject an ophthalmic viscosurgical device (OVD) to maintain the anterior chamber (AC) so as to prevent its collapse. Using a dispersive OVD, I would viscodissect the epinucleus and cortex (especially in the area of concern to tamponade the vitreous) and then remove them with bimanual irrigation and aspiration. I would start in the periphery and move toward the area of concern. Even if the capsule proved to be intact, I would avoid polishing it.
If, despite my best efforts, fragments of the cataract fell through a posterior capsular defect, I would avoid reaching deep into the vitreous cavity to retrieve them, because doing so might result in a retinal tear. Instead, I would perform a bimanual anterior vitrectomy and then secure the IOL using the optic capture technique.
After bringing down the pupil with acetylcholine chloride, I would inject triamcinolone into the anterior chamber to identify strands of vitreous. Most importantly, I would make sure the macular edema was treated appropriately prior to and after surgery with help from my retina colleague.

PAUL C. KANG, MD
Occasionally, anterior segment OCT can detect an irregularity in the contour of the posterior capsule to confirm the suspicion of a defect. Nevertheless, surgical planning should anticipate a compromised posterior capsule in an already vitrectomized eye. This would include appropriate informed consent for the possible need for future surgery.
Currently, I use a femtosecond laser for most of my cataract cases. I think the benefit here would be that the capsulorhexis and lens softening could occur with less stress on the lens, less lens manipulation, and less phaco energy. With the Catalys Precision Laser System (Abbott, now Johnson & Johnson Vision), I can control the depth of the lens treatment. In this case, I would extend the laser treatment to only two-thirds the depth of the lens and would stay far away from the posterior capsule.
Next, I would carefully hydrodissect in four quadrants but would stop the fluid wave short of the compromised area of the capsule. I would also aggressively hydrodelineate the lens, with a goal of levitating its nucleus and leaving behind an epinuclear shell. I would use a dispersive OVD to assist in the levitation of the lens and creation of space. Phacoemulsification would then be possible at the iris plane and anterior chamber. Because the laser would have presoftened the lens, it could likely be removed with high vacuum and very little phaco energy. I could then perform viscodissection of the epinuclear shell. Epinuclear and cortical cleanup could be done with irrigation and aspiration and a low bottle height.
The defect in the capsular bag appears to be round, with fibrosis at the edges. In such situations, a one-piece IOL can typically be inserted into the capsular bag. If not, I would place a three-piece IOL in the sulcus.
If, despite my best efforts, lens fragments fell to the posterior pole, I would proceed with caution. In an already vitrectomized eye, the pieces are likely to fall deep into the posterior pole. For this reason, it might be better for a retina surgeon to retrieve them. If the pieces were accessible, an anterior vitrectomy could be performed.

KEVIN M. MILLER, MD
The historical details and clinical findings in this case are consistent with an iatrogenic break in the posterior capsule. Because there was a previous vitrectomy, only a small amount of anterior cortical gel should remain.
I would approach the cataract exactly as I would a posterior polar cataract. I would prefer to use a femtosecond laser to create the capsulorhexis and fragment the nucleus. I would make sure there was at least a millimeter of untreated posterior epinucleus to keep gas bubbles from accumulating near the break.
As with a posterior polar cataract, I would avoid hydrodissection but would be generous with hydrodelineation, performed using an inside-out technique after nucleus removal. I would remove the posterior plate covering the hole as the very last step. If the hole looked irregular or began to expand, I would try to convert it into a posterior capsulorhexis.
A small amount of gel will likely prolapse through the break, so I would be prepared to perform a limited anterior vitrectomy. With a stable tear or a well-performed posterior capsulorhexis, it should be possible to place an IOL within the capsular bag. If not, with a nicely centered and sized anterior capsulorhexis, it should be possible to place an IOL in the ciliary sulcus and to capture the optic posterior to the capsulorhexis.
In the unlikely event that lens fragments drop posteriorly, it would be nice to have a retina specialist nearby, but I do not think one would need to be in the room.

R. BRUCE WALLACE III, MD
Considering the nature of the retinal surgery, the macular edema may reduce the possibility of any significant visual improvement in this case. The first steps are to assess the patient’s Lotmar potential visual acuity and to discuss in detail with her and her family the questionable cataract surgical prognosis based on her history of retinal disease and the probability of a tear in the posterior capsule. If macular disease continues, a further delay of surgical treatment may be warranted.
It would be advisable to wait 1 month and then repeat the macular OCT as well as the Lotmar visual acuity test. One avenue for treatment would be to have the patient return for a pars plana lensectomy by the retina surgeon, with a plan for an anterior chamber or sutured IOL later.
For the anterior segment specialist, surgical treatment will be a challenge because of the probable posterior capsular tear that likely will not have healed or even improved in the month before surgery. The tear will probably expand, even with low-flow, low-pressure phacoemulsification. One avenue to consider is a can-opener capsulotomy and planned extracapsular nuclear extraction to reduce the chance of a balanced salt solution-aqueous misdirection syndrome, which will be more likely with the additional fluid necessary for phacoemulsification. Without expansion of the capsular tear, in-the-bag placement of a posterior chamber IOL could be successful, but if the surgeon harbors any doubt, ciliary sulcus placement would be safer.
If lens fragments of significance flow posteriorly, a follow-up examination and treatment by the retina specialist should be considered.

ROBERT J. WEINSTOCK, MD
I would assume significant damage to the posterior capsule’s integrity and likely enlargement of the site of trauma during cataract removal, with the potential loss of nuclear and cortical material into the vitreous cavity. I would therefore perform a retrobulbar block and use the Stellaris PC Vision Enhancement System (Bausch + Lomb), which has 25-gauge vitrectomy capability. A laser could be used to make the capsulotomy, but I typically avoid laser fragmentation unless there is very dense nuclear sclerosis, because it obscures the red reflex and inhibits visualization of the potential hole in the capsule.
I usually start with a superior (for a left eye) or an inferior (for a right eye) transconjunctival 25-gauge PPV trocar system with port creation and a plug placed in the port to prevent vitreous from escaping. It is very important to perform these steps first, while the eye is still sealed with adequate pressure and resistance during the trocar’s placement. Then, I proceed with typical clear corneal cataract extraction, but I am extremely gentle during hydrodelineation and hydrodissection. I carefully watch the fluid wave to see if it passes in front of the potential defect in the posterior capsule or stops at the site of trauma, with fluid entering the posterior segment. If a complete fluid wave passes in front of the suspected hole in the posterior capsule, I have a lot more confidence that I will be able to preserve the posterior capsule and remove the cataract completely without any loss of material into the posterior segment.
Usually, I start with a high vacuum setting. I try to impale the nucleus and bring it into the supracapsular space so as to keep it trapped in the anterior chamber or at the level of the capsulorhexis and avoid moving it posteriorly through the potential capsular defect. I attempt to leave the cortex in place so that it can temporarily block vitreous or fluid movement between that potential hole and the anterior segment. If during the case I observe an enlargement of the posterior capsular hole or the prolapse of cataract material through it, I stop, perform a viscoelastic-balanced salt solution exchange in the anterior chamber, and remove the phaco handpiece. Because I perform bimanual microincisional cataract surgery, I then use the irrigating chopper through one of the incisions in the anterior chamber as a chamber maintainer.
With the 25-gauge vitrectomy instrument through the pars plana port, I perform an anterior vitrectomy and attempt to capture any cortical or nuclear material that is in the anterior vitreous space or starting to move through the hole in the posterior capsule. After a thorough anterior vitrectomy, I often place a retinal lens on the eye to visualize how much material has escaped into the vitreous cavity. If visibility is good, I will sometimes go a little deeper and remove any residual vitreous or cortical material that is floating in the midvitreous area. Afterward, I usually remove the vitrectomy instrument and return to conventional irrigation and aspiration in the anterior segment through the clear corneal incisions. I attempt to clean up the cortex in the capsule and preserve as much capsule as possible.
Next, I stain the anterior and posterior chambers with triamcinolone acetonide (Kenalog; Bristol-Myers Squibb) to ensure that no vitreous is presenting through the pupillary axis. Then, I usually implant an LI61AO monofocal lens (Bausch + Lomb) in the sulcus. After instilling acetylcholine (Miochol-E; Bausch + Lomb) and ensuring that there is no vitreous in the anterior chamber, I suture the main incision. Next, I inject triamcinolone acetonide and an antibiotic into the vitreous via the 25-gauge port and then remove the port.

CATHLEEN M. McCABE, MD
WHAT I DID:
Cataract surgery was delayed until 1 month after presentation to allow for fibrosis around the apparent posterior capsular defect. I was surprised that no further opacification or whitening of the cataract occurred and considered that the injury might not be full thickness after all.
After filling the anterior chamber with a dispersive OVD, I created a capsulorhexis small enough for optic capture (see video below). I carefully performed hydrodelineation and removed the nucleus with a chopping technique. As I was evacuating the last fragments, a tear in the posterior capsule became visible, and a nuclear fragment was lost through it into the vitreous.
I performed a bimanual anterior vitrectomy through a sclerotomy 3 mm posterior to the limbus. Then, I placed the vitrector through a paracentesis in order to remove the remaining cortex. A very useful tool in these cases is a disposable wide-view vitrectomy lens (Dutch Ophthalmic USA), which I used to view the posterior segment and the lost lens fragment. After removing vitreous adjacent to the lens fragment, I engaged and removed it with the vitrector. I left a small, thin epinuclear shell that could not be safely elevated from the retina. Next, I implanted a three-piece lens (LI61AO) in the sulcus with optic capture.
See how Cathleen M. McCabe, MD, managed this case.
The pupil was constricted with carbachol intraocular solution (Miostat; Alcon). I instilled triamcinolone/moxifloxacin solution (Tri-Moxi; Imprimis Pharmaceuticals) in the anterior chamber to identify any vitreous strands and provide a depot of drug. Next, I sutured the sclerotomy and securely closed all corneal incisions with ReSure Sealant (Ocular Therapeutix).
One day postoperatively, a small epinuclear fragment was visible inferiorly, which the patient’s retina surgeon recommended that we observe without surgical intervention. The patient has been using steroid drops, and 2 months after surgery, only a tiny remnant of the fragment can be seen. Her vision remains limited by an epiretinal membrane, with significant striae and distortion of the foveal contour, and by macular edema.
This case illustrates the importance of having a plan and the appropriate tools available for managing a damaged posterior capsule after posterior segment surgery. Disposable retinal lenses are useful for accessing the posterior segment and for limited retrieval of lost nuclear fragments. Safety is of the utmost concern, however, and it is better to leave a fragment behind than to risk injuring the retina. An examination with anterior segment OCT and/or ultrasound might have helped to confirm a full-thickness break in the posterior capsule prior to surgery.




