Digital Registration for Accurate IOL Placement
By Richard S. Davidson, MD

With rising expectations regarding cataract surgery’s refractive predictability, the procedure has become as much a refractive procedure as one that restores vision. It is no surprise, then, that the use of premium IOLs in the United States is growing by 48% annually.¹ Compared with standard monofocal lenses, greater accuracy is needed to confer the benefits for which toric and multifocal IOL platforms were designed. Surgeons who faithfully track their outcomes generally achieve ±0.50 D spherical equivalent accuracy for between 71% and 80% of cases.2-5 Only 6% of surgeons achieve that level of accuracy for 84% or more of cases, and less than 1% of surgeons achieve that level of accuracy for 92% or more cases.6 Research has shown that, for every 1º a toric lens is off axis, its effectiveness for reducing astigmatism is decreased by 3.3%; if the lens is off by 10º, its efficacy is decreased by 33%.7
A MARKERLESS OPTION
Intraoperative Alignment Tools
The advent of intraoperative toric alignment tools has helped to increase the theoretical accuracy of toric lens alignment. The Zeiss Cataract Suite (Carl Zeiss Meditec) features the IOLMaster 500 optical biometer, Callisto Eye markerless toric alignment system, and OPMI Lumera surgical microscope (Figure 1). These products assist physicians with the centration of IOLs along the visual axis and the alignment of toric IOLs along the optimal target axis via the use of graphical overlays projected in the eyepieces of the surgical microscope in a heads-up display-like fashion.
How It Works
The markerless version of the cataract suite, which received FDA 510(k) clearance in 2014, provides the benefit of accurate toric IOL alignment without needing to preoperatively mark the corneal surface. Reference axis marking (marking the cornea at 0º, 180º, and 270º), axis identification marking (marking the steep axis), and toric IOL alignment lead to a mean total error in toric IOL alignment of 4.9 ±2.1º (Figure 2).2-4,7 This “stackable error” is magnified by the need to mark both the 0º to180º horizontal meridian and the toric alignment axis. Callisto removes this error by using a reference image (Figure 3) captured by the IOLMaster as a means of registering the eye during surgery via the patient’s unique limbal blood vessel pattern and representing both the 0º to 180º meridian and the toric target axis digitally. The limbal vessel registration also enables Callisto to track and thus account for any eye movement along the x-y plane or cyclorotation so that the digital projections are properly positioned. Figure 4 demonstrates the capsulotomy overlay as viewed through the oculars of the microscope. The overlay acts as a template that allows surgeons to make a perfectly centered anterior capsulotomy.

Figure 1. The Zeiss Cataract Suite.
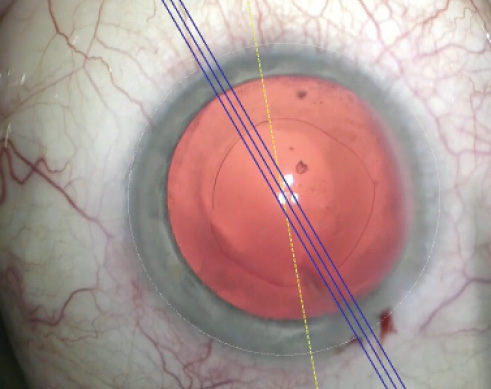
Figure 2. The Callisto Eye markerless alignment system. The yellow line represents the marking of the reference axis, and the blue lines represent the steep axis of the cornea.
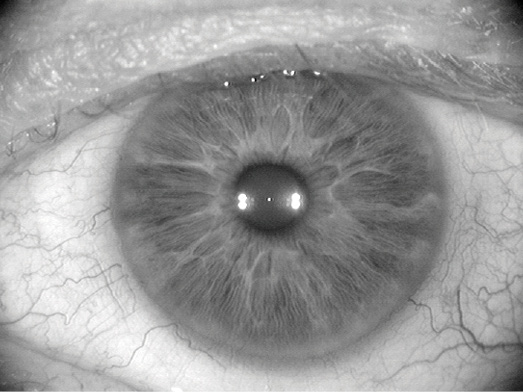
Figure 3. The reference image captured by the IOLMaster 500 as a means of registering the eye during surgery via the patient’s unique limbal blood vessel pattern.
INCREASED ACCURACY
Early experience suggests that this method of digitally registering the patient’s eye and representing the key centration point and toric alignment axis in the eyepieces of the microscope can dramatically improve the accuracy of IOL placement.8 At this year’s American Society of Cataract and Refractive Surgery annual meeting, Daniel Black, MBBS (Qld), FRANZCO, FRACS, A/FACAsM, presented data showing a statistically significant improvement in his patients’ outcomes with the markerless cataract suite. Of the 507 patients he treated with traditional ink markings, 94% were within 0.50 D of the target refraction, and 98% were within 0.75 D. In comparison, of the 161 patients who underwent markerless toric alignment with Callisto, 99.4% were within 0.50 D of the target, and 100% were within 0.75 D.8 The differences between the two groups in the percentage of patients who were within 0.50 D was statistically significant (P = .028).8
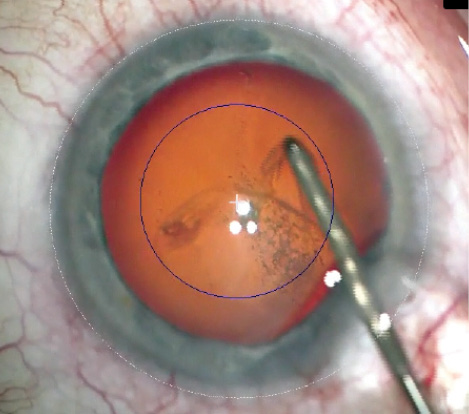
Figure 4. The capsulotomy overlay as viewed through the oculars of the microscope. The overlay acts as a template that allows surgeons to make a perfectly centered anterior capsulotomy.
EFFICIENT WORKFLOW
In addition to the clinical benefits, the Zeiss Cataract Suite enhances workflow and efficiency. Not only is there no longer a need to preoperatively mark patients’ eyes, but when used in conjunction with the Zeiss Forum data management system, the markerless cataract suite also seamlessly transfers biometry data between the IOLMaster, which is typically located in the physicians’ office, to Callisto, which is located in the OR. This connectivity increases efficiency, enhances the security of electronic health information, and eliminates manual data entry errors. Even without using Forum as a conduit, the ability to readily transfer the prerequisite biometry and reference image from the IOLMaster directly to Callisto via USB increases clinical precision without significantly changing cataract surgery workflow.
1. Market Scope Report. Toric IOL demand outpaces demand for all other premium IOLs. March 2012. http://market-scope.com/category/march-2012. Accessed May 11, 2015.
2. Miyake T, Kamiya K, Amano R, et al. Long-term clinical outcomes of toric intraocular lens implantation in cataract cases with preexisting astigmatism. J Cataract Refract Surg. 2014;40:1654-1660.
3. Hirnschall N, Hoffman PC, Draschi P, et al. Evaluation of factors influencing the remaining astigmatism after toric intraocular lens implantation. J Refract Surg. 2014;30(6):394-400.
4. Waltz KL, Featherstone K, Tsai L, Tretacost D. Clinical outcomes of Tecnis Toric intraocular lens implantation after cataract removal in patients with corneal astigmatism. Ophthalmology. 2015;122:39-47.
5. Behndig A, Montan P, Stenevi U, et al. Aiming for emmetropia after cataract surgery: Swedish National Cataract Registry. J Cataract Refract Surg. 2012;38(7):1181-1186.
6. Hill WE. Piggyback IOL case. Paper presented at: Cornea Subspecialty Day, ASCRS/ASOA Symposium; April 19, 2013; San Francisco, CA.
7. Visser N, Berendschot TT, Bauer NJ, et al. Accuracy of toric intraocular lens implantation in cataract and refractive surgery. J Cataract Refract Surg. 2011;37(8):1394-1402.
8. Black D. Evaluation of markerless alignment system for toric IOLs. Paper presented at: ASCRS/ASOA Annual Symposium & Congress; April 20, 2015; San Diego, CA.
Intraoperative Guidance for Heads-up, Three-Dimensional Surgery
By James A. Katz, MD

The transition from manual marking methods for astigmatic correction and toric IOL alignment to digital technologies took a leap forward in 2010 with the FDA’s 510(k) clearance of the TrueVision guidance software application (TrueVision 3D Surgical). The software, now called TrueGuide, uses both data and image inputs that act as an incision and IOL calculator for surgical planning and during live surgery.
CUSTOMIZABLE SETUP
With TrueGuide, I can use the ecosystem in total or in parts, based on my microscope configuration and interests. My setup includes the presurgical planning software called TruePlan, which has been configured to accept both image and data exports from the Cassini corneal analyzer (i-Optics). Patients’ parameters, including white-to-white, mesopic pupillary center and diameter, keratometry values, and high-resolution eye images, are exported from a standard Cassini topography acquisition into the TruePlan software. The software automatically suggests a primary incision axis, a toric IOL power and axis or an arc length, and an axis for limbal relaxing incisions (LRIs), depending on the magnitude of astigmatism and the surgical plan. The software uses a fully customizable, doctor-specific profile to create surgical plans that are unique to the surgeon’s preferences such as the operating position and surgically induced astigmatism (SIA). The software generates guidance templates and predicted residual astigmatism values that can be easily adjusted during surgical planning or live surgical setup. I also find the system useful for IOL centration guidance.
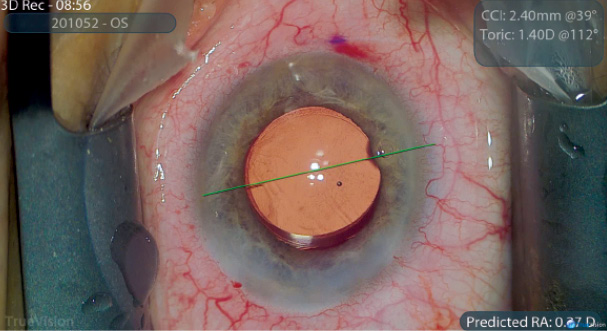
Figure 1. Toric IOL alignment using TrueGuide.
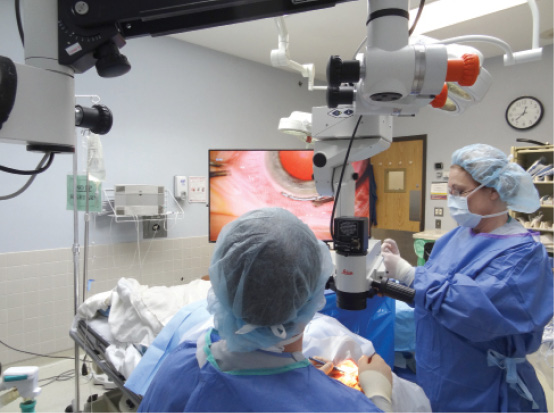
Figure 2. Heads-up 3-D cataract surgery.
The TrueGuide software was designed to automatically link the surgeon-specific SIA and astigmatic correction via a process called dynamic optimization, which allows for adjusting the location of incisions due to parameters such as the surgeon’s seated position and customized incision range. Real-time vector analysis recommends a placement for an incision that predicts the least residual astigmatism for toric IOL and LRI positioning (Figure 1).
LIVE SURGICAL FIELD
Using the latest generation of the TrueVision camera module and ultra HD 4K imaging, I routinely perform heads-up three-dimensional (3-D) cataract surgery, which enables 3-D registration and live eye tracking of the TrueGuide images and guidance templates (Figure 2). The camera module streams the live surgical field to a large or small 3-D monitor, depending on the surgeon’s preference for image size and ergonomics. I position TrueVision’s 55-inch 4K monitor perpendicular to me, which allows my surgical support staff to view the case easily.
PRESURGICAL PLANNING
The ecosystem can consist of the presurgical planning software in combination with the Cassini device or as standalone planning software with inputs for keratometry values and preoperative images. The surgical setup can be used in a heads-up live surgical manner with guidance provided on demand throughout the incision and IOL alignment stages, or it can simply be employed during the steps of astigmatic correction. Although the system works with any existing surgical microscope, its combination with the Leica microscope foot pedal increases surgeons’ control of the guidance features. They can use the system with a portable cart or by integration with a Leica microscope. The touchscreen control monitor and the Cassini axial topography map overlay provide an additional intuitive guide for astigmatic correction. The software allows for other secondary inputs such as an intraoperative aberrometry feed to be viewed in additional software windows. These controls permit the surgeon to operate with a fighter pilot’s cockpit-like setup and call up guidance tools as needed, while maintaining focus on the surgical field.
CONCLUSION
In my hands, version 10 of the TrueGuide software, which works with the 4K visualization setup, is a digitally precise tool for positioning the surgical incision and IOL. As the rapid pace of digital guidance innovation continues, I look forward to further customization.
At a Glance
• The Zeiss Cataract Suite features products that assist physicians with the centration of IOLs along the visual axis and the alignment of toric IOLs along the optimal target axis via the use of graphical overlays projected in the eyepieces of the surgical microscope in a heads-up display-like fashion.
• TrueGuide uses both data and image inputs that act as an incision and IOL calculator for surgical planning and during live surgery. The software is customizable to the surgeon’s preferences.
• Verion assists with preoperative planning, and ORA with VerifEye is used for intraoperative guidance.
Synergetic Technologies for Preoperative Planning and Intraoperative Accuracy
By Michael G. Woodcock, MD

The relationship between the Verion Image Guided System and the ORA System with VerifEye or VerifEye+ technology (all from Alcon) is analogous to that of a coach and the star players of a soccer team. When the coach (Verion) has a well-conceived game plan, the likelihood of a successful outcome is high, provided that the team performs as expected. Whenever the unexpected occurs and the original plan stops working, it falls on the shoulders of the star players (ORA functions) to adjust and win the game.
PREOPERATIVE PLANNING
With refractive cataract surgery, nothing can substitute for preoperative planning and preparation. I measure and register astigmatism in the office with Verion, and then I operate on the exact axis in the OR. I integrate data from Verion and other biometry into a game plan for IOL selection as well as astigmatic treatment with a laser, toric IOL, or a combination of the two. I use the digital overlay captured in the office to register the exact axis for the placement of operative tunnels, arcuate incisions, and IOL alignment throughout the surgical procedure.
Verion helps me to decrease patients’ postoperative astigmatism. Roughly 47% of the population has 0.75 D or less of corneal astigmatism.1 Verion allows me to eliminate problems due a patient’s tilted head and errors in marking and transcription. Some surgeons might argue that incisional correction guided by Verion could be extended into the 1.50 to 2.00 D range, but I prefer using a toric IOL guided by ORA to correct more than 1.25 D of corneal cylinder.
INTRAOPERATIVE GUIDANCE
What happens when the unexpected occurs and the preoperative game plan needs to change? For the past 5 years, my game-winning star player has been ORA with VerifEye, using Talbot-Moiré interferometry. ORA integrates preoperative biometry with real-time findings to allow me to adjust to the game’s (surgical) conditions.
Surgical outcomes are traditionally no better than the data surgeons input and the predictability of the formulas they use. Optical biometry has nailed axial length, but there are a plethora of methods to measure corneal power and astigmatism and just as many opportunities for inaccuracies to occur. For example, a poor-quality tear film, the lack of calibration, measurement errors, transcription errors, posterior astigmatism, and poor cooperation from patients all decrease accuracy. With ORA, I can observe the quality of the tear film, control IOP to physiologic conditions, ensure that measurements are centered on the patient’s visual axis, and obtain precise real-time refractive data. ORA uses these data to compute recommendations based on algorithms derived from over 100,000 prior refractive cataract procedures. Unlike other intraoperative guidance systems, the ORA recommends a spherical power for normal eyes, postradial keratotomy eyes, and those that have undergone myopic and hyperopic laser vision correction. I have found the recommendations from ORA for toric IOL power and orientation unparalleled and invaluable.

Figure. Surgeon’s right ocular dynamic image. The aphakic refraction and astigmatic axis are depicted along with the preplanned IOL. Along the right edge is the focus bar or target acquisition. Note VerifEye+ determined that a toric IOL was not indicated.
Wavefront power measurement incorporates both anterior and posterior corneal curvature and simplifies the power variable, increasing the accuracy of both spherical and cylindrical outcomes. The third-generation ORA System with VerifEye obtains 40 measurements in less than 2 seconds, but the preview mode with streaming refractive data allows me to observe the tear film and choose the most stable 2 seconds to measure. The new VerifEye+ version projects a dynamic reticle into the right ocular, allowing rapid, precise alignment of the toric axis without my leaving the microscope. For highly aberrated corneas, where a statistically verified prediction cannot be obtained with an algorithm, I can gather useful information during the streaming preview. Many times, the streaming preview has guided successful choices that I verify during a pseudophakic reading.
CONCLUSION
The ORA System with VerifEye technology and Verion are useful alone and are synergistic together. Both provide me with valuable tools for the refractive cataract surgery game. n
- Hill W. Expected effects of surgically induced astigmatism on AcrySof toric intraocular lens results. J Cataract Refract Surg. 2008;34(3):364-367.
Richard S. Davidson, MD
• associate professor and vice chair for quality and clinical affairs, University of Colorado Eye Center, Aurora, Colorado
• (720) 848-2500; richard.davidson@ucdenver.edu
• financial disclosure: consultant to Carl Zeiss Meditec
James A. Katz, MD
• director, Golf Laser Vision, The Midwest Center for Sight, Des Plaines, Illinois
• (847) 824-3127; jkatz@tmcfs.com
• financial disclosure: consultant to i-Optics and TrueVision 3D Surgical and has ownership interest in TrueVision 3D Surgical
Michael G. Woodcock, MD
• Carolina Vision Center, Fayetteville, North Carolina
• (910) 485-3937; lasikpro@gmail.com
• financial disclosure: consultant to Alcon


