The Galilei
By Neel R. Desai, MD

It is no coincidence that the biometric analysis of potential refractive cataract patients and LASIK patients is becoming nearly indistinguishable. The former has, appropriately, become increasingly sophisticated, as ophthalmologists' understanding of the influence of corneal topography and tomography, anterior and posterior corneal astigmatism, higher-order aberrations, and subtle corneal pathologies grows in step with patients' expectations for high-quality spectacle-independent vision.
The marketplace was recently flooded with devices that provide pre- and intraoperative guidance to the surgeon based on unique modes of measurement and interpretation of corneal indices. Working at a high-volume, technology-forward, cornea and refractive cataract practice, my colleagues and I have had experience with nearly every available device from traditional manual keratometers to the IOLMaster (Carl Zeiss), Lenstar (Haag-Streit), OPD II (Nidek), Orbscan (Bausch + Lomb), Cassini (i-Optics), Pentacam Comprehensive Eye Scanner (Oculus Surgical), and Galilei G4 (Ziemer Ophthalmic Systems). Some of these devices play key roles in our clinical decision making and daily practice. Clinicians may be asking, just as we did 1 year ago, “What really is the difference between these devices, and which one is going to give us the most information we need to make better clinical decisions for our patients?” In our view, the Galilei offers clinicians many benefits in one stand-alone device.
STRENGTHS
Topography and Tomography
Cornea, refractive, and cataract surgeons alike need corneal topography and tomography. Placido disk-based systems can directly measure corneal curvature, but data must be extrapolated to determine anterior and posterior corneal elevation and pachymetry. Conversely, Scheimpflug-based imaging technologies can directly measure—and allow interpretation of—corneal elevation maps but cannot directly assess corneal curvature. The Galilei uniquely combines both technologies in one device, allowing the simultaneous measurement of central corneal curvature, pachymetry, and anterior and posterior corneal elevation (Figure 1).
An additional point is that decentration of the cornea can decrease the accuracy of Scheimpflug pachymetry measurements when only one Scheimpflug camera is used. The dual-Scheimpflug system of the Galilei not only speeds the scanning process, but it also compensates for patient misalignment by allowing rotational scanning from opposing sides. An analytical report allows the technician to quickly assess the quality of the scan to ensure that accurate and useful data are transmitted to the surgeon at the time of the consultation and examination. These features are essential to fast and accurate workflow in a busy clinic.
Corneal Evaluation
Undetected on traditional biometry, irregular astigmatism can compromise results with toric and presbyopia-correcting IOLs. Subtle corneal pathology such as epithelial basement membrane dystrophy and Salzmann nodular corneal dystrophy or frank disease like keratoconus must be identified before refractive cataract surgery. The keratometry used by the IOLMaster and Lenstar devices may erroneously reflect regular astigmatism in a cornea that actually has basement membrane dystrophy or irregular astigmatism produced by keratoectatic disease. Placing limbal relaxing incisions, toric IOLs, or presbyopia-correcting IOLs based on these measurements will achieve lackluster results at best. We use the Galilei during every cataract consultation, because it identifies these often subtle issues preoperatively.

Figure 1. Dual-Scheimpflug imaging and Placido disk topography are combined in one powerful device.
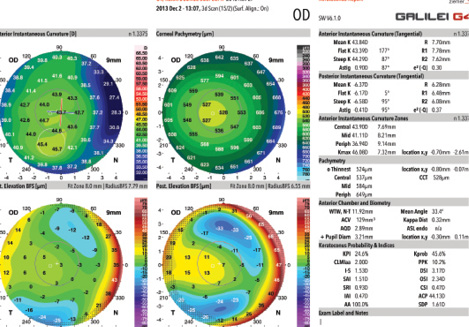
Figure 2. The Galilei G4 can detect subtle forme fruste keratoconus with high sensitivity and specificity.
Data are only as good as the indices used to interpret them and render them clinically relevant. Robust software programs built into the Galilei produce clinically useful indices. For example, the Asphericity Asymmetry Index (AAI) uses an artificial intelligence system and decision-tree algorithm to detect forme fruste keratoconus with impressive 93.7% sensitivity and 97.2% specificity (Figure 2).1,2 The posterior AAI is a qualitative index of posterior surface asymmetry, thought to be the first indication of keratoectasia risk, as described by Arce in 2010.3 Unlike most keratoconus indices, which are based on either pachymetry or strict cutoffs of deviations from a best-fit sphere, the AAI is calculated using a best-fit toric and aspheric reference surface, which improves the sensitivity for detecting keratoectasia.4
Lens Selection
Quality of vision diminishes with increasing total spherical aberration. IOLs are now available in spherical, aspheric-neutral, and negatively aspheric designs to offset corneal asphericity. The Galilei allows us to measure these higher-order aberrations and select the most appropriate IOL for a given patient. For example, if the device detects significant higher-order aberrations, we recommend against multifocal IOLs.
Douglas Koch, MD, and colleagues demonstrated the importance of accounting for posterior corneal astigmatism when planning to implant a toric IOL. The Galilei's ability to image and measure the posterior cornea has increased our understanding of why we often overcorrected with-the-rule astigmatism and undercorrected against-the-rule astigmatism with toric IOLs when we used only anterior keratometric data. We are now able to factor in this effect using the Galilei and more accurate nomograms.
WEAKNESSES
With so many new technologies, methodologies for measurement, and indices for interpretation, there is a risk that practitioners may become overwhelmed by the mass of available data and have difficulty knowing how to prioritize this information when making clinical decisions on the fly. Like other next-generation corneal analyzers, the Galilei offers so many different metrics that are not yet fully understood. Time and the ongoing work of a number of clinician-scientists will help to elucidate the meaning and value of these metrics.
ON THE HORIZON
With the anticipated addition of optical coherence tomography-based biometry and axial length measurements, the Galilei G6 Lens Professional Suite (not yet available in the United States) will allow surgeons to perform all necessary biometry, lens selection, and even limbal relaxing incision planning on one device. This, I believe, will be a true “game-changer” for busy practices with limited physical space and budgets.
1. Maeda N, Klyce SD, Smolek M. Automated keratoconus screening with corneal topography analysis. Invest Ophthalmol Vis Sci. 1994;35:2794-2757.
2. Smadja D, Touboul D, Cohen A, et al. Detection of subclinical keratoconus using an automated decision tree classification. Am J Ophthalmol. 2013;156(2):237-246.
3. Arce C. Qualitative and quantitative analysis of aspheric symmetry and asymmetry on corneal surfaces. Poster presented at: ASCRS/ASOA Symposium and Congress; April 9-14, 2010; Boston, MA.
4. Smadja D, Santhiago MR, Mello GR, et al. Influence on the reference surface shape for discriminating between normal corneas, subclinical keratoconus, and keratoconus. J Refract Surg. 2013;29(4):274-281.
iTrace
By Paul Mann, MD

Managing corneal astigmatism using toric IOLs is the standard of care in cataract surgery, and perfecting outcomes is a common topic of discussion. Preoperative planning is important to successful treatment, which has prompted cataract surgeons to use topography and software to improve results.
ABOUT THE SYSTEM
Surgical planning requires an evaluation of corneal curvature and astigmatism and imaging for axial placement. Curvature is no longer described using simple keratometry. Instead, the system assesses the corneal curvature in diopters taken via reflection at four points on the central 3-mm ring. Simulated keratometry uses topography data to calculate the keratometry values using the corresponding four points on the 3-mm ring. Refractive keratometry is an average of all curvature data measured within the central 3 mm from the topographic map, and it better reflects corneal power.
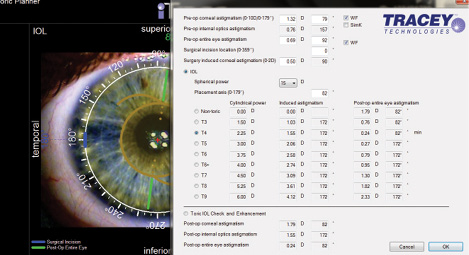
Figure 1. The iTrace toric calculator uses information from the unit's examination to help the surgeon select the best toric power for a patient.
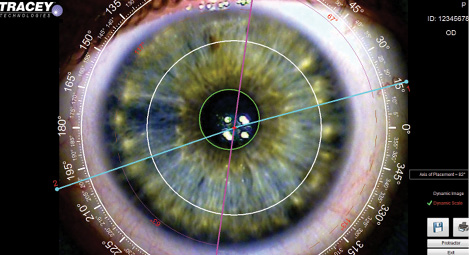
Figure 2. A color image with caliper can help improve the precision of the toric IOL's placement.
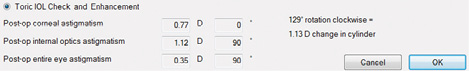
Figure 3. The PostOp Toric Check 1 shows the difference between the patient's measured corneal and internal astigmatism, both in power and axis.

Figure 4. Three examples of the utility of the PostOp Toric Check. In case 1, a small rotation of the IOL may be necessary, but the outcome is not significant enough to warrant an enhancement. In case 2, rotation could improve the outcome by more than 3.00 D. In case 3, rotating the IOL will not improve the outcome; the patient's residual astigmatism is due to an overpowered toric lens.
In addition, the iTrace (Tracey Technologies) provides a similar wavefront keratometry value in its toric planner. This keratometry measurement is based on the central 4-mm disk of information but is translated through Zernike calculations back to the cylindrical power and axial keratometry value. I have found using refractive and wavefront keratometry values to be more accurate in modern formulas, especially for eyes with irregular and nonorthogonal astigmatism.
STRENGTHS
Overlays
The iTrace's toric planner includes unique eye image overlays to assist with intraoperative axial alignment. This tool incorporates information on corneal and refractive astigmatism, IOL spherical power based upon biometry, incision location, and surgically induced astigmatism to help me choose the appropriate toric IOL (Figure 1). It compares the residual astigmatism with each toric IOL.
The overlays guide axial placement of the IOL based upon a color image of the external eye. I can use simulated placement of the IOL axis relative to conjunctival landmarks intraoperatively to reduce positional errors when the patient is supine (Figure 2).
Postoperative Measurements
Careful preoperative evaluation with the iTrace has improved my outcomes with toric IOLs. In rare cases, however, a lens may rotate in the days after implantation. Postoperatively, the iTrace can measure the power and axis of corneal cylinder and the toric IOL's axial location to help me assess the surgical correction (Figure 3). From this information, I can determine if the lens should be rotated in situ to optimize the outcome (Figure 4). This feature can be reassuring, because I prefer not to enter the eye a second time unless the improved outcome warrants it.
WEAKNESSES
At this time, the iTrace cannot be used intraoperatively to measure the eye's power. I might argue, however, that this is not an ideal time to obtain accurate measurements, owing to the many factors at work during surgery, including lid retraction and issues with the tear film.
Cassini
By P. Dee G. Stephenson, MD

I enjoy being an early adopter of technology. I am a solo practitioner, however, so it is very important that the technology provide advantages to my practice. Because topography is an essential aspect of planning astigmatic management, I hesitated to change from my Placido-based topographer when the Cassini (i-Optics) first became available. I decided to purchase the device based on my own research into the technology and the endorsements from my peers.
ABOUT THE SYSTEM
The unit takes a different approach than the traditional Placido disk topographers. The former uses point-to-point light-emitting diode ray tracing analysis to acquire images. I find that the 679 points provide a highly repeatable axis and magnitude of astigmatism for my surgical planning.
Total Corneal Astigmatism (TCA) functionality is a recent addition to the system. The Cassini uses the second Purkinje reflections off the posterior surface of the cornea to measure the posterior corneal axis and magnitude of astigmatism. The system analyzes the anterior and posterior measurements to calculate a total corneal magnitude and axis of astigmatism.
Posterior astigmatism was shown to be a major cause of postoperative refractive surprises by Li Wang, MD, PhD; Douglas Koch, MD; and the entire Baylor team.1 The Baylor nomogram works off a population mean and provides reliable results in 85% to 90% of my patients. With the Cassini, the idea is to customize the treatment of each patient through the TCA measurement (Figure).
STRENGTHS
Repeatable Data
The axis' repeatability with the Cassini has proven superior to other methods of measuring astigmatism. Starting with the correct axis of placement is critical, because studies indicate a 3.3% drop in corrective power for each degree to which the toric lens is placed on the wrong axis.2 Studies have shown the repeatability in terms of magnitude to be comparable with Scheimpflug cameras and Placido-based topographers.3
TCA Measurement
I find that capturing a posterior measurement only adds 2 seconds to the topographic examination. The Cassini provides me with anterior, posterior, and total corneal magnitude and axis as well as keratometry readings. The information is displayed where I can easily compare and gather data for planning.
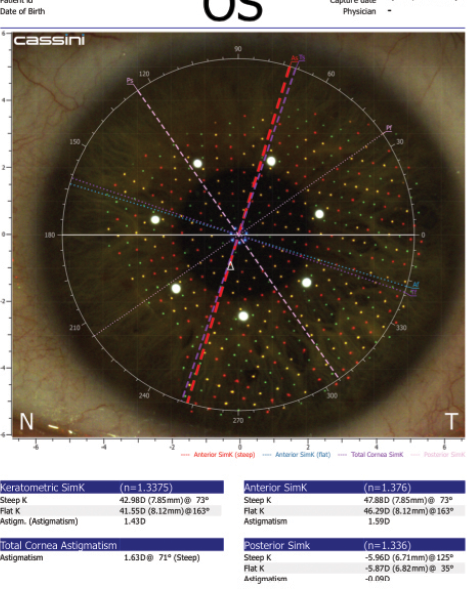
Figure. The Baylor nomogram would recommend undertreating this patient (with-the-rule astigmatism) based on anterior data alone. In contrast, the Cassini's TCA indicates a treatment plan correcting more astigmatism.
Integration
One of the factors that prompted me to add this device to my diagnostic package was its integration with my femtosecond laser. The new Streamline package uploads preoperative data from the Cassini directly to the Lensar Laser System (Lensar). Iris registration is then used to determine where to place incisions to reduce the amount of residual astigmatism. The Cassini also integrates with the TrueGuide Computer-Guided Surgery system (TrueVision Systems).
As an avid user of intraoperative aberrometry, I found it important to add a device that factors in posterior astigmatism to help confirm aberrometry measurements. In my experience, the Cassini's TCA measurement better matches the measurements from my ORA System (Alcon WaveTec) than do those obtained with my other diagnostic devices that only measure anterior data. This agreement gives me extra confidence at the outset of each case.
At a Glance
• Robust software programs built into the Galilei
produce clinically useful indices.
• The iTrace's toric planner includes unique eye
image overlays to assist with intraoperative axial
alignment.
• The new Streamline package uploads preoperative
data from the Cassini directly to the Lensar Laser
System.
• The Pentacam's Percentage Thickness Increase map
is particularly useful for detecting early keratoconus,
because it shows the change in thickness from the
center to the periphery of the cornea.
WEAKNESSES
Brightness
The light-emitting diode illumination during data capture is bright for patients who are light sensitive, which can affect the amount of coverage in each image. The company seeks to improve patients' experiences with the new “flash mode” functionality available with the latest software update. The Cassini will flash once the unit is in proper alignment, and I have seen a noticeable difference in my patients' level of comfort.
Compliance
As with any corneal topographic measurement, the Cassini requires patients to hold their eyelids open for a few seconds until the system is properly aligned. It can be difficult to capture measurements if patients have difficulty fixating. I sometimes find it necessary to hold open droopy eyelids to improve coverage by the system. Because of certain corneal issues, a small subset of patients does not have a visible second reflection off the posterior cornea, which means a TCA measurement is not attainable.
Posterior Elevation
Currently, the Cassini does not offer posterior elevation maps, which might be helpful to refractive and cornea surgeons.
CONCLUSION
Preoperative diagnostics are a critical aspect of today's premium practice. The Cassini's TCA combined with Lensar's Streamline integration are two exciting upgrades in my quest to improve patients' refractive outcomes.
1. Koch DD, Jenkins RB, Weikert MP, et al. Correcting astigmatism with toric intraocular lenses: effect of posterior corneal astigmatism. J Cataract Refract Surg. 2013;39(12):1803-1809.
2. Shimizu K, Misawa A, Suzuki Y. Toric intraocular lenses: correcting astigmatism while controlling axis shift. J Cataract Refract Surg. 1994;20(5):523-526.
3. Klijn S, Reus NJ, Sicam VA. Evaluation of keratometry with a novel color-LED corneal topographer. J Refract Surg. 2015;31(4)249-256.
Pentacam
By William B. Trattler, MD

Many patients with cataracts are interested in premium IOLs. The preoperative workup can determine whether or not they are candidates for these lenses as well as whether astigmatic keratotomy or limbal relaxing incisions would be beneficial and/or if patients are candidates for a PRK or LASIK enhancement for residual refractive error. Several technologies can help make the decision clearer, which is why upgrading to more advanced imaging systems is often warranted.
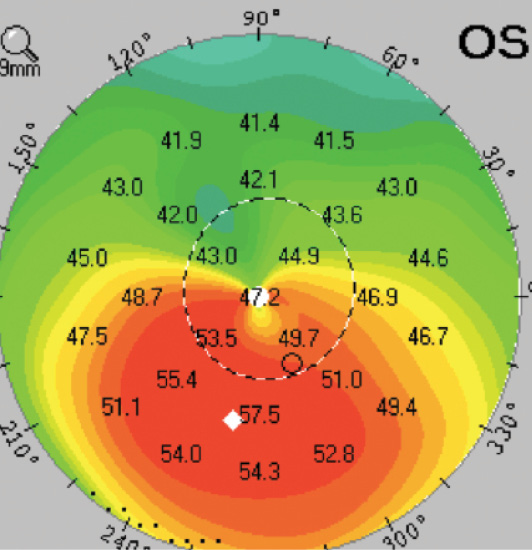
Figure 1. The Pentacam shows that the thinnest part of the cornea (marked by a white diamond) is decentered inferiorly. This case is consistent with a pellucid pattern form of keratoconus.
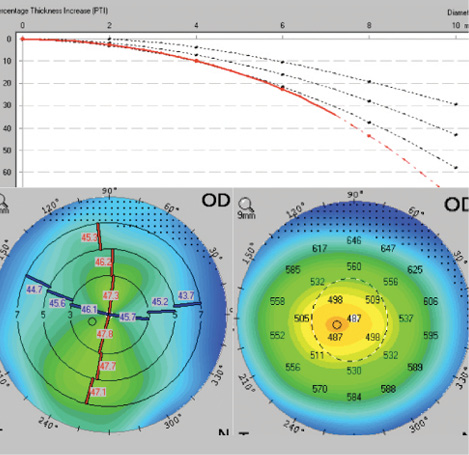
Figure 2. An eye with inferior steepening and a thin cornea. The red dashed line in the PTI map drops off the curve, meaning that the rate of change in corneal thickness from center to periphery is greater than normal.
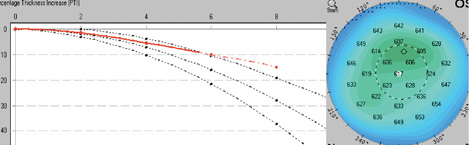
Figure 3. The PTI in an eye with Fuchs corneal dystrophy. The red dashed line in the PTI map stays above the curve, which means that the rate of change in corneal thickness from center to periphery is less than normal.
STRENGTHS
Both topography and tomography can be used (along with keratometry) to identify the magnitude and axis of astigmatism for surgical planning, but it is important to identify patients with mild pellucid marginal degeneration or mild keratoconus, because incisional methods for reducing astigmatism should be avoided in these cases. The Pentacam Comprehensive Eye Scanner (Oculus Surgical) can help. It not only shows the shape of the anterior and posterior cornea, but it can also identify whether the thinnest part of the cornea has decentered inferiorly, as occurs in early pellucid marginal degeneration (Figure 1).
The Pentacam's Percentage Thickness Increase (PTI) map is particularly useful for detecting early keratoconus, because it shows the change in thickness from the center to the periphery of the cornea. In ectatic corneas, such as those with keratoconus, the central cornea has thinned, but the peripheral cornea is unchanged (Figure 2). Conversely, in eyes with Fuchs corneal dystrophy, the central cornea is thicker than normal, so the percentage change from center to periphery is less than normal (Figure 3).
WEAKNESS
Multiple studies have found significant subjectivity in the interpretation of corneal maps, whether topography or tomography.1,2 Overall, however, these maps provide useful information for surgical planning. It is reasonable to expect that technologies such as the Pentacam will continue to evolve to enhance surgical outcomes and help ophthalmologists avoid treating patients who may have a reduced chance of achieving their desired outcome. n
1. Ramos IC, Correa R, Guerra FP, et al. Variability of subjective classifications of corneal topography maps from LASIK candidates. J Refract Surg. 2013;29(11):770-775.
2. Randleman JB, Akhtar J, Lynn MJ, et al. Comparison of objective and subjective refractive surgery screening parameters between regular and high-resolution Scheimpflug imaging devices. J Cataract Refract Surg. 2015;41(2):286-294.
Neel R. Desai, MD
• practices in the Tampa Bay region of Florida
• director of corneal refractive surgery and the Center for Ocular
Surface Disease at The Eye Institute of West Florida
• (727) 581-8706; desaivision@hotmail.com
• Financial disclosure: None acknowledged
Paul Mann, MD
• cataract and refractive surgeon at Mann Eye Institute and Laser
Center, Houston
• (713) 580-2568; paul.mann@manneye.com
• Financial disclosure: None acknowledged
P. Dee G. Stephenson, MD
• president, American College of Eye Surgeons
• private practice, Stephenson Eye Associates, Venice, Florida
• associate professor of ophthalmology, University of South Florida
College of Medicine, Tampa
• (941) 468-6363; eyedrdee@aol.com; Twitter @deestep03136405
• financial disclosure: on the speakers' bureaus of Bausch + Lomb,
i-Optics, and Lensar
William B. Trattler, MD
• director of cornea at the Center for Excellence in Eye Care in
Miami
• (305) 598-2020; wtrattler@earthlink.net; Twitter @wtrattler
• financial disclosure: speaker for Oculus


