
In 2015, clinical research and innovation in point-of-care diagnostics contributed to continued evolution in the diagnosis and treatment of ocular surface disease (OSD). Progress in these areas leads to an important question: How can we best utilize these new tools, data, and therapies? This article reviews some of the new technologies available for OSD diagnosis and treatment and offers some of my own observations about how best to use these technologies in practice.
Most optometrists see patients with varying levels of OSD every day. No other disease state is so commonly seen in optometric practice.1 As the US population ages, primary eye care providers will continue to see increasing numbers of patients with macular degeneration, cataract, and glaucoma. Many of these patients will also have varying levels of comorbid OSD. The increasing need for access to care, coupled with continued progress in early detection, makes this a very exciting time for clinicians with a special interest in OSD. We have never been in a better position to help preserve stability of vision and to help our patients feel more comfortable. .
Most clinicians who treat OSD might agree that the conjunctiva and cornea are the most critical areas to zoom in on when carefully examining the patient with OSD. It is just as important, however, to zoom out and scrutinize the eyelid and adnexa of these patients (Figure 1). Instrumental work by Donald R. Korb, OD, and others has provided us with the fundamental understanding that the meibomian glands are the leading cause of dry eye disease in the majority of OSD patients.2-4 A comprehensive evaluation of the eyelid margin, including the meibomian glands, is imperative.
Figure 1. A gap between meibomian glands demonstrates gland atrophy and dropout.
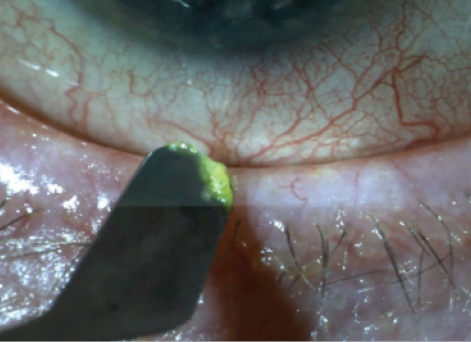
Figure 2. Conjunctival debridement.
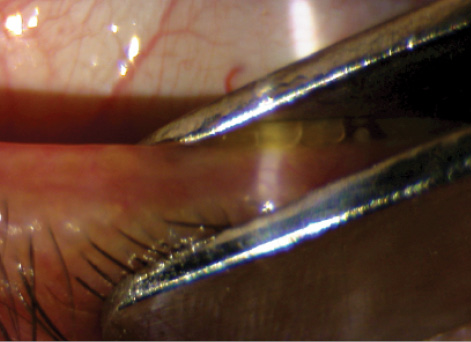
Figure 3. Therapeutic in-office meibomian gland expression using eyelid forceps.
COMMUNICATION
Today’s patient needs a detailed explanation of his or her condition using information from multiple sources. Connecting with patients can be more difficult than understanding the clinical picture and knowing what to do about it. The best plan increasingly demands a convincingly articulated explanation. Diagnostic tools available today allow us not only to pick up on subtle signs of disease but also to use these metrics to explain our treatment rationale to patients.
OSD DIAGNOSTICS
Osmolarity testing with the »TearLab Osmolarity System (TearLab) is an important diagnostic test for the OSD specialist. In addition to helping us understand which main category our OSD patient might fall into (evaporative or aqueous deficient), it also helps us to see trends that can help guide the treatment plan. In early dysfunction of the tear film, asymmetry is a hallmark that is worth discussing with the patient. It is also important to remember that, in the early stages of disruption of the tear film, as the compensatory lacrimal gland becomes more active, the OSD patient may be symptomatic despite a normal osmolarity score. These patients must be monitored closely. Although osmolarity is an excellent predictor of an unhealthy tear film, treatment should not be centered on any one given test.5 Rather, a meticulous interest in the rest of the clinical picture and diagnostic data is key.
»InflammaDry (Rapid Pathogen Screening) is another diagnostic tool that I find very useful, especially in eyes with surface irregularities such as anterior basement membrane dystrophy, pterygia, or Salzmann nodules or with an irregular surface after corneal surgery.
Dual-mode imaging of the meibomian glands with the »LipiView II (TearScience) is a giant step forward in accurately assessing the extent of meibomian gland dysfunction (MGD) and, just as important, photographically illustrating the dysfunction to the patient. Before this became available earlier this year, I was mostly using my penlight to evert the eyelid and retroilluminate the glands. This method can yield great detail. I can see individual glands and terminal ductal obstruction, and I can use the images to explain the process of truncation and gland atrophy to patients.
Dual-mode imaging provides detail of the meibomian gland at a level that most patients can understand and appreciate. This technology consistently delivers an elegant and detailed view of each gland on every patient. The next step using this technology must be to detect and categorize progression of MGD. We can now determine if two images of the same eyelid, taken 12 months apart, show stability or new areas of truncation and atrophy. It would be nice to be able to quantify progression much more precisely, in some ways similar to nerve fiber layer imaging and automated visual field progression analysis.
OSD TREATMENT
»LipiFlow (TearScience) is a very useful in-office treatment for patients with mild to moderate MGD. Using the stabilizers (stickers on the patient’s nose) is critical to the success of the treatment. The stability of the activators during the treatment is vital for complete and diffuse gland expression. I increasingly rely on patients during the procedure to tell me how the treatment feels on each eyelid. If there is asymmetry, I often interrupt the treatment and reposition the activator.
Choosing the right patients for LipiFlow treatment is important. I find it most useful in patients with early to moderate MGD. I generally characterize these two stages by examining the meibum from each gland. Even if the meibum is so thick that it is cheese-like in consistency, I find that patients feel better after treatment with LipiFlow.
For severe OSD patients with only a few meibomian glands remaining, treatment with LipiFlow may not provide significant improvement in oil delivery to the ocular surface. The remaining glands may need even more aggressive heating and evacuation. This may possibly be achieved with intense pulsed light therapy followed by ductal probing and further expression at the slit lamp.
Maintaining the impact of the LipiFlow treatment often requires more in-office therapy in addition to home therapy. For this reason, the decision by TearScience to offer a lower price for its single-use activators, announced in October 2015, should prove to be a successful strategy. Unlike cataract or refractive surgery, OSD is a chronic condition. Successful OSD treatment may need to be repeated. I find it useful to combine intense pulsed light and LipiFlow treatments in more severe cases.
Meibomian gland ductal probing can also be helpful in some eyes with obstructive MGD. The key to success is to use the procedure more superficially, without penetrating deep into the duct. I often combine this with eyelid debridement (Figure 2) to clear the path for the meibum to reach the ocular surface, and then I express the glands with eyelid forceps (Figure 3).6
We providers have learned how crucial healthy, patent meibomian glands are, and we now have increasingly improved methods to detect and treat MGD. Just as important as the health of the gland, of course, is the health of the tissue it resides in. The recognition of the pervasiveness of Demodex blepharitis has sparked important discussions regarding how relevant these little creatures are and what should be done about them. Should we get rid of all cylindrical dandruff and kill each parasite? Is that possible? In my clinic, treating Demodex blepharitis has helped patients who mostly complain of itching yet whose clinical picture is not consistent with allergy. The best way to get patients on board with a treatment plan for Demodex blepharitis is to show them the Demodex mite on a microscope slide. This can easily be done using a portable digital microscope (Figure 4).

Figure 4. Multiple Demodex mites on an eyelash after epilation.
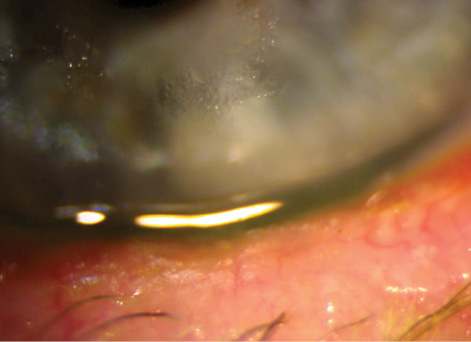
Figure 5. Untreated blepharitis leading to epithelial keratitis.
For these patients, daily use of a comfortable, effective eyelid cleaner such as Avenova (NovaBay Pharmaceuticals) has been a successful strategy. In comparison with other lid wipes, I have found Avenova to yield excellent patient feedback and healthier eyelids. We need more data on how lid-wipe products are different from each other in order to design a more scientifically supported protocol. It is clear that we need to critically examine possible sources of blepharitis just as much as we now carefully assess the meibomian glands (Figure 5).
For patients who have heaps of cylindrical dandruff, multiple in-office debridement treatments are required. I think »BlephEx (BlephEx) is a great tool, although I find that it is not aggressive enough for debridement of significant cylindrical debris or seborrheic debris. I look forward to seeing the next version of the BlephEx device.
Over the years, multiple drugs developed for the treatment of OSD have failed to gain approval from the FDA. Hundreds of millions of dollars have been invested in drug development for OSD, yet due to the complex scientific, clinical, and regulatory challenges, we do not have multiple topical drugs indicated for the treatment of OSD.
Lifitegrast (Shire) may be approved by the FDA in early 2016 for the treatment of signs and symptoms of dry eye disease in adults. Shire announced in October 2015 that, in response to the company’s new drug application for lifitegrast, the FDA asked for an additional clinical study, and Shire intends to use the results of a recently completed phase 3 clinical trial to fulfill that request. Lifitegrast is a first-in-class nonpreserved small-molecule integrin antagonist targeting T cells to keep them from becoming active. This molecule has a lot riding on its shoulders, and many clinicians are looking forward to its approval as a healthy sign of more topical drugs in the coming years.
For patients with severe OSD and significant keratitis, I find adjunctive topical therapy with autologous serum to be a useful strategy.7 The main advantage that autologous serum can offer the patient is epithelial healing. In my experience, treatment with autologous serum drops has been helpful for patients with Sjögren syndrome and other autoimmune diseases. I find autologous serum eye drops to be superior to other compounded medications such as testosterone-progesterone drops. Autologous serum is a fantastic addition to available topical treatments. In my experience, autologous serum has been the most useful topical therapy for patients with severe OSD.
Another valuable option for the treatment of a compromised corneal epithelium is the application of amniotic membrane. »Prokera biologic corneal devices (Bio-Tissue) are an effective and convenient option for treatment of epithelial damage that has not responded to topical and oral medication. The stability of the Prokera device can be a challenge for patients with significant lid scarring or ectropion. Tegaderm (3M) medical tape can be used to carefully close the lids to help keep the lens in place and comfortable for these patients.
LOOKING AHEAD
Looking forward to 2016, I think the most important progress in OSD treatment might come not in the form of more new diagnostic tests but rather in how we understand and apply the recent advances in diagnostics described herein to provide smarter treatments and superior outcomes. These innovations in diagnostic tools have shifted the treatment pendulum away from simply responding to patients’ symptoms.8 Given that clinical evidence of OSD precedes symptoms, it is reasonable to argue that symptoms are not the most relevant piece of information in OSD. A careful primary eye care provider will treat early, before tissue damage occurs.
I completely agree with this mindset. At the very least, I offer OSD patients a baseline protocol that includes adequate hydration, dietary supplementation with omega-3 fatty acids, and lid hygiene. A more aggressive approach is imperative perioperatively in patients undergoing surgery. We know that patients can have signs of mild OSD that will affect keratometry readings essential for IOL power calculation.9 Catching these patients and treating them early and appropriately will undoubtedly improve surgical outcomes.
At the same time, I do not think it is appropriate to perform extensive testing on patients who are completely asymptomatic. A correct balance between the utilization of diagnostic tests and listening to patients’ symptoms is the key. Finding that right balance is definitely an art. We are fortunate to be practicing at a time when such innovative advances are available.
1. Lemp MA, Nichols K. Blepharitis in the United States 2009: a survey-based perspective on prevalence and treatment. Ocul Surf. 2009;7(2 suppl):S1-S22.
2. Lemp MA, Crews LA, Bron AJ, et al. Distribution of aqueous deficient and evaporative dry eye in a clinic-based patient cohort: a retrospective study. Cornea. 2012;31(5):472-478.
3. Knop E, Knop N, Millar T, et al. The International Workshop on Meibomian Gland Dysfunction: report of the subcommittee on anatomy, physiology, and pathophysiology of the meibomian gland. Invest Ophthalmol Vis Sci. 2011;52(4):1938-1978.
4. Knop E, Korb DR, Blackie CA, et al. The lid margin is an underestimated structure for preservation of ocular surface health and development of dry eye disease. Dev Ophthalmol. 2010;45:108-122.
5. Bron AJ, Tomlinson A, Foulks G, et al. Rethinking dry eye disease: a perspective on clinical implications. Ocul Surf. 2014;12(2S):S1-S31.
6. Korb D, Blackie C. Debridement-scaling: a new procedure that increases meibomian gland function and reduces dry eye symptoms. Cornea. 2013;32(12):1554-1557.
7. Ariza AA, Rapuano CJ. Autologous serum eye drops for the treatment of ocular surface disease. Eye Contact Lens. 2015;41(3):133-140.
8. Sullivan BD, Crews LA, Messmer EM, et al. Correlations between commonly used objective signs and symptoms for the diagnosis of dry eye disease: clinical implications. Acta Ophthalmol. 2014:92:161-166.
9. Trattler W, Reilly C, Goldberg D, et al. Cataract and dry eye: Prospective Health Assessment of Cataract Patients’ Ocular Surface Study. Poster presented at: American Society of Cataract and Refractive Surgery Annual Meeting; March 25-29, 2011; San Diego, CA.
Ahmad M. Fahmy, OD, FAAO, Dipl ABO
• director of clinical operations, Minnesota Eye Consultants, Bloomington, Minnesota
• (651) 398-5728; amfahmy@mneye.com
• financial interest: none acknowledged


