
In this month’s installment of “Residents and Fellows,” Drs. Pitcher and Ho review the basics of the most common systemic disease we see in our ophthalmology practice: diabetes. This is a great primer on the epidemiology, pertinent history and examination findings, billing nuances, patient education and counseling considerations, and the impact on our day-to-day practice.
—Section Editor Sumit “Sam” Garg, MD


According to the American Diabetes Association, nearly 10% of Americans are diabetic, and approximately 1.4 million are newly diagnosed with the disease each year. Among diabetic individuals aged 40 years or older, 28.5% have some degree of diabetic retinopathy, the leading cause of visual impairment in adults of working age.1
Diabetes alone results in more than $176 billion in direct medical costs annually.1 The detection and appropriate treatment of diabetic retinopathy reduce blindness by 52%, resulting in systemic gains of productivity that far outweigh the cost of care.2 These societal implications represent an enormous responsibility for ophthalmologists.
The essence of quality care is a strong partnership between patient and physician. In many cases, a diabetic patient may see his or her ophthalmologist more frequently than any other health care professional. Each office visit offers a tremendous educational opportunity.
A conversation with a physician is often one of the strongest motivators of patients’ adherence to therapy. This is especially true for eye care providers, because vision loss is one of the most feared complications of diabetes. Nonmydriatic photoscreening and telemedicine are gaining traction, but as helpful as these modalities may be, removing direct contact with the ophthalmologist eliminates a beneficial relationship.
A FEW SIMPLE QUESTIONS
By asking a few simple questions, the ophthalmologist can have a huge impact on patients’ quality of life.
Are You Type 1 or Type 2?
People with type 1 diabetes should have annual screenings beginning 5 years after the onset of the disease. The development of vision-threatening diabetic retinopathy may begin as soon as 6 years after the onset of the disease, but it is rare prior to puberty.3
Individuals with type 2 diabetes should be screened yearly upon diagnosis. They often have the disease for many years before being diagnosed, and the actual onset may be difficult to assess. As many as 30% of patients have retinopathy at initial diagnosis, and up to 3% may have macular edema or high-risk features.4
The type of diabetes is also important for coding and billing. Appropriate documentation will help support the 10th revision of the International Statistical Classification of Diseases and Related Health Problems (commonly known as ICD-10), because type 1 and type 2 diabetic retinopathy codes are categorized separately. For example, type 1 diabetes mellitus mild nonproliferative retinopathy without macular edema is E10.329, whereas type 2 diabetes mellitus mild nonproliferative retinopathy without macular edema is E11.329.
When Were You Diagnosed?
The duration of diabetes is a major risk factor for the development of vision-threatening diabetic retinopathy. Knowing the length of time since a patient’s diagnosis will aid in predicting the probability and severity of retinopathy. Approximately one in four patients with type 1 diabetes will have retinopathy after 6 years. The risk increases to 60% and 80% at 10 and 15 years, respectively.3 Signs of neovascularization will be present in up to 50% of patients who have had the disease for at least 20 years.
Retinopathy will be present in at least 24% of patients who have had type 2 diabetes for 5 years, increasing to 84% at 19 years’ duration. After 25 years, 25% of type 2 patients may have proliferative diabetic retinopathy.3
What Is Your Glycosylated Hemoglobin Level?
The most essential modifiable risk factor for the development and progression of vision-threatening diabetic retinopathy is glycemic control. Severity of hyperglycemia seems to be more important than duration once retinopathy is present. Historically, recommendations have included maintaining a glycosylated hemoglobin (commonly known as A1C) level of less than 7%, but some studies have recommended a target of 6.5% or lower.5 Routinely asking patients about their A1C level will reinforce its importance, and documenting it in the chart at each visit will help track a patient’s progress. It may also be helpful to consider shortening a patient’s follow-up period if his or her disease is poorly controlled, as this puts patients at higher risk of vision loss.
Chronic hyperglycemia can play a role in the development of cataract. Diabetic patients may require additional preoperative counseling because of elevated risk during and after cataract surgery. Studies have shown that hyperglycemia may contribute to delayed wound healing, an increased risk of postoperative infection or inflammation, and postoperative progression of retinopathy and macular edema.6 This has prompted some, such as medical governing bodies in Japan, to recommend against cataract surgery in patients with an A1C level greater than 10%.
Informing surgery patients that their visual outcomes can be improved with blood sugar optimization can help motivate them to improve their lifestyle and compliance. Caution should be urged, however, regarding rapid preoperative glycemic correction, as this may accelerate retinopathy.7
Less is known about the connection between diabetes and outcomes of refractive surgery. Although early FDA approvals included relative contraindications for LASIK in diabetics, there is growing evidence and acceptance that LASIK can be safely performed in those with good glycemic control and no systemic or ocular complications.
Significant fluctuations in blood sugar are known to cause morphologic changes in the crystalline lens. In addition, diabetic patients have altered corneal biomechanical properties, which can alter the response to laser treatment. These facts may account for higher surgical enhancement rates that have been reported in patients with diabetes.
What Medications Are You Taking?
Keeping track of a diabetic patient’s medications is an important component of the Meaningful Use Stage 2 incentive program for electronic health records. Additionally, there are ways to improve patients’ vision by monitoring their systemic medications. For example, pioglitazone and rosiglitazone have been associated with the development of macular edema, which resolves with cessation of the drugs.8
Aspirin therapy does not slow the progression of diabetic retinopathy. It also is not related to more severe, frequent, or persistent vitreous hemorrhage in patients with proliferative diabetic retinopathy. Therefore, no changes in medically administered aspirin therapy are indicated.
Intensive management of hypertension can slow retinopathy progression. Most diabetics are on an angiotensin II receptor blocker or angiotensin-converting enzyme inhibitor for kidney protection as well as blood pressure reduction. Although a practical target maximum blood pressure is 140 mm Hg/90 mm Hg, a lower goal is often sought for patients with chronic kidney disease or proteinuria.9
Who Is Your Medical Doctor?
Diabetes is a multisystem disease, so a multidisciplinary approach is necessary. Each patient may have several providers, including an ophthalmologist, nephrologist, endocrinologist, podiatrist, nutritionist, neurologist, and others, not to mention a primary care provider. If patients have other microvascular complications of diabetes, especially renal failure or proteinuria, they have a greater probability of vision-threatening diabetic retinopathy. The primary care provider, usually a general practitioner or internist, helps to coordinate care among members of this team. Communication with the primary care provider is an essential component of quality care.
The Physician Quality Reporting System measures for diabetic eye care include communication of examination results to the physician managing the diabetes care for patients with retinopathy. With many electronic health record systems, this is easily accomplished through digital transmission. It may be useful to copy the other members of the patient’s medical team on each report. Doing so will help facilitate care decisions, such as referral to an endocrinologist if retinopathy is progressing rapidly or planning eye surgery around a patient’s dialysis schedule.
What Do You Do for Exercise?
Many diabetic patients with neuropathy or cardiovascular disease think that they are unable to exercise. They may be surprised to hear that even small quantities of physical activity can be beneficial. Recording patients’ answers to this question in their charts will give insight into their functional status. Suggesting low-impact activities such as walking 30 minutes per day or joining a water aerobics class may encourage patients toward lifestyle changes they really need.
Obesity is strongly related to metabolic syndrome and insulin resistance. Weight loss has been proven to lower A1C levels and reduce patients’ need for oral hypoglycemic medication and insulin.10 This can be true for as little as a 5% reduction in body weight. A high body mass index is correlated with suboptimal glycemic control and the progression of diabetic retinopathy.11
Patients may be motivated by the fact that losing weight and lowering blood sugar levels will ultimately result in fewer procedures such as intravitreal injections and retinal laser. The United Kingdom Prospective Diabetes Study showed a 29% reduction in the need for retinal photocoagulation in a group undergoing intensive glucose management.12
CONCLUSION
The role of the ophthalmologist in caring for diabetic patients is, at its core, one of education. Patients need to know the importance of maintaining near-normal glucose and blood pressure levels, and they must understand that this is supported by years’ worth of evidence. Patients must be informed that effective treatment for diabetic retinopathy depends on timely intervention, even for patients who are asymptomatic with “good” vision.
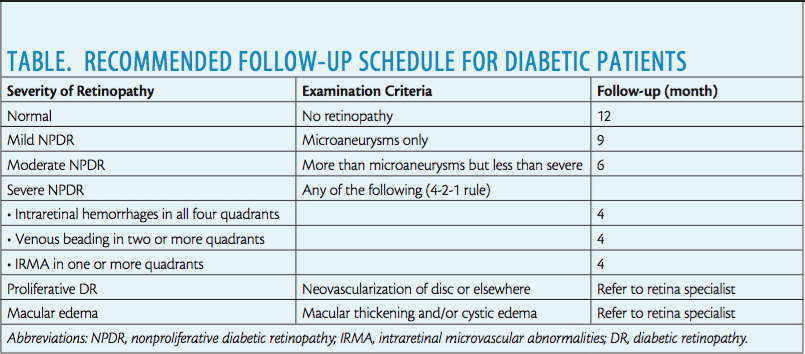
The ophthalmologist should therefore emphasize and encourage annual dilated eye examinations in patients without retinopathy and follow-up visits according to best recommendations for those with mild to severe nonproliferative disease (Table). Best practices require prompt referral to a retina specialist for treatment in the event of the development of macular edema or signs of neovascularization.
There is growing evidence that macular edema can be eliminated, and retinopathy can be reversed with appropriate antivascular endothelial growth factor therapy.13 Good visual outcomes can be achieved with a team approach to management that includes patients’ education and adherence.
1. Statistics About Diabetes: Data from the National Diabetes Statistics Report, 2014. http://www.diabetes.org/diabetes-basics/statistics/. Accessed February 10, 2016.
2. Crijns H, Casparie AF, Hendrikse F. Continuous computer simulation analysis of the cost-effectiveness of screening and treating diabetic retinopathy. Int J Technol Assess Health Care. 1999:15:198-206.
3. Klein R, Klein BE, Moss SE, et al. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. Arch Ophthalmol. 1984;102:520-526.
4. Diabetes Prevention Program Research Group. The prevalence of retinopathy in impaired glucose tolerance and recent-onset diabetes in the Diabetes Prevention Program. Diabet Med. 2007;24:137-144.
5. American Diabetes Association. Standards of medical care in diabetes—2013. Diabetes Care. 2013;36 (suppl 1):S11-66.
6. Squirrel D, Bhola R, Bush J, et al. A prospective case controlled study of the natural history of diabetic retinopathy and maculopathy after uncomplicated phacoemulsification cataract surgery in patients with type 2 diabetes. Br J Ophthalmol. 2002;86:565-571.
7. Suto C, Hori S, Kato S, et al. Effect of perioperative glycemic control in progression of diabetic retinopathy and maculopathy. Arch Ophthalmol. 2006;124(1):38-45.
8. Ryan EH Jr, Han DP, Ramsay RC, et al. Diabetic macular edema associated with glitazone use. Retina. 2006;26(5):562-570.
9. Nicholas SB, Vaziri ND, Norris KC. What should be the blood pressure target for patients with chronic kidney disease? Curr Opin Cardiol. 2013;28(4):439-445.
10. Delahanty LM, Dalton KM, Porneala B, et al. Improving diabetes outcomes through lifestyle change—a randomized controlled trial. Obesity. 2015;23(9):1792-1799.
11. Kastelan S, Salopek Rabatic J, Tomic M, et al. Body mass index and retinopathy in type 1 diabetic patients. Int J Endocrinol. 2014;2014:387919.
12. UK Prospective Diabetes Study Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes. UKPDS 33. Lancet. 1998;352:837-853.
13. RIDE and RISE Research Group. Long-term outcomes of ranibizumab therapy for diabetic macula edema: the 36-month results from two phase III trials: RISE and RIDE. Ophthalmology. 2013;120(10):2013-2022.
Section Editor Sumit “Sam“ Garg, MD
• medical director, vice chair of clinical ophthalmology, and an assistant professor of ophthalmology at the Gavin Herbert Eye Institute at the University of California, Irvine, School of Medicine
• serves on the ASCRS Young Physicians and Residents Clinical Committee and is involved in residents’ and fellows’ education
• gargs@uci.edu
Allen C. Ho, MD
• attending surgeon and director of retina research, Wills Eye Hospital, Philadelphia
• professor of ophthalmology, Sidney Kimmel College of Medicine, Thomas Jefferson University, Philadelphia
• chief medical editor, Retina Today
• acho@att.net
John D. Pitcher III, MD
• retina specialist, Eye Associates of New Mexico, Albuquerque, New Mexico
• jdpitcher@eyenm.com




