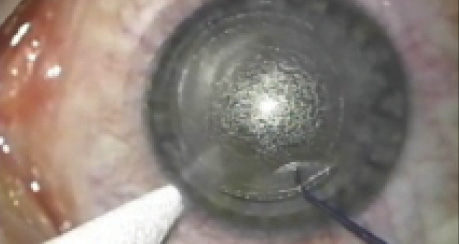
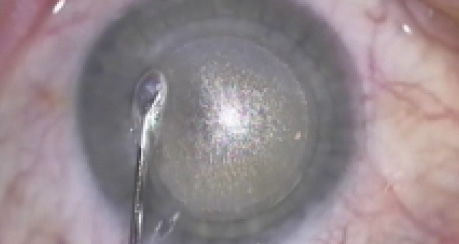
Small-incision lenticule extraction (SMILE; not available in the United States) is currently indicated (outside the United States) for the correction of myopia or myopic astigmatism of up to -10.00 D spherical equivalent or -5.00 D of astigmatism. Because the procedure currently cannot be used in hyperopic treatment, the immediate answer to the question posed in this headline is: “No, it won’t overtake LASIK.” With that said, a recently published report has shown promising initial results with hyperopic SMILE,1 and further research and clinical studies are being undertaken. Hence, this situation may well change in the near future.
AT A GLANCE
• The treatment of hyperopia with SMILE has been approached in two ways: (1) resection of a hyperopic- shaped lenticule that is thicker in the periphery and thinner in the center and (2) implantation of a myopic- cut lenticule following removal from another patient, donor cornea, or, depending on refractive error, the other eye of the same patient.
• SMILE has not overtaken LASIK as of yet, and the caveat is that surgeons must continue to watch this space.
The treatment of hyperopia with SMILE has been approached in two ways: (1) resection of a hyperopic-shaped lenticule that is thicker in the periphery and thinner in the center and (2) reimplantation of a myopic-cut lenticule following removal from another patient, donor cornea, or, depending on refractive error, the other eye of the same patient.2,3 We are currently performing animal studies to analyze the outcomes of both these approaches.
CHALLENGES IN HYPEROPIC TREATMENT
Compared with myopic correction, there are two considerable challenges in hyperopic SMILE treatment.
Challenge No. 1. The lenticule must be larger than in myopic treatment, with most of the correction occurring in the peripheral cornea. This may require a larger cone size, which is difficult in certain populations such as Chinese patients due to their small white-to-white measurements. Using an S-cone, I have found that most lenticules are created just at the limits of possibility for cone lenticule creation. Interestingly, however, lower dioptric hyperopic corrections (eg, 2.00 D) were easier to remove than myopic ones (eg, -2.00 D), as the lenticule periphery was thicker and, therefore, easier to delineate. The central lenticule thickness was set at a default of 30 µm.
Challenge No. 2. The firing sequence in lenticule creation is different in myopia and hyperopia. In myopic treatments, the refractive lenticule is created by initially forming the posterior lenticule plane from the periphery to the center, followed by the anterior plane from the center to periphery. In hyperopic lenticule creation, by contrast, the lenticule is created from an optical zone of 5.5 mm, whereby the posterior surface is first created from the periphery to the center, followed by an extension of the posterior plane (transition zone) into the periphery, followed by creation of the anterior plane from the center to the periphery, and then the sidecut. This complex photodisruption profile is necessary to achieve the desired refractive change in hyperopic treatments.
Although lenticule extraction in both 2.00 and 4.00 D treatments was straightforward, more work is needed to assess the refractive outcomes of this treatment with respect to wound healing, refractive accuracy, and long-term stability.
Reimplantation of a myopic-cut lenticule is an interesting approach that may avoid some of the challenges of the hyperopic SMILE approach outlined previously2,3; initial results have shown a good level of accuracy.3 Although the procedure itself is technically not difficult, a main concern is the refractive accuracy of correction following reimplantation of the lenticule. Fine-tuning of the result may require the use of an excimer laser (eg, LASIK or advanced surface ablation).
Other issues to be addressed before indications for lenticule reimplantation are expanded include determining the optimal method for lenticule storage4 and whether using donor cornea or lenticule from another patient (which is the most likely scenario) should require serology testing of the allograft donor. Another option would be to make the lenticule immunogenic by decellularization.
ARGUMENTS AGAINST LASIK
The vast majority of patients with refractive errors have myopia or myopic astigmatism. LASIK has been the dominant refractive procedure of choice in this group of patients for the past 20 years,5 and it is a highly successful procedure. Although patients’ satisfaction rates are high, and despite advances in flap creation with the use of femtosecond lasers and the development of faster excimer lasers with eye-tracking technology, certain limitations of the procedure have become apparent.
LASIK’s success has been propagated thanks to its painless visual recovery, which is achieved because tissue ablation is carried out intrastromally, under a flap. For any new procedure to overtake an existing one, it must, at a minimum, offer the benefits of the existing procedure and then improve on them. With respect to visual outcomes, multiple studies have shown equivalent visual outcomes in terms of safety, efficacy, and predictability between SMILE and LASIK.6,7 These results have been reproduced by several centers around the world.
The consistency of ReLEx SMILE (Carl Zeiss Meditec) results internationally, now approaching more than 120,000 cases, offers strong validation for the procedure. The creation of the flap in LASIK poses several issues such as the creation of a neurotrophic ocular surface and the biomechanical weakness induced by flap creation itself. The ability of the femtosecond laser to create a refractive lenticule that can be removed without the creation of a flap is a significant advance, and studies in both animal models and patients show better preservation of subbasal nerve density8 and faster nerve regeneration in SMILE than in LASIK.9 This has been further supported by clinical studies examining corneal nerve sensation, confirming faster neural recovery in SMILE than in LASIK.9
Preservation of the nerve plexus and the subsequent effect on the tear film may persuade many surgeons to switch from LASIK to SMILE. We previously examined ocular surface function following flap creation with the IntraLase (Abbott Medical Optics) and VisuMax (Carl Zeiss Meditec) femtosecond lasers in a randomized, controlled trial. Although all patients had a healthy ocular surface preoperatively, as assessed by tear breakup time and corneal sensation, and although some recovered to near-normal tear function by 3 months postoperatively, a subgroup of patients had no recovery of their corneal function by this timepoint.10 Because the ability to predict postoperative corneal function preoperatively is difficult, a procedure that preserves the subbasal nerve layer better and allows faster tear film recovery could eliminate this variability.
Patients also like the fact that frequency of topical artificial tear use is reduced postoperatively with SMILE in comparison with LASIK, and patients’ satisfaction has a significant impact on the refractive surgery experience.
The most feared complication for all LASIK surgeons is ectasia. Despite the development of better topography machines and advanced indices for keratoconus detection, post-LASIK ectasia still occurs. The option of an intrastromal procedure that may be more biomechanically stable than LASIK is an attractive proposition for ophthalmologists and also for patients. Good clinical devices to measure ocular biomechanics are currently lacking. The Ocular Response Analyzer (Reichert Technologies) and Corvis ST (Oculus) offer limited information, and clinical studies using these instruments have begun to show greater postoperative biomechanical strength in SMILE than LASIK in moderate to high myopia.11,12
If mathematical and finite element modeling analysis is supported by clinical information, many surgeons may consider this factor alone enough to switch from LASIK to SMILE. In fact, results with newer devices studying corneal biomechanics in more detail may provide evidence to expand the clinical range for SMILE.
CONCLUSION
Since we first started performing SMILE in 2010, we have seen a significant shift in patients’ perception of the procedure. Patients like the appeal of small-incision surgery, especially if we are able to maintain some of the strength of the cornea.
My response to the original question, “Will SMILE overtake LASIK?” is this: potentially yes. The caveat is that we must watch this space; it is only a matter of time.
1. Blum M, Kunert KS, Voßmerbäumer U, Sekundo W. Femtosecond lenticule extraction (ReLEx) for correction of hyperopia – first results. Graefes Arch Clin Exp Ophthalmol. 2013;251(1):349-355.
2. Riau AK, Angunawela RI, Chaurasia SS, et al. Reversible femtosecond laser assisted myopia correction: a non-human primate study of lenticule re-implantation after refractive lenticule extraction. PLoS One. 2013;8(6):e67058.
3. Ganesh S, Brar S, Rao PA. Cryopreservation of extracted corneal lenticules after small incision lenticule extraction for potential use in human subjects. Cornea. 2014;33:1355-1362.
4. Mohamed-Noriega K, Toh K-P, Poh R, et al. Cornea lenticule viability and structural integrity after refractive lenticule extraction (ReLEx) and cryopreservation. Mol Vis. 2011;17:3437-3449.
5. Yuen LH, Chan WK, Koh J, et al. A 10-year prospective audit of LASIK outcomes for myopia in 37,932 eyes at a single institution in Asia. Ophthalmology. 2010;117(6):1236-1244.e1.
6. Sekundo W, Kunert K, Russmann C, et al. First efficacy and safety study of femtosecond lenticule extraction for the correction of myopia: six month results. J Cataract Refract Surg. 2008;34:1513-1520.
7. Shah R, Shah S, Sengupta S. Results of small incision lenticule extraction: all-in-one femtosecond laser refractive surgery. J Cataract Refract Surg. 2011;37(1):127-137.
8. Mohamed-Noriega K, Riau AK, Lwin NC, et al. Early corneal nerve damage and recovery following small incision lenticule extraction (SMILE) and laser in situ keratomileusis (LASIK). Invest Ophthalmol Vis Sci. 2014;55(3):1823-1834.
9. Li M, Niu L, Qin B, et al. Confocal comparison of cornea reinnervation after small incision lenticule extraction (SMILE) and femtosecond laser in situ keratomileusis (FS-LASIK). PLoS One. 2013;8:e81435.
10. Petznick A, Chew A, Hall RC, et al. Comparison of corneal sensitivity, tear function and corneal staining following laser in situ keratomileusis with two femtosecond laser platforms. Clin Ophthalmol. 2013;7:591-598.
11. Roy AS, Dupps WJ Jr, Roberts CJ. Comparison of biomechanical effects of small-incision lenticule extraction and laser in situ keratomileusis: finite-element analysis. J Cataract Refract Surg. 2014;40(6):971-980.
12. Wu D, Wang Y, Zhang L, Wei S, Tang X. Corneal biomechanical effects: small-incision lenticule extraction versus femtosecond laser-assisted laser in situ keratomileusis. J Cataract Refract Surg. 2014;40(6):954-962.
Jodhbir S. Mehta, FRCS, FRCOphth
• associate professor of ophthalmology and head of corneal research at the Singapore Eye Research Institute
• jodmehta@gmail.com
• financial disclosure: consultant to Carl Zeiss Meditec and Ziemer; has a patent/part ownership and royalty agreement with Lenticor