In cataract surgery, the ultimate goal is also the biggest challenge: the removal of the natural lens through an opening called the capsulotomy, which is 50% smaller than the lens itself. Although a surgeon may approach each case with a plan for lens removal, it is often the lens itself that dictates how the procedure actually goes. Fortunately, using a femtosecond laser can help make the process far more predictable.
SUCCESSFUL LENS REMOVAL
Successful lens removal requires breaking the lens into smaller pieces, and there are several ways this can be done. In traditional cataract surgery, the lens is broken up manually after sculpting grooves with a phaco tip. A second instrument is often used to provide countertraction in order to crack or manually cut the lens. Lens fragments will flow more easily into the phaco tip if they are a predictable size and are removed in a specific order—the first and last piece typically being the most difficult to extract—but variables including lens density, depth, and the capsulotomy’s size will have an impact on the size of the lens fragments and how easy they are to extract.
FEMTOSECOND LASER
Removal of the lens nucleus can be compared to the demolition of a building. A wrecking ball will knock down the building in a somewhat random fashion, but a better option is to use strategically placed explosives to break apart key locations and let gravity do the rest. This more controlled approach creates smaller pieces that can be more easily stacked and removed. Watch the procedure here: eyetube.net/?v=arosi.
Using the femtosecond laser and patient-specific optical coherence tomography (OCT) imaging provides a similar opportunity by allowing surgeons to create strategic cuts and measurements to aid in lens removal. The laser can be used to create a capsulotomy of specific size and shape and to make precise cuts in the lens at 90% depth. Thus, during phacoemulsification, the nuclear pieces will crack into predictable pieces that allow consistent ease of removal.
Surgeons can also choose the size and quantity of central cylinders cut into the lens and cut within 500 µm of the posterior capsule based on the OCT images. A chop pattern may also be applied to divide the lens even further.
EFFECTIVE PATTERNS
In my experience, selecting four cylinders with the largest at 4.7 mm and a chop pattern of 5.3 mm is most effective. The central lens is typically the densest, with the peripheral lens’ being softer and requiring less energy to remove. By using the laser to break the central lens into 20 pieces, I can usually remove the densest part of the lens with less energy, with it remaining in the capsular bag and away from the cornea. With a cylinder bowled out to 4.7 mm in diameter, the remaining peripheral quadrants, each approximately 2.6 mm, can be removed inside the capsular bag. Since the laser will cut within 500 µm of the posterior capsule, it is unusual for a remaining epinuclear plate to impede cracking of the quadrants and moving them into this workspace (Figures 1 and 2).
Cylinders with a smaller diameter may not allow enough workspace for the surgeon to remove pieces in the capsular bag. For example, a cylinder of 3 mm in diameter will leave peripheral quadrants that are about 3.5 mm when cracked—too large to fit in the bowled-out area. These pieces will need to be removed at the iris plane, closer to the cornea, and are likely to require more energy to remove since they contain more of the dense central lens material.
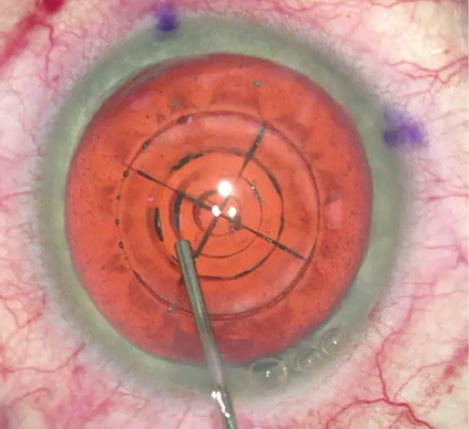
Figure 1. The femtosecond laser can create cylinders with a diameter set by the surgeon. The chop pattern often extends beyond the cylinder cuts and even beyond the capsulotomy. This cylinder size will have an impact on nuclear removal.
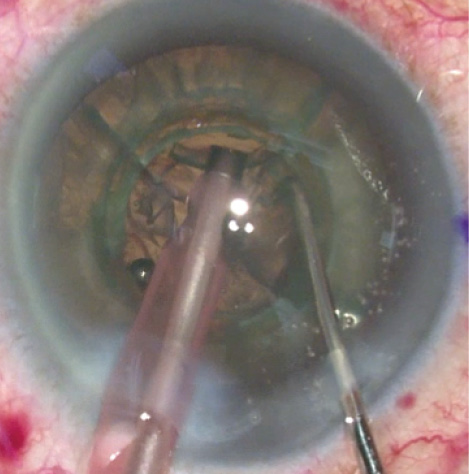
Figure 2. The densest part of the lens was easily removed using four cylinders, with the largest cylinder 4.7 mm, and a chop pattern of 5.3 mm. This bowled-out area created a workspace for the remaining four quadrants to stay in the capsular bag, away from the cornea, during the remaining nuclear removal.
A cylinder of larger diameter can also make it more difficult to remove the peripheral quadrants. For example, a 6-mm cylinder will leave soft peripheral quadrants that are about 2 mm in diameter, but the benefit of removing more of the nuclear lens in the bag is offset by the risk of a remaining nuclear lens that is too soft to crack into quadrants. In this case, the phaco tip is grabbing these small, soft peripheral pieces closer to the capsular bag. Also, if the capsulotomy is 5 mm, then work is being done in the bag under the anterior capsule, causing the surgeon to feel as though he or she is reaching for pieces. A smaller pupil would also be quicker to affect visualization of lens removal.
PHACO SETTINGS
Phaco settings can be adjusted to take full advantage of the benefits of using the femtosecond laser to cut pieces in the nuclear lens. I created a new setting for laser cataract surgery that uses high vacuum, no traditional phaco energy, and reduced linear-controlled torsional phaco energy. This allows me to remove the 4.7-mm core lens material, usually with very low energy and fluid exchange. An adjustment can also be made to the quadrant setting to use no traditional phacoemulsification and linear control of the torsional energy, because the quadrants are often not as dense compared to traditional cases.
At a Glance
• Using a femtosecond laser can help make the process of lens removal far more predictable.
• Lens fragments will flow more easily into the phaco tip if they are a predictable size and are removed in a specific order. In the author’s experience, selecting four cylinders with the largest at 4.7 mm and a chop pattern of 5.3 mm is the most effective.
• Phaco settings can be adjusted to take full advantage of the benefits of using the femtosecond laser to cut pieces in the nuclear lens. The author created a new setting for laser cataract surgery that uses higher vacuum, no traditional phaco energy, and reduced linear-controlled torsional phaco energy.
The integration of OCT imaging and the femtosecond laser into nuclear lens removal can change the way surgeons approach cataract surgery and have a positive impact on phaco energy and fluid exchange, while allowing the energy necessary to be farther away from the cornea. By creating predictably sized fragments and treating more than 90% of the lens depth, removal of the lens becomes safer and more routine.
CONCLUSION
Many surgeons agree that we are only at the tip of the iceberg with femtosecond laser technology, and that it will continue to evolve over time. Perhaps in the future, the capsulotomy will be a trapezoidal shape, and the lens will be cut into linear tubular shapes. There are endless possibilities and combinations to explore. Already, however, the femtosecond laser invites surgeons to think outside the box of traditional surgery to achieve greater precision and predictability. n
Scott E. LaBorwit, MD
• president of Select Eye Care in Towson, Maryland
• assistant professor, part-time faculty, at The Wilmer Eye Clinic, Johns Hopkins Hospital, Baltimore
• (410) 821-6400; sel104@me.com